-
工矿、印刷等行业的快速发展,大量含铅污染物被排放到水环境中,水质铅污染物检测及治理正成环境与材料领域的研究热点。传统的重金属检测技术主要包含质谱分析法[1]、原子吸收光谱测试法[2]、荧光分析法[3]和色谱分析法[4]等,但这些方法存在设备大、价格昂贵、操作复杂以及便携性差等局限性,限制了其在水环境中的实时监测应用。基于电化学方法的碳基传感器技术因具有便于携带、操作简单、灵敏度高、价格低廉等优势,在水质实时测定分析与治理方面具有极大优势[5]。作为一种新型介观碳材料,有序介孔碳因具有大的比表面积、有序的介孔结构、良好的导电性能,可望成为潜在的新型电极传感器材料。
最近,越来越多的研究[6-7]证实,通过氮磷等杂原子掺杂能在生物炭结构上形成更多的富电子边缘和空位缺陷等活性位和更高的极化度,进而增强材料对极性有机污染物和重金属的吸附固定;同时,改性掺杂的氮磷原子具有更高的电负性和更强的给电子能力,掺杂改性置入多孔碳材料的碳晶格中后,不仅能增强多孔碳材料的电子传递能力,并可以诱导与氮磷相邻的C原子或氧原子产生较大的自旋和电荷密度,作为氧化还原反应的活性位点,增强碳材料的氧化还原性能,从而提高其对检测物的循环伏安响应能力[8-9],如高杨等[10]研究表明,氮掺杂能明显增加石墨烯表面的活性点位,提升石墨烯的自由载流子密度和导电性能,从而增强其对硝酸根的检测性能。但也有研究表明,N/P共掺杂后多孔碳材料的氧化还原活性不仅没有提高,甚至降低[11-12],这可能是由于掺杂改性新生成的氮磷基团破坏或阻塞了多孔碳材料的微孔与介孔孔道,降低了氧化还原反应的有效活性位点和被检测物质在孔道中传递性能所致[13]。为此,本研究探索在氮磷双掺杂的同时,控制多孔碳材料的微中孔结构,规则的孔径结构与发达的孔隙率,使得氮磷等官能活性位点可以分散在不同长度的孔中并且增大了界面运输面积、提高电子转移速率,从而提升其电化学性能,保证制备的氮磷双掺杂多孔碳材料对水中痕量重金属铅的响应度与测定精确性。
在本研究中,以间苯二酚、甲醛和F127为碳源和模板剂制备出富含微孔的介孔碳,并使用生活中常见的磷氮双成份磷酸二氢铵为改性剂,在氮气气氛下,通过一步碳化实现碳材料的双重掺杂。在碳化改性过程中,磷酸二氢铵的分解可引起磷酸化反应,可有效减少碳材料中原有碳氧官能团的分解;同时,磷酸二氢铵中的氮和磷元素,在高温碳化的过程中与碳材料原有碳氧官能团结合,在碳骨架上生成大量活性氮磷结构,这将大大改善碳基材料在水性电解质中的润湿性;而且,磷酸二氢铵在高温下分解释放的氨气对多孔碳进行再次活化,可在制备碳材料上生成丰富的微孔结构。氮磷双掺杂、中孔结构的保持和微孔结构的增加,将增加氮磷掺杂介孔碳的有效活性点位,从而提升其对水中铅离子的识别敏感程度,实现对水中痕量铅离子的电化学检测。
-
间苯二酚、磷酸、无水乙醇、模板剂F127、盐酸、甲醛、磷酸二氢铵均为分析纯,购于南京化学试剂有限公司。主要仪器包括比表面及微孔径分析仪(3H-2000PM2,贝士德仪器科技有限公司)、管式炉(OTF-1200X,合肥科晶材料技术有限公司)、傅里叶变化红外光谱分析仪(Nicolet 6700,美国)、元素分析仪(Elementar Vario MICRO,德国)、透射电子显微镜 (JEM-2100F,日本)等。
-
将1.729 g磷酸(磷源)和3.3 g间苯二酚溶解在30 g质量分数50%的乙醇溶液中,加入3.0 g F127,搅拌至透明后,加入0.60 g盐酸,继续搅拌1 h,滴加3 g甲醛,继续搅拌1.5 h,将得到的混合物在室温下静置96 h,去除上层清液,用玻璃棒搅拌下层沉淀物,直至形成乳白色的粘稠物,将此黏稠物在室温静置48 h,先在80 ℃下固化48 h,而后在氮气保护下于500 ℃下恒温碳化3 h,冷却至室温,获得介孔碳OMC。将磷酸二氢铵与制得的OMC材料按2.3∶1的质量比溶于水,在室温下浸渍12 h,将所得到的混和材料在500 ℃恒温碳化1 h后自然降温,将得到的改性碳材料命名为OMC-MAP。
-
在进行玻碳电极预处理时,将直径3 mm的裸玻碳电极依次用粒径为1.0、0.3、0.05 μm的Al2O3粉在麂皮上抛光至洁净,每次抛光后先冲洗掉表面污物,再移入超声水浴中精洗2~3 min,重复3次,然后再依次用质量浓度为1∶1的乙醇、HNO3和蒸馏水超声清洗;最后将清洗干净的电极在0.1 mol·L−1 H2SO4溶液中,通过循环伏安法在0~0.6 V扫描10次进行活化备用。
在进行玻碳电极改性时,将50 mg过200目筛的改性介孔碳溶入5 mL 1%的Nafion溶液中,超声20 min获得均一的分散液;以微量注射器移取2.5 μL分散液,均匀地滴涂在预处理合格的裸玻碳电极表面,自然晾干后在45 ℃下固化15 min,得到OMC-MAP/GCE电极。
-
用贝士德微孔吸附仪测定碳样品在77.3 K下的氮吸附量,并通过Brunauer-Emmett-Teller (BET)、Horvath-Kawazoe(HK)和Barrett-Joyner-Halenda(BJH)理论计算材料比表面积、微孔孔容和中孔孔容,并通过Density functional theory(DFT)与BJH表征材料的全孔和介孔孔径分布特征;利用傅里叶变化红外光谱分析仪和X射线光电子能谱仪分析碳材料的表面化学官能团性质。
-
在(25±1) ℃下,在铂电极、饱和甘汞电极和氮磷介孔碳改性玻碳工作电极组成的三电极体系中,使用循环伏安法(CV)对水中痕量铅离子进行电化学检测,扫描区间为−1~0.2 V、采样间隔为0.001 V、静止时间为2 s、扫速为0.1 V·s−1,铅离子浓度为1 mg·L−1、底液浓度为0.2 mol·L−1。在优化实验中,探讨底液 (磷酸氢二钠盐-磷酸二氢钠、醋酸-醋酸钠、氯化铵-氨水)、pH(3.3~4.2)、富集电位(−1.4~−1.0 V)、富集时间(180~300 s) 对溶出电流的影响,进行优化时,除变量外,底液、pH、富集电位、富集时间设为醋酸-醋酸钠、3.8、240 s、−1.2 V和240 s。实际水样检测和抗干实验均在优化条件下测定。
-
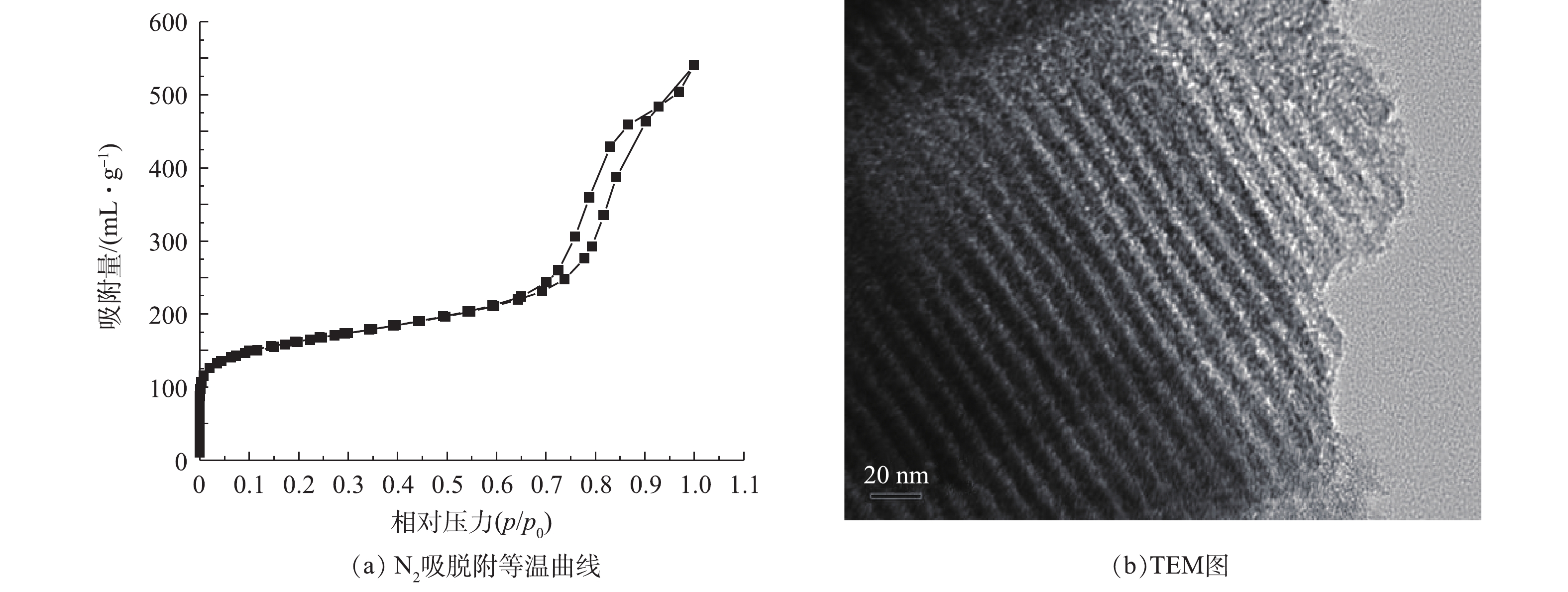
图1(a)为氮磷改性碳OMC-MAP的N2吸脱附等温曲线。在p/p0<0.01的低压区,介孔碳OMC-MAP的氮吸附量随着相对压力的增加而迅速增加,而后出现一缓慢吸附平台,这是由于微孔对氮气单分子层吸附达到饱和后,又出现了多层吸附引起的现象,这表明材料存在丰富的微孔结构,并含有一定量孔径接近于微孔的微中孔结构[14];当0.7<p/p0<0.98时,氮吸附等温线陡峭式的上升,发生毛细管凝聚,脱附线出现明显的滞后回线,这一吸附段的氮吸附量约为350 cm3·g−1,占总吸附量的60%,表明OMC-MAP材料存在大量孔径尺寸集中介孔结构。由图1(b)可见,在OMC-MAP的结构中,明显具有二维六方有序介孔结构的有序介孔碳。
图2为介孔碳OMC-MAP的DFT全孔与BJH介孔孔径分布。由DFT全孔分布(图2(a))可以看出,碳材料OMC-MAP的孔主要包括微孔和介孔,峰值孔径主要分布在0.813、1.178和7.45 nm处,且介孔分布集中于5~15 nm;由BJH介孔孔径分布(图2(b))也可以看出,OMC-MAP的介孔孔径主要分布在5~15 nm。根据氮等温吸附线数据,通过BET、HK和BJH可以计算出OMC-MAP的比表面积为579 m2·g−1,介孔孔容、微孔孔容和总孔孔容分别为0.681、0.232和0.835 cm3·g−1,介孔占总孔容的81.5%。结果表明:OMC-MAP材料是属于富含微孔和二维六方有序介孔结构的有序介孔碳,该孔结构特征有利吸附捕获溶液铅离子以及氧化还原反应产生的电子传递,从而增强该电极材料对水中痕量重金属离子的识别与测定。
-
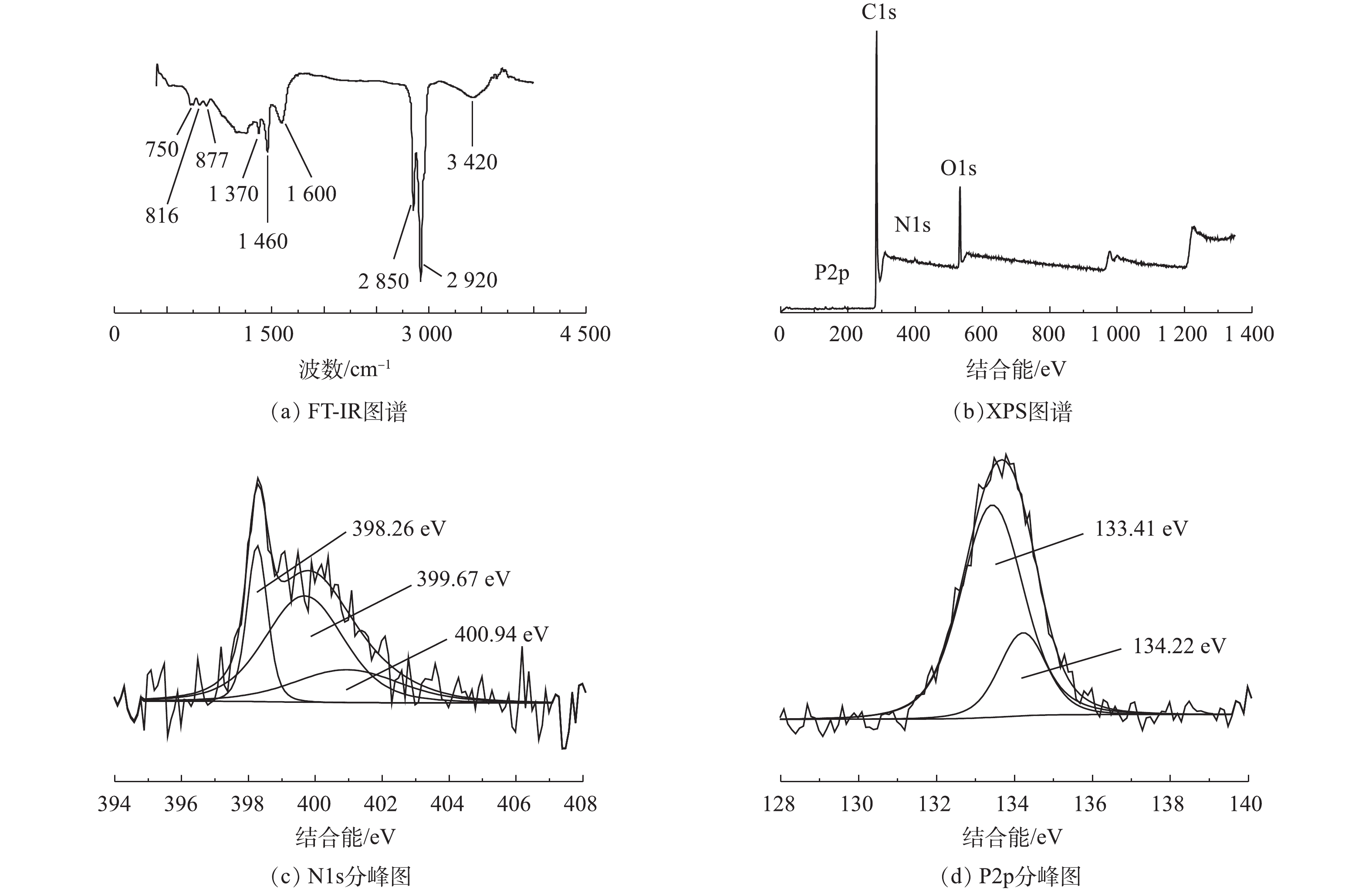
图3为碳材料FT-IR与XPS图谱。OMC-MAP的FT-IR红外光谱如图3(a)所示,可以看出OMC-MAP在750、816、877、1 370、1 460、1 600、2 850、2 920、3 420 cm−1处有吸收峰,1 460、2 850和2 920 cm−1处的吸收峰尖锐,750、816、870、1 370、1 600和3 420 cm−1处的峰谱带较宽。750 cm−1和816 cm−1处的吸收峰属于含磷官能团,其归因于P—C的伸缩振动[15]和P—O—C的对称伸缩振动[16];877、1 370和3 420 cm−1的吸附峰是由=C—H面外弯曲振动、—COOH拉伸振动和—OH伸缩振动所引起的[17-19];而1 600 cm−1吸附峰在双键区(2 000~1 500 cm−1),可能是由芳环与氧的其他不饱和体系发生共轭带而分裂出来的吸收带引起的[20]。以上结果表明:材料表面含氧官能团可能包含羟基、羧基、羰基及醌基等多种含氧酸基团;1 460 cm−1和2 850 cm−1是由氨基NH2的拉伸振动[21]和伯胺N—H键不对称伸缩[22]引起的吸收峰,这表示在介孔碳的表面含有氨基等含氮官能团。上述结果表明,介孔碳OMC-MAP的表面除含有羟、羧基等含氧酸基团外,还含有P—C、P—O—C、N—H、—NH2等含氮磷官能团。
XPS分析用于鉴定OMC-MAP的表面元素组成和化学结构。图3(b)为OMC-MAP的XPS光谱。可以看出,图谱中显示出O1s(532 eV)、C1s(284 eV)、P2p(135 eV)和N1s(399 eV)峰,其所对应的原子含量分别为85.7%、11.0%、1.75%和0.79%,这进一步说明改性样品成功掺入了N、P元素。为了进一步验证N、P元素的状态,N1s图谱可以解卷积成3个不同的分量,由图3(c)可见,其峰值分别在398.26、399.67、400.94 eV,这可归因于吡啶氮[23],吡咯氮[24]和氨基(—NH2)[25];P2p峰的精细结构表明样品存在2个主要的含磷基团,如图3(d)所示,峰值为134.22 eV表明其含有P—O—C基团[26],而峰值在133.41 eV附近可归因于P—C键[27]。这与FT-IR的分析结果保持一致。
-
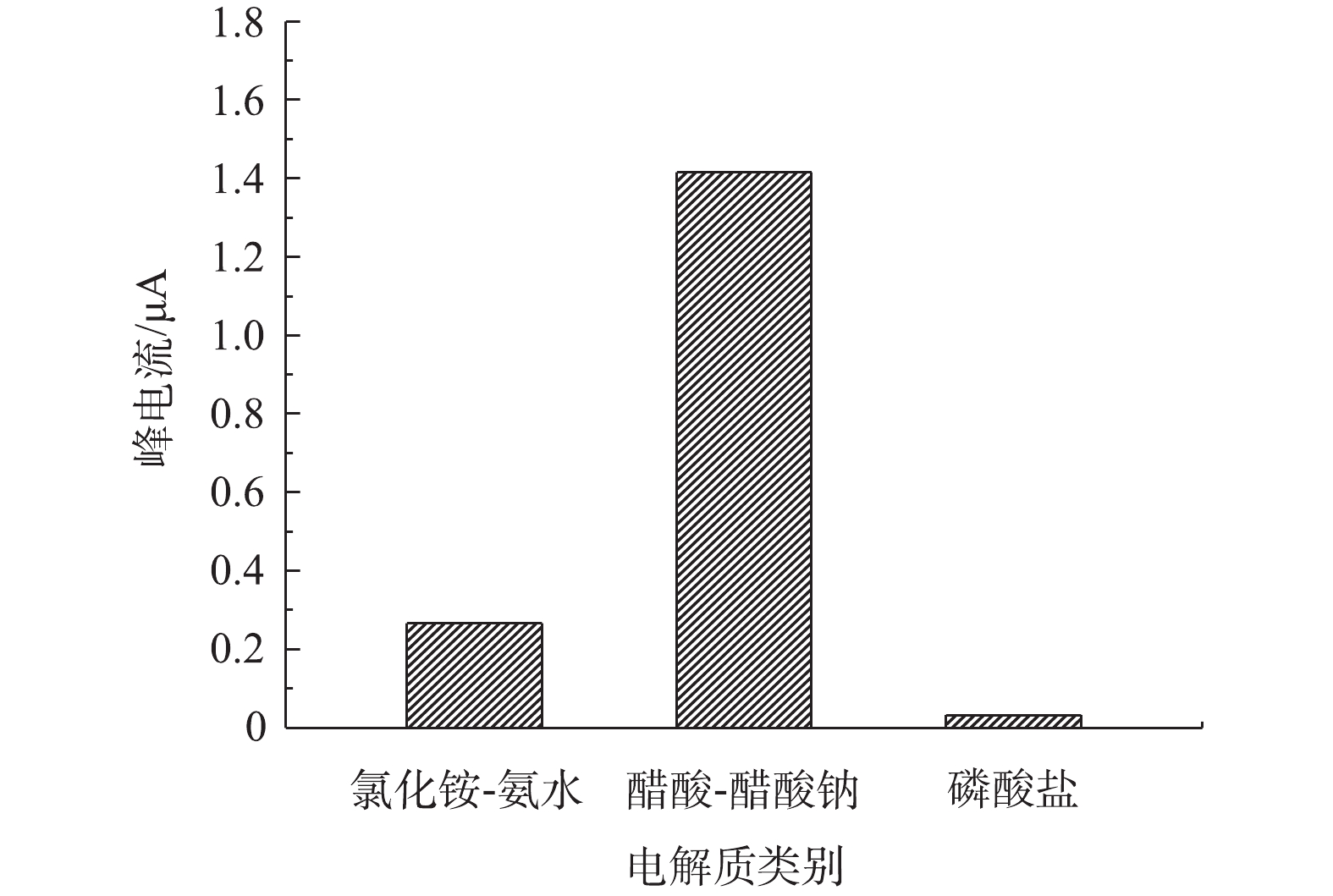
1)不同支持电解质底液对改性电极检测痕量铅的影响。由图4可知,在以磷酸氢二钠盐-磷酸二氢钠、醋酸-醋酸钠、氯化铵-氨水3种缓冲液作为支持电解质底液时,支持电解质底液类别对改性电极检测铅离子的性能具有较大影响。此外,当醋酸-醋酸钠作为支持电解质底液时,改性电极对水中铅离子的溶出电流峰值最大。这主要是由于醋酸-醋酸钠和介孔碳改性电极对目标铅离子的还原均可能产生较强的催化作用,而且醋酸也可与铅离子发生配位作用,从而成为铅离子的扩散载体,这2个方面的原因均会导致扩散速度和峰电流增大。李静等[28]比较了Na2SO4、KCl、Na2HPO4-NaH2PO4和醋酸-醋酸钠等作为支持电解质溶液对铋改性碳电极测定水中痕量铅离子的影响,也得到了相似的结论,即以醋酸-醋酸钠缓冲液作为支持电解质底液时,改性电极对铅检测响应敏感度最优。为此,在后续实验中选用醋酸-醋酸钠缓冲液作为OMC-MAP改性电极铅检测的支持电解质底液。
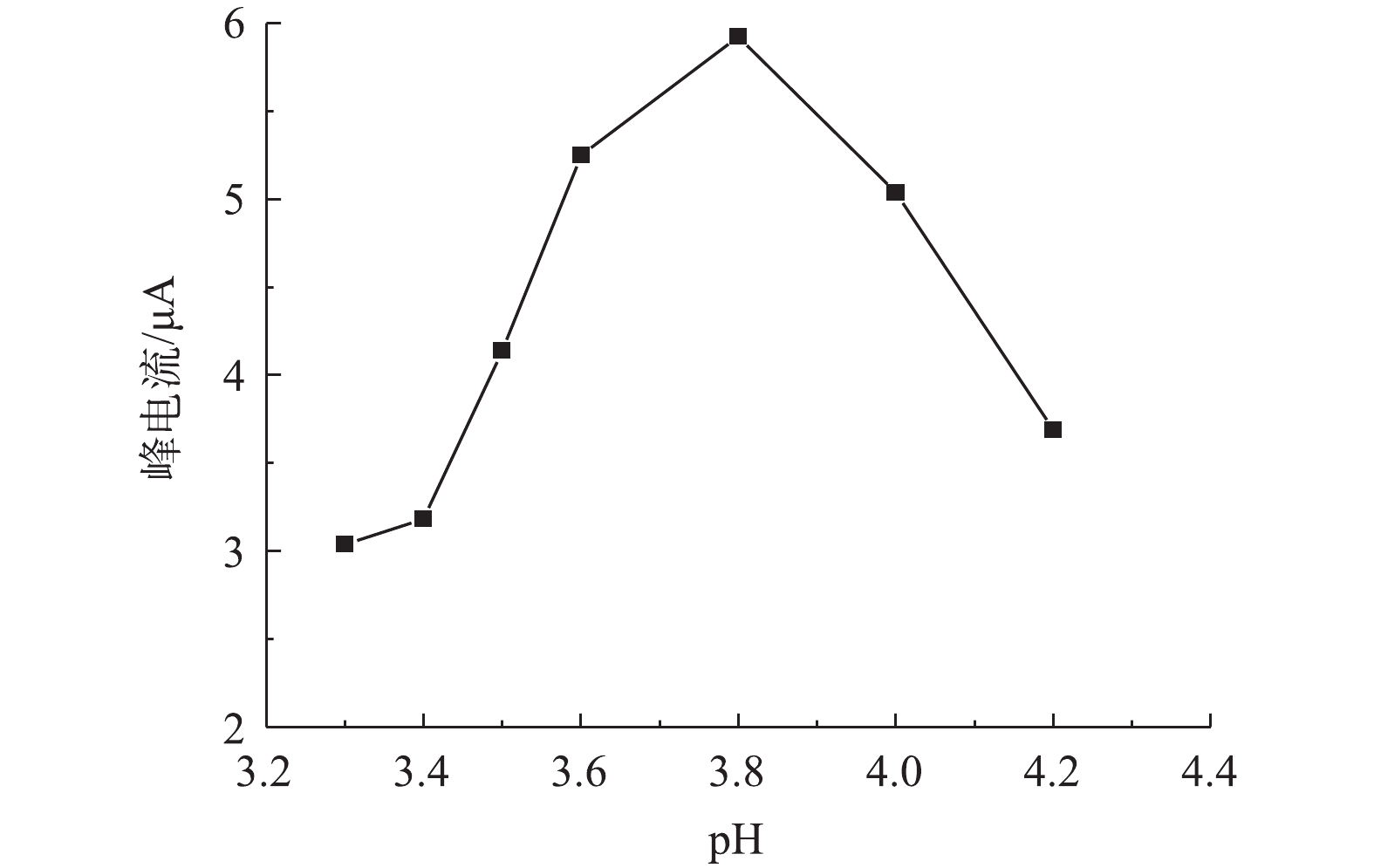
2)溶液pH对改性电极测定痕量铅的影响。由图5可知,当溶液pH由3.3增加4.2时,改性电极的铅溶出电流先增大后减小;在溶液pH=3.8时,改性电极对铅检测的峰电流值最大,说明溶液pH对改性电极检测铅有较大影响,这主要是由于改性电极表面修饰的介孔碳材料表面富含氮磷官能团,在不同pH下,氮磷官能团质子化程度发生变化,如吡咯或吡啶上的氮孤对电子在不同酸性条件下质子化程度不同,因此,其所表现出来的电性不同,导致与溶液中铅离子的静电或化学作用也有所不同,进而影响铅离子在改性电极表面结合能力和结合量,最终导致循环伏安电流变化。戴兴欣[29]在研究氨基改性对介孔硅电极测定铅的影响时,也发现溶液pH对氨基改性电极测定铅有较大影响。在对于本研究中OMC-MAP改性的玻碳电极测定溶液中铅,在pH为3.8时峰电流最强,在后续研究中选择其为最优pH。
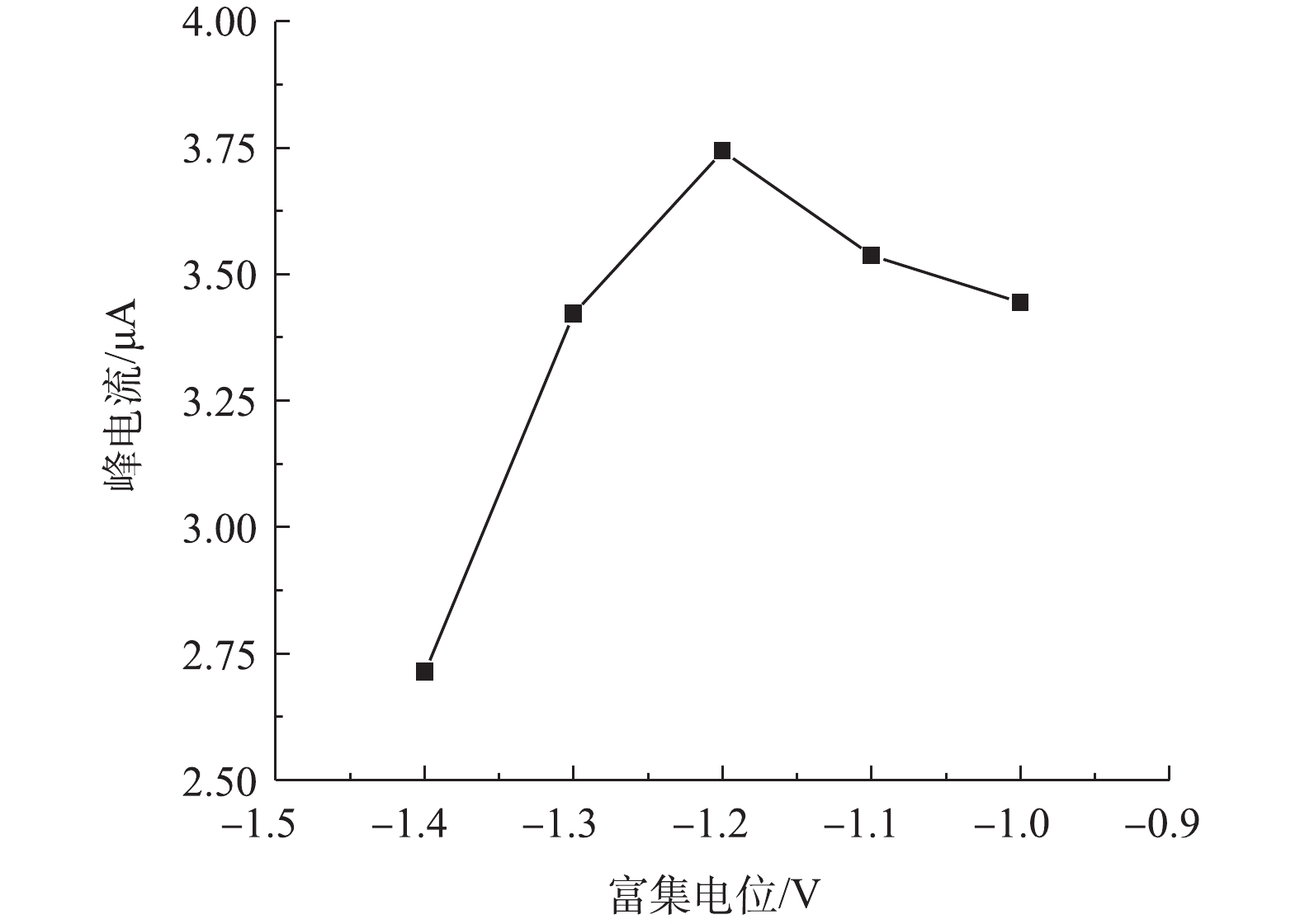
3)富集电位对改性电极检测痕量铅的影响。由图6可见,改性电极对铅的响应峰电流随着富集电位的升高先升高而后降低,当富集电位为−1.2 V时,峰电流达到最大值。当富集电位由−1.0 V降到−1.2 V时,随着富集电位绝对值的增大,峰电流增大,这可能是高电位下水中铅离子在改性电极上的还原更加充分所致;当富集电位由−1.2 V降到−1.4 V时,响应峰电流降低,这可能是高电位下溶液发生了析氢,电位越高,析氢增大,而氢离子的析出阻碍了铅离子在电极表面的沉积[30],进而导致电化学还原阶段峰电流的降低。
4)富集时间对改性电极测定铅的影响。由图7可见,当富集时间为240 s时,改性电极对铅的响应溶出电流达最高。当富集时间小于240 s时,铅离子在改性电极上的富集不完全,未达到最大值,因而响应溶出电流值较低。当富集时间大于240 s时,溶出峰电流随着富集时间的增加非但没有进一步增加,并且还有所降低,这可能归因于改性电极对铅离子的还原富集在240 s时已达最高值,进一步增加富集时间,改性电极对铅离子的还原富集量因活性位点饱和不再增加,反而导致已还原的铅与改性介孔碳表面氮磷官能团等活性位结合成了更稳定的化学物,阻碍电子传输,使得在随后的氧化溶出时变得困难,从而导致检测响应溶出峰电流降低。尽管不少研究表明,改性电极对铅检测的响应峰电流随富集时间的增加一直增加,达到一峰值后峰响应电流不变,但也有研究[31-32]得出与本研究相类似结果,这说明在高活性官能团改性电极测定金属离子时,优化控制富集时间对提高电极敏感性有重要的意义。
-
不同浓度铅离子在改性电极上的溶出伏安图及峰电流与铅离子浓度之间的线性关系见图8。图8(a)为OMC-MAP改性玻碳电极在醋酸-醋酸钠作为支持电解质底液、pH=3.8、富集电位−1.2 V、富集时间280 s的优化条件下对水中1~10 000 µg·L−1中10个不同浓度的铅离子的溶出响应伏安电流图。可以看出,响应电流峰值随着铅离子浓度的增大而增大。对响应电流值与铅离子浓度作线性相关分析可知,溶出响应电流与铅离子的浓度间呈两段线性相关(图8(b)),在铅离子为1~150 µg·L−1和150~10 000 µg·L−1时,对应的拟合方程为y=0.022 1x+1.798 71和y=0.004 8x+4.676 79,R2均大于0.98。每次测定并加相反电压后,在空白底液中经过循环伏安扫描除去电极表面的铅,以恢复电极活性,对2.18 µg·L−1的铅溶液平行测定5次的相对标准偏差为2.16%,这表明电极有良好的重现性。上述结果表明,经氮磷改性介孔碳OMC-MAP修饰的改性电极,对水中的痕量铅有良好的响应度和稳定性。
本实验的检测方法同其他文献报道出来的检测方法相比,如表1所示,具有更宽的线性范围及较低的检测限,说明氮磷改性介孔碳作为玻碳电极的修饰材料,无论在线性范围以及检测限方面都有非常大的优势,对水中的痕量铅有良好的响应度与灵敏度。
-
在最优实验条件下,使用OMC-MAP改性玻碳电极5次重复测定长江实际取水水样中的Pb2+浓度,测定结果分别为2.08、2.12、2.04、2.13和2.11 μg·L−1,平均值为2.096 µg·L−1。使用ICP-AES法检测实际长江水样的Pb2+离子浓度为2.13 μg·L−1。OMC-MAP改性玻碳电极与ICP-AES 2种方法检测的Pb2+离子浓度的相对偏差为3.85%,表明本方法具有可靠性,其可用于实际环境水样中Pb2+浓度的检测。在长江水样中加入100倍的Ca2+、Na2+、Mg2+干扰物质后,在优化的实验条件下,对OMC-MAP改性玻碳电极检测长江水中Pb2+的浓度并无影响,这说明OMC-MAP改性玻碳电极具有良好的选择性。
-
1)以磷酸二氢铵为磷氮改性剂、间苯二酚和F127为碳源和模板剂成功制备了具有丰富微中孔结构的磷氮双改性介孔碳OMC-MAP,材料的介孔、微孔孔容分别为0.681 cm3·g−1和0.232 cm3·g−1。
2) FT-IR与XPS表征结果表明,OMC-MAP介孔孔道镶嵌有—NH2、N—H、P—C、P—O—C等氮磷官能团以及C=O、C—O、COOH等含氧官能团。
3)在以醋酸-醋酸钠支持电解质底液、pH=3.8、富集电位为−1.2 V和富集时间为240 s的条件下,当线性范围为1~10 000 µg·L−1时,循环伏安溶出响应电流与对应的铅离子呈良好的两段线性相关关系,线性相关度R2>0.98,对应的检测限为0.2 µg·L−1,这表明电极对水中的痕量铅有良好的响应度和灵敏度。
氮磷双改性酚醛树脂基有序介孔碳电极对水中痕量铅的测定
Determination of trace lead in water by nitrogen and phosphorus double modified phenolic resin-based ordered mesoporous carbon electrode
-
摘要: 以常见的磷氮双成份磷酸二氢铵为改性剂,间苯二酚/甲醛和F127为碳源和模板剂,成功制备了对痕量铅具有高灵敏度的氮磷双改性介孔碳OMC-MAP改性玻碳电极。通过N2吸附脱附等温线、FT-IR、XPS和阳极溶出伏安法对OMC-MAP的物化性质及其改性玻碳电极电化学性能进行了表征和分析。结果表明:OMC-MAP具有较高的孔容(0.835 mL·g−1)、比表面积(579 m2·g−1)和丰富的氨基、羧基、羰基等氮氧官能团,以及P—C、P—O—C等含磷官能团,介孔主要分布在5~10 nm区域,峰值在7.45 nm;OMC-MAP良好的介孔结构及其分散在微介孔表面的氮、磷、氧活性官能团为其改性玻碳电极传感器提供了良好的电子传递通道和高识别铅离子的活性位点。在醋酸-醋酸钠支持电解质底液下,OMC-MAP介孔碳改性玻碳电极传感器对溶液铅显示极优的电催化还原活性,当pH=3.8、富集电位为−1.2 V和富集时间为240 s时,循环伏安溶出电流的响应值达到最大。在该条件下,OMC-MAP改性玻碳电极在1~10 000 µg·L−1宽范围内对铅离子均表现出极优的响应性,R2>0.98,灵敏度高、检测范围广,说明OMC-MAP是一种潜在痕量铅的电极材料。Abstract: N/P co-doped OMC-MAP modified glassy carbon electrode with high sensitivity for lead ions was prepared when common ammonium dihydrogen phosphate, formaldehyde/resorcinol and F127 were taken as modifier, carbon source and template agent of OMC-MAP, respectively. N2 adsorption-desorption isotherm, FT-IR, XPS and anodic stripping voltammetry were used to characterize the physicochemical properties of OMC-MAP and electrochemical performance of modified glassy carbon electrode. The results showed that OMC-MAP had high pore volume (0.835 mL·g−1) and large specific surface area (579 m2·g−1), which contained amino nitrogen, carboxyl, carbonyl and other nitroxide functional groups, as well as P—C, P—O—C and other phosphorus functional groups. The mesopores were mainly distributed in the region of 5~10 nm, with a peak at 7.45 nm. The great mesoporous structure of OMC-MAP and the active functional groups of nitrogen, phosphorus and oxygen dispersed on the surface of the micro-mesopores provided a good electron transfer channel and active sites for high lead ions identification. Under the condition of acetic acid-sodium acetate supporting electrolyte solution, modified glassy carbon electrode sensor of OMC-MAP showed excellent electrocatalytic reduction activity on lead solution. When pH was 3.8, enrichment potential was −1.2 V and enrichment time was 240 s, the cyclic voltammetric stripping current response value reached the maximum. Under this condition, the OMC-MAP modified glassy carbon electrode showed excellent responsiveness and regularity to lead ions within a wide range of 1~10 000 µg·L−1. The linear correlation of R2 was higher than 0.98, with high sensitivity and wide detection range. This indicated that OMC-MAP modified glassy carbon electrode was a potential electrode material for trace lead ions detection.
-

-
-
[1] BLAIR S M, BRODBELT J S, MARCHAND A P, et al. Evaluation of binding selectivities of aged crown ligands toward heavy metals by electrospray ionization/quadrupole ion trap mass spectrometry[J]. Analytical Chemistry, 2000, 72(11): 2433-2445. doi: 10.1021/ac991125t [2] BAZZI A, KREUZ B, WUOKILA J, et al. Separation and determination of Cr(III) and Cr(VI) with cation-exchange chromatography and atomic absorption spectroscopy. An experiment for quantitative methods of analysis[J]. Journal of Chemical Education, 2005, 82(3): 435-438. doi: 10.1021/ed082p435 [3] WANG L, XIA T, LIU J, et al. Preparation and application of a novel core/shell organic nanoparticle as a fluorescence probe in the selective determination of Cr(VI)[J]. Spectrochimica Acta Part A: Molecular & Biomolecular Spectroscopy, 2005, 62(1): 1386-1425. [4] WILLIAMS T, JONES P, EBDON L. Simultaneous determination of Cr(III) and Cr(VI) at ultratrace levels using ion chromatography with chemiluminescence detection[J]. Journal of Chromatography A, 1989, 482(2): 361-366. doi: 10.1016/S0021-9673(01)83924-8 [5] 袁彩霞. 碳基材料电化学传感研究[D]. 兰州: 西北师范大学, 2014. [6] FUJITA S I, WATANABE H, KATAGIRI A, et al. Nitrogen and oxygen-doped metal-free carbon catalysts for chemoselective transfer hydrogenation of nitrobenzene, styrene, and 3-nitrostyrene with hydrazine[J]. Journal of Molecular Catalysis A: Chemical, 2014, 393: 1381-1169. [7] LIU N, DING L, LI H, et al. N-doped nanoporous carbon as efficient catalyst for nitrobenzene reduction in sulfide-containing aqueous solutions[J]. Journal of Colloid and Interface Science, 2017, 490: 677-684. [8] QIAO X, PENG H, YOU C, et al. Nitrogen, phosphorus and iron doped carbon nanospheres with high surface area and hierarchical porous structure for oxygen reduction[J]. Journal of Power Sources, 2015, 288: 253-260. doi: 10.1016/j.jpowsour.2015.04.118 [9] ZHANG Z A, LAI Y Q, LI J, et al. Electrochemical behavior of wound supercapacitors with propylene carbonate and acetonitrile based nonaqueous electrolytes[J]. Journal of Central South University of Technology, 2009, 16(2): 247-252. doi: 10.1007/s11771-009-0042-2 [10] 高杨, 岳荣, 鲁青, 等. 基于聚吡咯纳米线修饰玻碳电极的硝酸根电流型传感器研究[J]. 分析科学学报, 2012, 28(5): 6-10. [11] DUAN X, INDRAWIRAWAN S, SUN H, et al. Effects of nitrogen-, boron-, and phosphorus-doping or codoping on metal-free graphene catalysis[J]. Catalysis Today, 2015, 249: 84-191. [12] YANG L, JIANG S, ZHAO Y, et al. Boron-doped carbon nanotubes as metal-free electrocatalysts for the oxygen reduction reaction[J]. Angewandte Chemie International Edition, 2011, 50(31): 7132-7135. doi: 10.1002/anie.201101287 [13] PEREIRA L, PEREIRA R, PEREIRA M F, et al. Effect of different carbon materials as electron shuttles in the anaerobic biotransformation of nitroanilines[J]. Biotechnology and Bioengineering, 2016, 113(6): 1194-1202. doi: 10.1002/bit.25896 [14] 李坤权, 李烨, 郑正, 等. 富含中孔与酸性基团的生物质炭的制备与吸附性能[J]. 环境科学, 2013, 34(6): 2479-2485. [15] WU X, ZHAO W, WANG H, et al. Enhanced capacity of chemically bonded phosphorus/carbon composite as an anode material for potassium-ion batteries[J]. Journal of Power Sources, 2018, 378: 460-467. doi: 10.1016/j.jpowsour.2017.12.077 [16] LIU Y, GONG X, DONG W, et al. Nitrogen and phosphorus dual-doped carbon dots as a label-free sensor for curcumin determination in real sample and cellular imaging[J]. Talanta, 2018, 401: 330-354. [17] 翁诗甫. 傅里叶变换红外光谱分析[M]. 北京: 化学工业出版社, 2010. [18] PARK J A, JUNG S M, YI I G, et al. Adsorption of microcystin-LR on mesoporous carbons and its potential use in drinking water source[J]. Chemosphere, 2017, 177(6): 15-23. [19] YANG H, LI S, CHEN J, et al. Adsorption of Pb(II) on mesoporous activated carbons fabricated from water hyacinth using H3PO4, activation: Adsorption capacity, kinetic and isotherm studies[J]. Applied Surface Science, 2014, 293: 160-168. doi: 10.1016/j.apsusc.2013.12.123 [20] TORRELLAS S A, LOVERA R G, ESCALONA N, et al. Chemical-activated carbons from peach stones for the adsorption of emerging contaminants in aqueous solutions[J]. Chemical Engineering Journal, 2015, 279: 788-798. doi: 10.1016/j.cej.2015.05.104 [21] LI K, CAO J, LI H, et al. Nitrogen functionalized hierarchical microporous/mesoporous carbon with a high surface area and controllable nitrogen content for enhanced lead(II) adsorption[J]. RSC Advances, 2016, 6: 9218-92196. [22] WAN Z, LI K. Effect of pre-pyrolysis mode on simultaneous introduction of nitrogen/oxygen-containing functional groups into the structure of bagasse-based mesoporous carbon and its influence on Cu(II) adsorption[J]. Chemosphere, 2017, 194: 370-389. [23] YAN X, LIU Y, FAN X, et al. Nitrogen/phosphorus co-doped nonporous carbon nanofibers for high-performance supercapacitors[J]. Journal of Power Sources, 2014, 248: 745-751. doi: 10.1016/j.jpowsour.2013.09.129 [24] XIAO L, XU H, ZHOU S, et al. Simultaneous detection of cd(II) and Pb(II) by differential pulse anodic stripping voltammetry at a nitrogen-doped microporous carbon/Nafion/bismuth-film electrode[J]. Electrochimica Acta, 2014, 143: 143-151. doi: 10.1016/j.electacta.2014.08.021 [25] JOSE P, JUAN J, JUANA M, et al. Selective nitrogen functionalization of phosphorus-containing activated carbons[J]. Fuel Processing Technology, 2017, 156: 236-241. [26] YAN X, YU Y, RYU S K, et al. Simple and scalable synthesis of phosphorus and nitrogen enriched porous carbons with high volumetric capacitance[J]. Electrochimica Acta, 2014, 136: 466-472. doi: 10.1016/j.electacta.2014.05.031 [27] LIN Y, ZHANG J, PAN Y, et al. Nickel phosphide nanoparticles decorated nitrogen and phosphorus co-doped porous carbon as efficient hybrid catalyst for hydrogen evolution[J]. Applied Surface Science, 2017, 422(15): 828-837. [28] 李静, 王文成, 范钦莉, 等. 铋膜修饰碳离子液体糊电极测定痕量铅离子的研究[J]. 海南师范大学学报(自然科学版), 2015, 28(4): 400-403. [29] 戴兴欣. 氨基改性介孔硅修饰电极在检测重金属离子中的应用[D]. 苏州: 苏州大学, 2015. [30] 童基均, 汪亚明, 黄文清, 等. 基于平面印刷碳电极的重金属离子检测[J]. 传感技术学报, 2004, 17(1): 22-25. doi: 10.3969/j.issn.1004-1699.2004.01.006 [31] 王栋萍. 类石墨相氮化碳电极材料的制备及其在铅离子检测中的应用研究[D]. 广州: 华南理工大学, 2013. [32] 于璐洋, 王会才, 赵修青, 等. 巯基功能化石墨烯修饰玻碳电极测定水中痕量重金属镉[J]. 天津工业大学学报, 2014, 33(3): 34-39. doi: 10.3969/j.issn.1671-024X.2014.03.007 [33] SLAVEC M, HOEVAR S B, BALDRIANOVA L, et al. Antimony film microelectrode for anodic stripping measurement of cadmium(II), lead(II) and copper(II)[J]. Electroanalysis, 2010, 22(14): 1617-1622. doi: 10.1002/elan.200900583 [34] DAI X, QIU F, ZHOU X, et al. Amino-functionalized MCM-41 for the simultaneous electrochemical determination of trace lead and cadmium[J]. Electrochimica Acta, 2014, 144: 161-167. doi: 10.1016/j.electacta.2014.08.093 [35] KEFALA G, ECONOMOU A. Polymer-coated bismuth film electrodes for the determination of trace metals by sequential-injection analysis/anodic stripping voltammetry[J]. Analytica Chimica Acta, 2006, 576(2): 283-289. doi: 10.1016/j.aca.2006.06.006 -



 下载:
下载: