-
世界卫生组织(World Health Organization,WHO)指出氟存在于水中会导致大规模健康问题。适量的氟有助于人类的健康,但过量氟摄入会导致骨质疏松、脑损伤等许多疾病。WHO建议饮用水中氟浓度应低于1.5 mg·L−1;我国规定工业废水中F−浓度应低于1 mg·L−1(GB 5749-2006),饮用水中F−浓度应低于1 mg·L−1。在自然界中,矿石中的氟元素通过地质风化作用释放到地下水中,但人类活动已经成为更加主要的氟污染来源。
煤炭是我国的主要能源,每年相关产业会产生约3×108 t焦化废水[1]。焦化废水组分十分复杂,其中就包括高浓度的氟及难降解的有毒有机物,如喹啉、吡啶、吲哚等[2-4]。除了焦化废水,玻璃陶瓷工艺、半导体制造、电镀等工业废水中也含有高浓度的氟。焦化废水的处理主要采用活性污泥法,如A/O或A/A/O法。活性污泥法具有成本低、运行和维护简单等优点[3, 5]。然而,由于焦化废水中还存在大量难生物降解的有机物,活性污泥法出水往往难以达到我国的焦化废水排放标准(GB 16171-2012)。因此,需要进一步处理相关工艺出水。
目前,絮凝沉淀[6-7]、吸附[2]、高级氧化[4, 8-10]、膜滤[11-12],以及这些方法的组合[13]都已被应用于焦化废水的处理。在这些方法当中,絮凝沉淀具有操作简单、去除效率高、成本低等优势,所以受到广泛关注。采用聚合硫酸铁絮凝和聚酰胺-胺(poly(amidoamine),PAMAM)树形分子改良的粉末活性炭吸附结合的方法可以去除焦化废水中85.3%的COD[6];采用聚合氯化铝进行絮凝预处理可以有效降低焦化废水中的污染物浓度,从而降低后续膜蒸馏过程中的膜污染发生的可能性[11]。化学沉降是另一种常见的除氟的方法,常采用的沉降剂有Ca2+、Mg2+、Al3+等金属离子。Ca2+可以通过与F−形成氟化钙沉淀从废水中去除氟,最适宜的pH为9~10。但是这种方法最主要的缺点是CaF2颗粒非常细,还需要采取其他的方法提高其沉降性能,如加入晶种[14]或采用膜滤的方法[15]。但目前少有研究考查絮凝与化学沉降方法结合的去除效果。
本研究考察了2种絮凝剂对有机物和F−的去除效果,并探究了Ca2+对去除效果的影响。采用铁铝复配的絮凝剂以提高絮凝的效率,通过研究FeCl3和AlCl3的比例,以及pH对去除效率的影响,实现了FeCl3和AlCl3协同絮凝去除焦化废水中氟和难降解有机物。
全文HTML
-
原水取自永鑫焦化废水处理厂的活性污泥工艺出水。原水的TOC为(99.8±7.0) mg·L−1,F−浓度为(90.5±2.5) mg·L−1,pH为8.02±0.06。图1为原水的三维荧光图,峰值表示可溶性微生物副产物[5]。所用试剂包括AlCl3、FeCl3、NaOH、HCl、CaCl2,均为分析纯。
-
在室温((25±1)℃)下,将原水溶液在50 r·min−1下搅拌60 s;之后加入不同浓度的絮凝剂,并加入0.1 mol·L−1的HCl或NaOH调节原水的pH;再将转速调到200 r·min−1,搅拌20 min;反应结束后,分别收集上清液和絮体进行后续的检测。
-
F−浓度采用GB 7484-1987中的F−电极选择法进行测量。根据GB 7484-1987中的方法,配置测F−专用的总离子强度缓冲液TISAB Ⅱ。上清液经过0.22 μm滤膜过滤之后,取2.5 mL加入37.5 mL去离子水中,再加入10 mL TISAB Ⅱ。混合液在磁力搅拌器上搅拌均匀,用F−选择电极测量电位。通过事先测定的电位-F−浓度标准曲线确定溶液中F−浓度。
收集的絮体经过冷冻干燥后,采用傅里叶变换红光谱(NICOLET 8700,Thermal,USA)进行分析;分别采用紫外-可见光谱(UV-2600,Shimadzu,Japan)和TOC分析仪(TOC-VCPH,Shimadzu,Japan)测量溶液在254 nm的吸光度和DOC;溶液中具有紫外活性有机物的分子质量分布采用装有UV254吸收检测器的凝胶色谱分析。10 mmol·L−1醋酸钠(Aldrich,USA)作为流动相,流速为1 mL·min−1。待测样注射体积为100 μL,紫外-可见分析器检波长为254 nm。检测前,以2 mL·min−1的流速注入流动相,以清除残余的污染物。用分子质量为33 500、14 900、6 530和1 100 Da的聚苯乙烯(polystyrene sulfonate,PSS)标准物(American Polymer Standard Corp.,U.S.)校准化合物分子质量与停留时间的关系。所有样品在检测前都经过0.22 μm的滤膜过滤。
1.1. 实验原料
1.2. 实验方法
1.3. 分析方法
-
采用AlCl3和FeCl3研究了不同絮凝剂去除有机物和氟的效率和作用机理,结果如图2所示。由图2(a)可知,AlCl3在氟去除方面明显优于FeCl3。随着AlCl3投加量从1 mmol·L−1升至20 mmol·L−1,原水中F−的浓度从92.8 mg·L−1降至5.2 mg·L−1,AlCl3投加量为20 mmol·L−1时达到最高的氟去除率94.4%。然而,对于FeCl3来说,最高的F−去除率仅为27.3%。其可能的反应机理为:当Al3+和Fe3+投加到水中时,会水解形成金属氢氧化物;金属氢氧化物可以通过库仑引力或配体交换作用吸附焦化废水中的F−;对于金属离子M,当pH < pHpzc时,库仑引力和配体交换同时作用,则会发生反应(式(1)和式(2))。而当pH > pHpzc时,吸附的主要原理是配体交换,即发生了反应(式(3))。
金属铝的pHpzc高于金属铁的pHpzc,因此,在中性pH下AlCl3的F−去除率比FeCl3的要高。该结论与之前的研究结果[16]一致,且F−可以与Al3+形成AlFn (n = 1~6)络合物,并且进一步与羟基形成三级络合物Al(OH)mFn (m + n ≤ 4)[17]。
Al3+和Fe3+都可以通过絮凝从原水中去除有机物。图2(b)显示了不同投量AlCl3和FeCl3絮凝后的TOC去除率。从图2(b)看出,FeCl3对有机物的去除效果比AlCl3的更好。此外,图2(c)~图2(d)显示了通过不同浓度AlCl3和FeCl3处理后溶液中有机物的分子质量分布。从图中可以看出,2种絮凝剂都能有效去除分子质量为1 000~10 000 Da的有机物。膜滤也常用于焦化废水的处理[11]。图2(e)显示了采用截留分子质量为1 000 Da的PA膜处理前后的有机物分子质量分布,与图2(c)~图2(d)比较可以看出,絮凝对有机物的去除效果与采用膜过滤的效果相近。随着絮凝剂投加量增多,溶液中剩余有机物的浓度下降。当2种絮凝剂投加量均为10 mmol·L−1时,FeCl3比AlCl3去除了更多的大分子有机物(1 000~10 000 Da)。采用FTIR对Al3+絮凝得到的絮体进行分析,结果如图2(f)所示。3 374 cm−1处的峰表示分子间氢键,而1 624 cm−1附近的峰表示弱吸附水分子H—O—H键角弯曲振动;1 100 cm−1和556 cm−1处的峰分别表示Al—O—H弯曲振动和Al—O伸缩振动[18],并且它们的强度随絮凝剂投量的增加而增大。
-
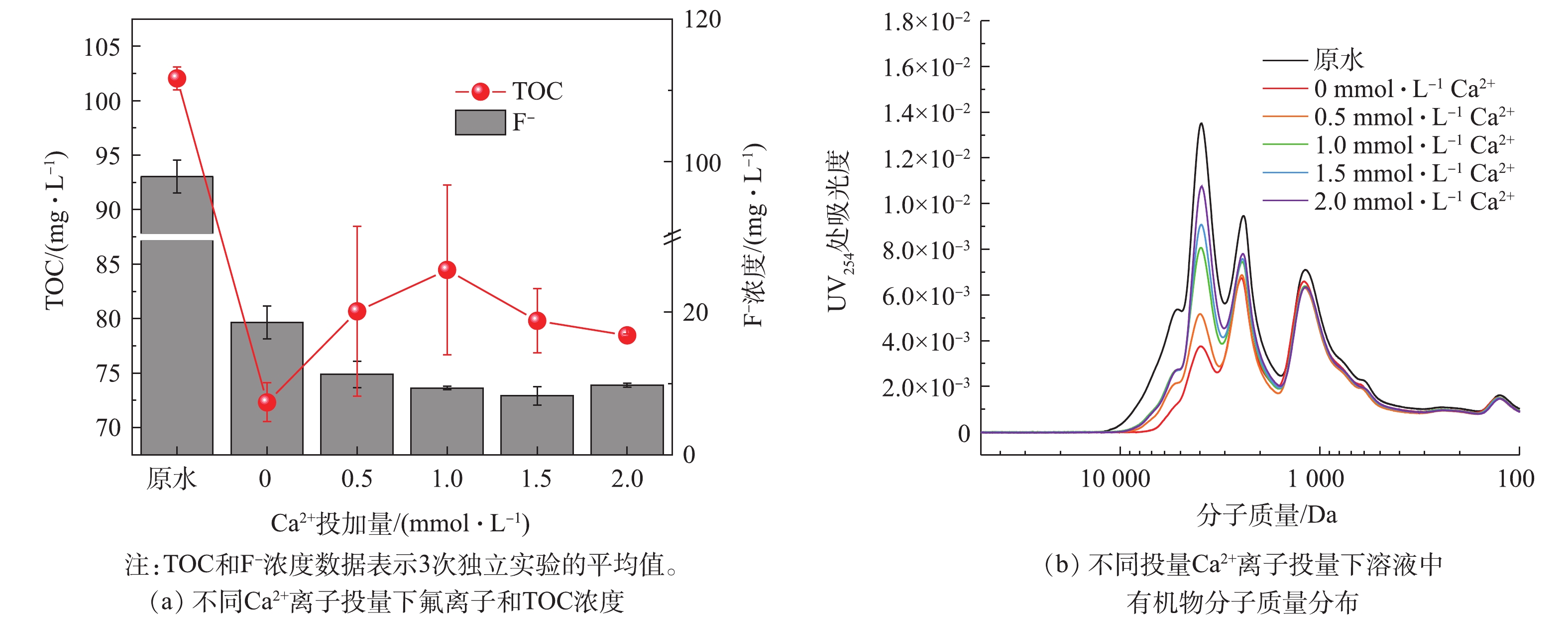
工业上常采用Ca2+沉淀的方法从废水中去除和回收氟[14-15],因此还考察了Ca2+对絮凝效果的影响。图3显示了在10 mmol·L−1 AlCl3絮凝体系中加入不同浓度Ca2+时,溶液中TOC浓度和F−浓度的变化。Ca2+可以通过与F−结合形成氟化钙沉淀来去除氟。反应过程如式(4)所示。
当加入1 mmol·L−1 Ca2+时,F−浓度从18.57 mg·L−1降至9.36 mg·L−1,这与CaF2在水中的溶解度接近(20 ℃时CaF2的溶解度为17 mg·L−1,F−溶解度为8 mg·L−1)[19]。然而,加入1 mmol·L−1 Ca2+使得TOC浓度从72.32 mg·L−1上升到84.47 mg·L−1,并且凝胶色谱的结果也表明Ca2+会影响絮凝去除有机物的效率。分析其原理,可能有2个方面:1)Ca2+与有机物络合,降低了絮凝剂对有机物的絮凝效率;2)由于AlCl3在中性条件下发生絮凝作用的原理是网捕卷扫,AlCl3水解形成的无定型的氢氧化物颗粒聚集在一起形成絮体,吸附水中的溶解性和小颗粒杂质[20-21];絮体表面的—OH和—OH2的活性位点在絮体生长方面起着重要的作用[16],然而CaF2颗粒非常细小,可能会吸附到絮体表面占据一部分活性位点,从而导致有机物的去除率下降。
-
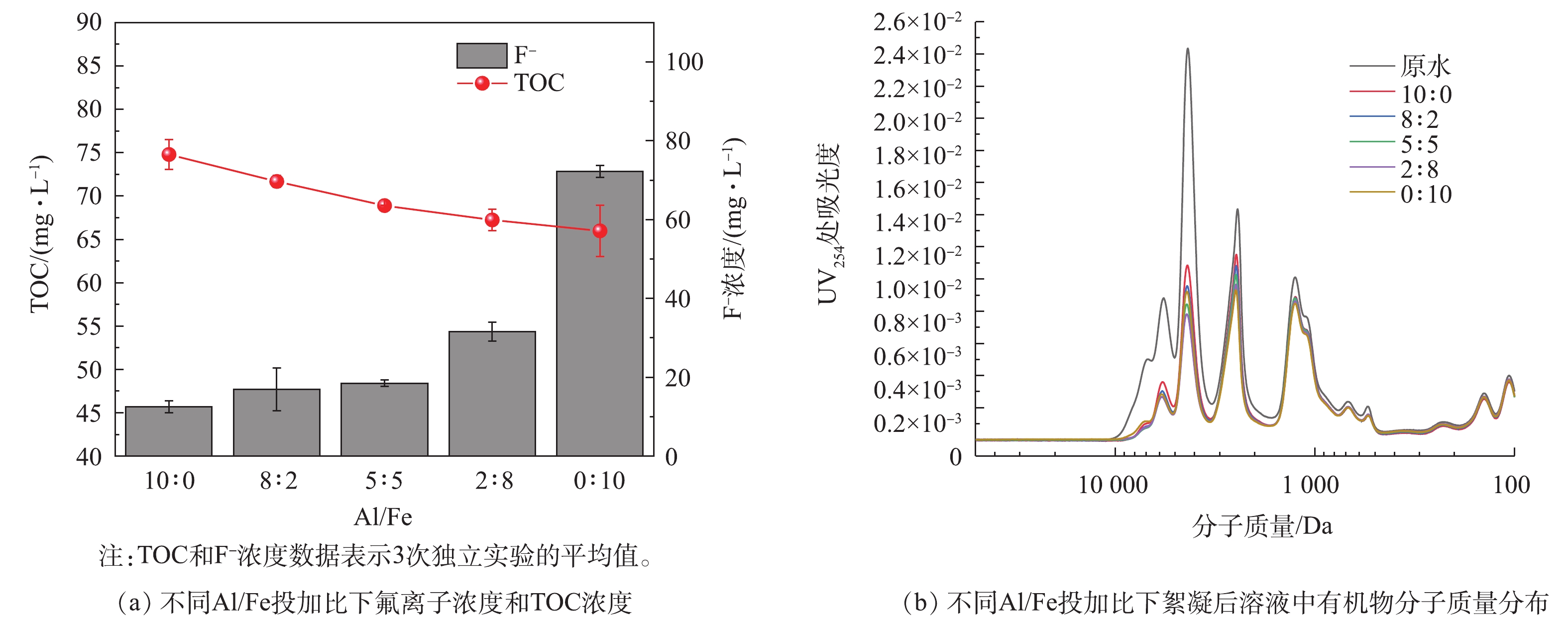
Al/Fe投加比例对絮凝的影响见图4。考虑到FeCl3的有机物去除性能优于AlCl3,并且原料成本也更低,在焦化废水的大规模处理中采用FeCl3和AlCl3复配絮凝剂进行絮凝的方式,可能会更高效且经济。因此,继续考查了Al/Fe的投加比例对絮凝效果的影响。FeCl3和AlCl3的总投加量均为10 mmol·L−1。从图4(a)可以看出,当FeCl3的比例从0上升到100%时,絮凝后溶液的TOC从71.11 mg·L−1降至64.21 mg·L−1,而F−浓度的变化则相反,从7.65 mg·L−1升至88.86 mg·L−1。当FeCl3的比例不超过50%时,F−浓度保持在一个较低的范围内。该结果与前面已验证过的AlCl3除氟效率较高,而FeCl3对有机物去除效率较高的结论吻合。图4(b)表明溶液中有机物的浓度随着FeCl3投加比例增加而下降。当Al/Fe投加比例下降时,絮体的FTIR谱图(图4(c))中556 cm−1处Al—O峰逐渐消失,而Fe—O—H弯曲振动的特征峰在710 cm−1附近出现,同时另一位于1 072 cm−1处的特征峰强度也增加[22],表明随着铁投加比例的增加,铁絮体的生成也增多。此外,谱图中3 300、2 996和2 894 cm−1附近的峰分别对应絮体表面不同的羟基官能团[23]。随着FeCl3比例的增加,这些峰的强度逐渐增大,表明Fe的水解产物形成的氢氧活性基团多于Al的水解产物[24],从而解释了FeCl3的有机物去除效率更高的原因。
值得一提的是,溶液的初始pH随Al/Fe投加比有较大变化(图4(d)),pH随着FeCl3比例上升而下降。为了考查调整pH对絮凝效果的影响,在加入不同比例复配絮凝剂后,将所有的pH值调到6.5进行实验,结果如图5所示。当FeCl3的比例从0增至100%,TOC从74.79降至65.98 mg·L−1时,F−浓度从12.51增至72.25 mg·L−1。分子量分布结果也表明有机物去除效率随着FeCl3比例上升而增加。尽管总体的变化趋势与图4中的相似,当AlCl3投量在5~10 mmol·L−1范围内,F−浓度比未调pH时要高。这表明pH对絮凝后的F−浓度有较大影响,这将在下一部分进行讨论。
-
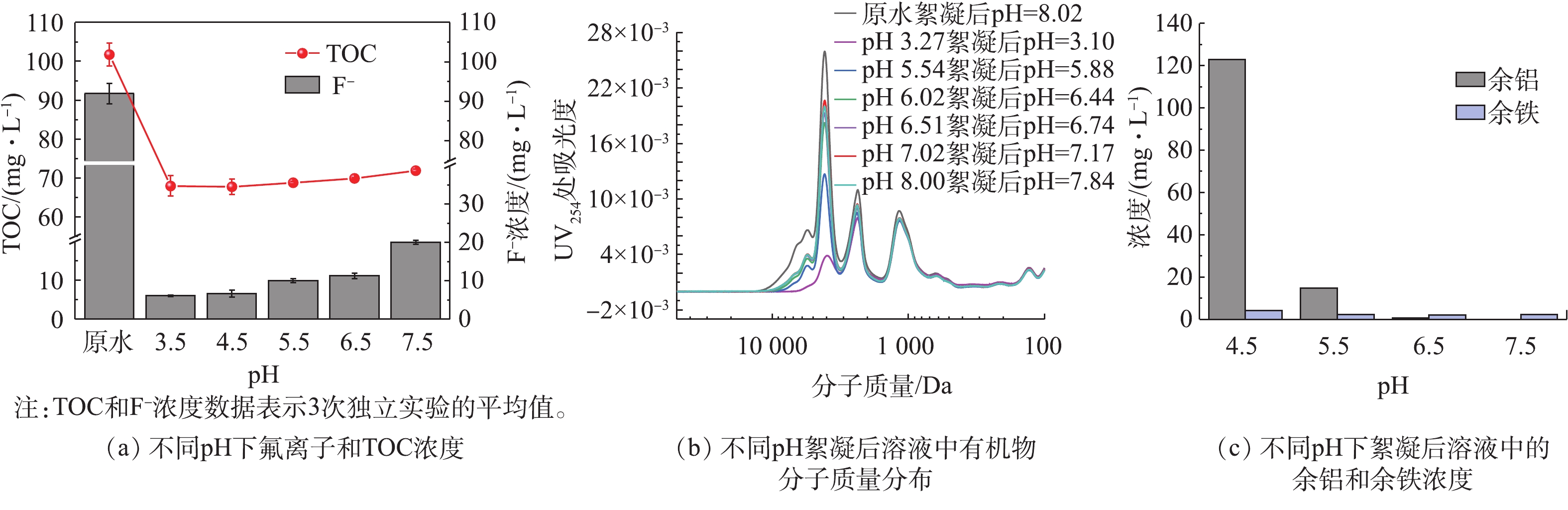
为了进一步研究pH对铁铝复配絮凝的影响,将Al/Fe投加比例控制在8∶2,通过改变初始pH来分析絮凝去除有机物和氟的效率。由图6数据可知,TOC随pH的变化影响很小,而有机物分子量分布的结果显示大分子有机物(5 000~10 000 Da)的去除效率在低pH下较高。这表明焦化废水中的大分子有机物主要通过电性中和机理被去除。此外,在低pH条件下,溶液中F−浓度更低。有研究表明,在低pH和高F−浓度条件下,可溶性Al-F络合物的形成可能会降低氟的去除效率[25]。因此,推测在低pH时F−去除率较高是由于F−与Al3+络合形成了Al-F物质,而这部分络合态的Al-F中的F−不能被F−选择电极方法检测到,故其实际去除率比观察到的去除率要低。为证实这个猜测,采用ICP-OES测量不同pH条件下絮凝反应后溶液中的Al3+和Fe3+浓度。根据图6(c)的结果,溶液中余铝浓度在酸性条件下明显高于中性条件下的浓度,表明有大部分的Al以可溶性Al-F络合物的形式存在。溶液中余铁的浓度随着pH的上升而下降,但始终保持在一个较低的范围。因此,尽管酸性条件下溶液中的表观F−浓度较低,但由于Al-F的存在,F−的实际去除率被高估了。综合考虑F−浓度和剩余Al3+浓度的结果,确定絮凝处理焦化废水的最佳pH为6.5。在此pH下,剩余Al3+浓度低,形成的Al-F络合物较少,实际F−去除率与检测到的F−去除率接近。而在更低的pH条件下,处理后溶液中的表观F−浓度虽然更低,但却是以Al-F形式存在,实际并未从溶液中去除。
2.1. AlCl3和FeCl3对F−和有机物的去除
2.2. Ca2+对絮凝效果的影响
2.3. Al/Fe投加比例的影响
2.4. pH对铁铝复配絮凝的影响
-
1) AlCl3比FeCl3的pHpzc高,因此,在中性条件下,AlCl3与氟络合的能力要优于FeCl3;但FeCl3表面具有更多的氢氧活性基团,在去除有机物方面的效率优于AlCl3。
2) Ca2+可以通过形成CaF2沉淀增强对F−的去除。Ca2+一方面会与有机物络合影响其絮凝去除效率,另一方面CaF2颗粒会占据絮体表面活性位点从而对有机物的去除产生不利影响。
3) Al/Fe复配絮凝能提高对有机物和F−的去除,最适宜的Al/Fe投加比例为8∶2,pH为6.5。在酸性条件下,F−与铝易形成可溶Al-F络合物,从而降低F−的去除效率。




 下载:
下载: