-
随着化石燃料的燃烧、工农业生产的加速发展,越来越多的氮进入水环境中。含氮化肥使用、废水排放、畜禽养殖、垃圾渗滤等[1], 使得农村地下水环境硝酸盐超标现象较多,但由于硝酸盐的总体超标浓度不高[2],去除难度大。然而长期饮用硝酸超标水源,会增加患消化道癌症的风险,干扰人体红细胞的输氧功能,造成身体缺氧,同时诱导脑、神经系统等方面的疾病[3-6]。因此,在2022年国家最新发布的《生活饮用水卫生标准》(GB57 49-2022)中对地下水源硝酸盐指标限值做了调整,由20 mg·L−1降低到10 mg·L−1。目前水体中硝酸盐的去除技术主要有吸附[7]、反渗透[8]、离子交换[9]、电渗析[10]、生物处理[11]、电凝聚[12]等,这些技术各有优劣,实际推广应用存在一定困难。
电容去离子(capacitive deionization, CDI)由于其节能、高效、再生成本低、不产生污染物等优点,成为一种新兴的脱盐技术。CDI的吸附即通过向两极板施加电压,使无机离子停留在电极表面,以达到去除目的;当吸附达饱和后,采用施加反向电位、短接等方式使离子离开电极孔实现再生[13]。为促进CDI技术的发展,研究人员在CDI结构设计和电极材料研制方面开展了大量研究工作。膜电容去离子技术(membrane capacitive deionization, MCDI)是CDI的升级,因离子交换膜的加入降低了共离子驱逐效应[14],大大提高了离子存储能力和再生效率,延长了电极使用寿命。LIU等[15-17]开展了CDI电极复合电极材料研制工作,进一步提高了电极的吸附容量。
近年来,有研究表明,CDI技术对离子的去除不具备选择性,对硝酸盐去除能力较差,特别是对低浓度硝酸盐、与其他离子共存时,去除后硝酸盐再生脱附困难。为解决这些问题,LI等[18]利用戊二酸(GA)和磺基琥珀酸(SSA)制备了一种新型离子交换聚合物,修饰电容去离子电极,考察其对于亚硝酸盐的选择去除能力。KIM等[19]将阴离子选择性聚合物与聚苯乙烯大孔硝酸盐选择性树脂涂覆在碳电极上,用于城市污水脱盐和膜电容去离子过程中NO3−回收,实验表明其对NO3−去除有较好的选择性。GAN等[20]将阴离子交换树脂(A520E)和羧基功能化多壁碳纳米管涂覆在活性炭电极上,组装CDI反应器,测试其对硝酸盐的选择性去除能力,实验表明涂覆两种电极后选择性有显著提高。目前开展的研究大多将离子交换树脂采用黏附方式直接附于电极上,该方法存在材料易脱落、两极板间电阻增大等缺点。
本研究通过静电纺丝法直接一体成型制备出对硝酸盐具有选择去除性能的薄膜,并将NSRM膜和碳纤维、离子交换膜组装成复合电极开展了硝酸盐去除性能研究,通过分析测试阐明了NO3−在复合电极表面的选择性电吸附过程。
-
Fumasep FAS-50阴离子交换膜,Fumasep FKS-50阳离子交换膜,德国FuMa Tech公司;N,N-二甲基甲酰胺(DMF,99.5%),上海麦克林生化科技有限公司;聚丙烯腈(PAN, Mw=150 000) 购自Sigma-Aldrich (St. Louis, MO, USA)公司;IRA402强碱型阴离子交换树脂,上海阿拉丁生化科技股份有限公司;硝酸钠(NaNO3,分析纯),国药集团化学试剂有限公司;其他试剂均购自国药集团化学试剂有限公司;实验中所使用的水均来自超纯水净化系统。
-
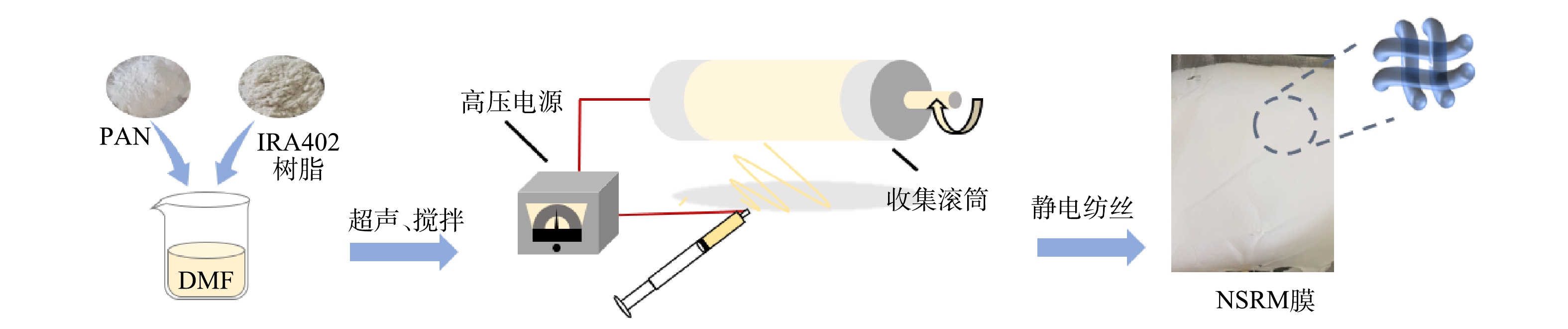
纺丝工艺流程如图1所示。将1.62 g聚丙烯腈(PAN)和15.0 g DMF磁力搅拌12 h,加入0.514 g IRA402搅拌3 h后超声分散2 h,配置得NSRM纺丝液。制备条件为:电压18 kV,间距15 cm,速率1 mL·h−1,相对湿度≤40%,纺丝时间分别为1.5、3、5 h。干燥后按集电极尺寸10 cm×10 cm剪裁组装即可。
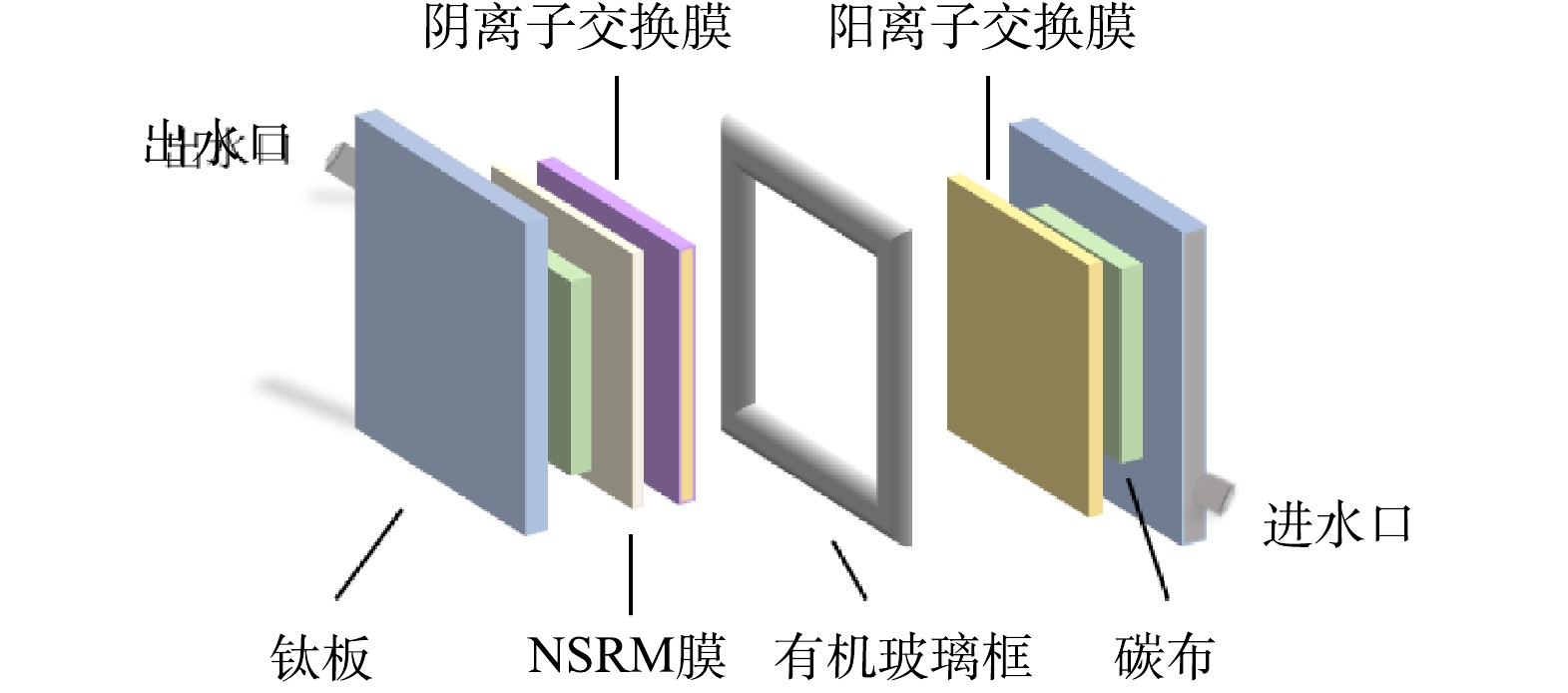
碳纤维布根据集电极尺寸10 cm×10 cm剪裁后,采用沸煮的方式进行清洗,每隔30 min换水,清洗2~3次后在60 ℃烘箱中烘干后组装。离子交换膜直接剪裁组装。MCDI电极对装配如图2所示,从左到右依次叠加钛板、碳布、NSRM膜、离子交换膜、硅胶垫片、有机玻璃框、隔网。
-
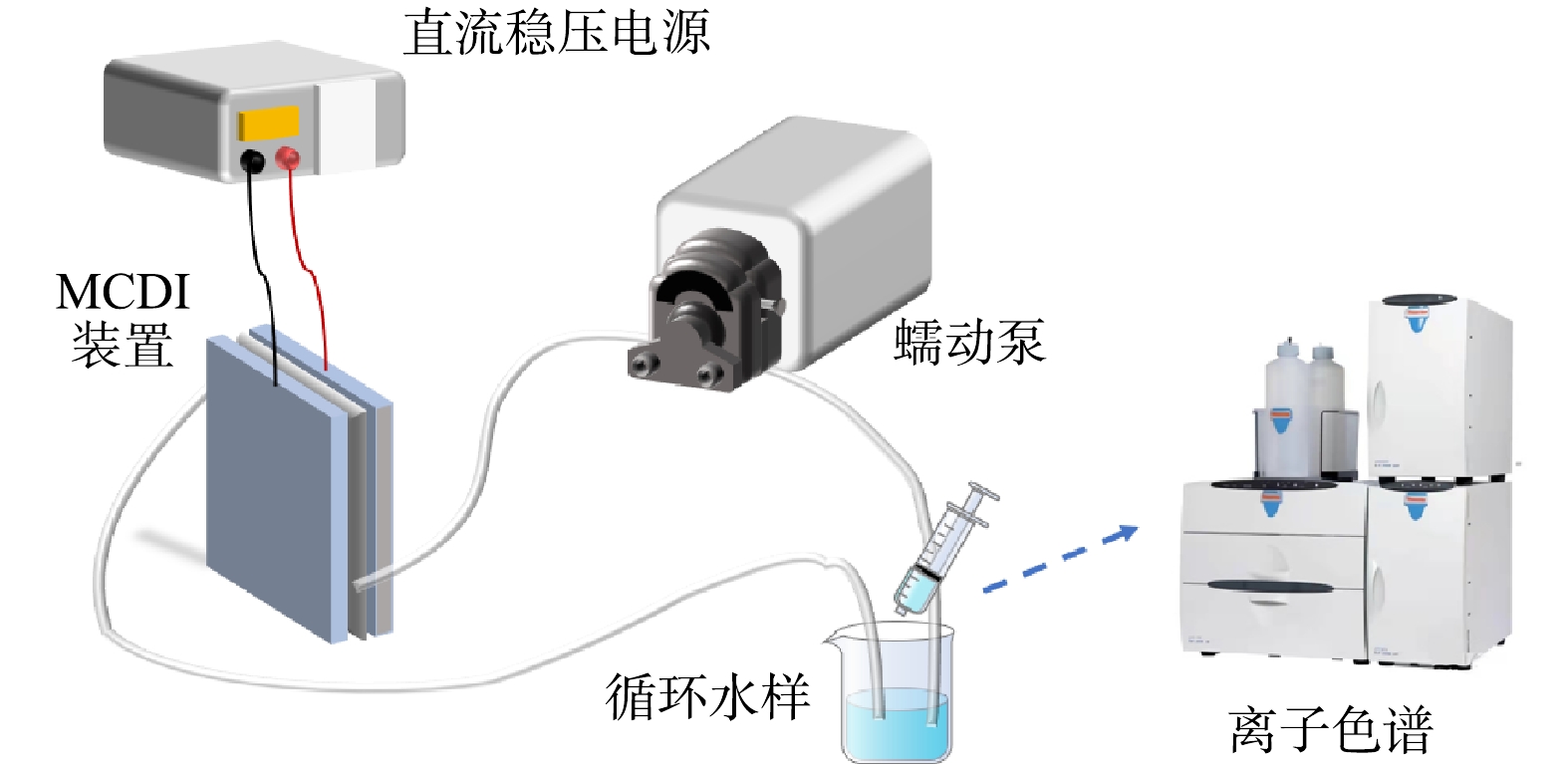
MCDI去除硝酸盐性能测试系统如图3所示。直流稳压电源(UTP3315TFL,优利德科技股份有限公司)给MCDI电极供电,水样通过蠕动泵(YZ1515x,兰格恒流泵有限公司)实现循环流动。采用电导率仪实时监测离子去除情况,定时取样通过离子色谱测试硝酸根浓度变化。
各工艺条件对硝酸盐选择去除性能的影响实验。设置不同操作电压(1.6、1.8、2.0、2.2 V)、循环流速(40、60、80、100 mL·min−1)、硝酸盐初始质量浓度(50、100、150、200 mg·L−1),取200 mL水样循环吸附,采用电导率仪定时记录溶液电导率,并通过公式(1)计算硝酸盐去除率,正向施加电压60 min后,反接10 min实现电极再生完成1次循环,重复2次以验证其可重复性。
装置对硝酸盐选择去除性能的影响实验。取水中常见阴离子(F−、NO3−、Cl−、SO42−、PO43−)不同种类、不同比例混合,在2.0V的外加电压下,取200mL水样进行循环吸附实验。定点取1mL水样进行稀释,后通过0.25µm过滤头注入离子色谱,测定主要阴离子含量,通过反接实现电极再生。
-
分别采用扫描电子显微镜(SEM)、傅里叶红外光谱仪(FTIR)表征NSRM膜的表面形貌、化学成分及官能团组成;采用Zeta电位测试NSRM膜电荷性能。用离子的去除率、单位面积吸附量、选择性、电荷效率对去除效果进行评估。阴离子的电吸附去除率根据式(1)进行计算,单位面积吸附量根据式(2)进行计算,离子在某段时间的电吸附选择性根据式(3)进行计算,电荷效率根据式(4)进行计算。
式中:
$ \eta $ 表示电吸附去除率,%,$ {c}_{0} $ 表示水样的初始浓度,$ \mathrm{m}\mathrm{m}\mathrm{o}\mathrm{l}\cdot {\mathrm{L}}^{-1}{;c}_{t} $ 表示时间t时水样的浓度,$ \mathrm{m}\mathrm{m}\mathrm{o}\mathrm{l}\cdot {\mathrm{L}}^{-1} $ 。式中:
$ {A}_{\mathrm{i}} $ 表示离子i的单位面积吸附量,g·m−2;$ {\mathrm{\Delta }M}_{\mathrm{i},t} $ 表示时间t内离子含量变化,g;S为NSRM膜面积,m2。式中:表示S离子的选择性,
$ {A}_{\mathrm{i}} $ 表示离子在时间t的单位面积吸附量,g·m−2。式中:
$ \Lambda $ 表示电荷效率,%;n是电吸附离子电荷值;F是法拉第常数,96 485.3 C·mol−2;I表示电流密度,A·m−2;A表示电极有效面积,m2。 -
图4(a)和图4 (b)分别为空白和NSRM的SEM表征结果。由图4(a)可见,未掺杂树脂的空白薄膜的纤维丝管径尺寸分布均匀,表面光滑无明显缺陷,而NSRM薄膜(图4(b))树脂颗粒与纤维丝相互交缠形成一体,与树脂相连纤维的管径尺寸显著变小,球状树脂颗粒表面呈现多孔的形貌,有利于吸附水体中的离子。
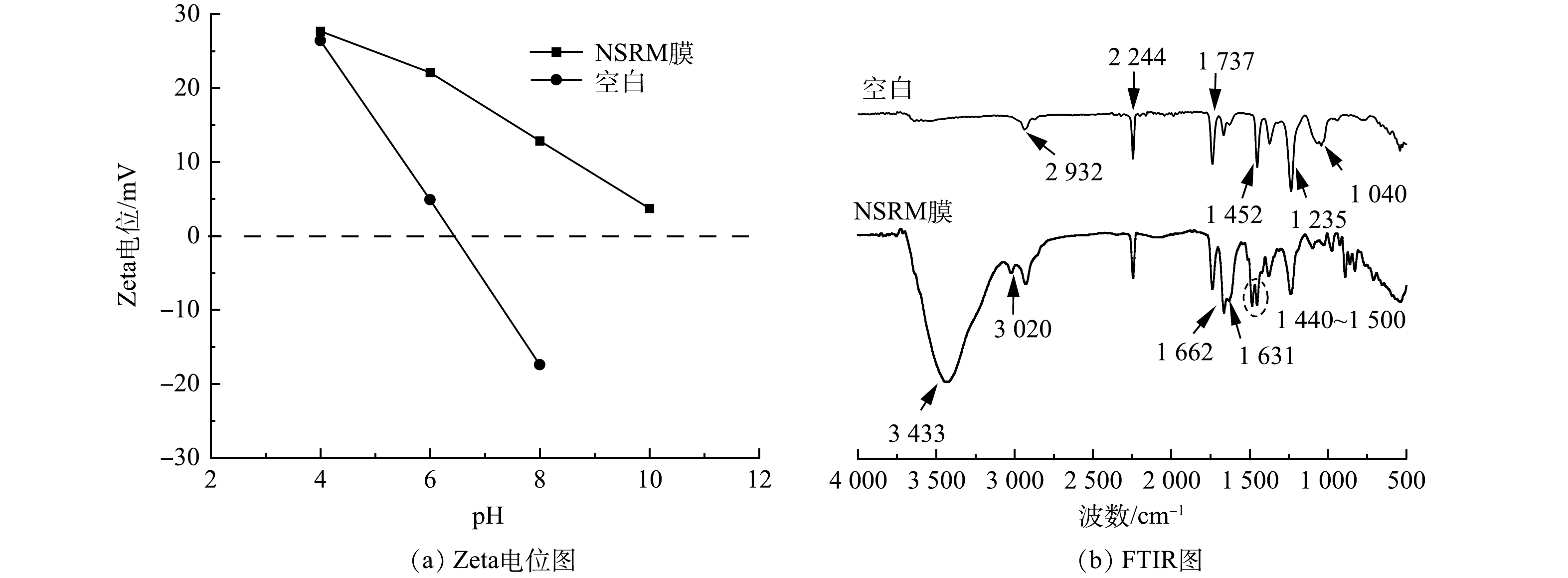
图5(a)为空白膜和NSRM膜在不同pH下的Zeta电位。可见,空白膜在pH小于6.44时带正电,大于6.44时带负电,而NSRM膜在pH为4~10内均带正电。这表明IRA402树脂颗粒的添加使得NSRM膜在较宽的范围内表面带正电荷,更有利于吸附水溶液中的阴离子。图5(b)为空白和NSRM膜的FTIR光谱。由图5(b)中空白图谱可见,PAN的特征峰有2 240 cm−1和1 040 cm−1的伸缩振动峰[21],2 932、1 235、1 452 cm−1处的C—H振动峰[22]和1 737 cm−1处的C=O伸缩振动峰[23]。将离子交换树脂加入PAN后,出现了较多新的吸收峰,其中3 433 cm−1 为树脂O—H和N—H振动吸收峰[24-25],3 020 cm−1为弱乙烯基=CH2不对称伸缩峰[24],1 662 cm−1处峰在空白基础上有加强,表明PAN与树脂发生了环化或其他共价作用。1 631 cm−1 左右为C=C或C=N伸缩振动峰[26],1 440~1 500cm−1为C=C及—CH2的伸缩振动[27],该处峰值产生分叉峰可能是由于树脂加入后C(O)NH键的生成。此外谱图中1 000 cm−1左右的指纹区为C—N的伸缩振动,由于该键的生成,使得N原子的电子发生转移,缺失电子从而表现出正电性,提升了阴离子的吸附效率。
-
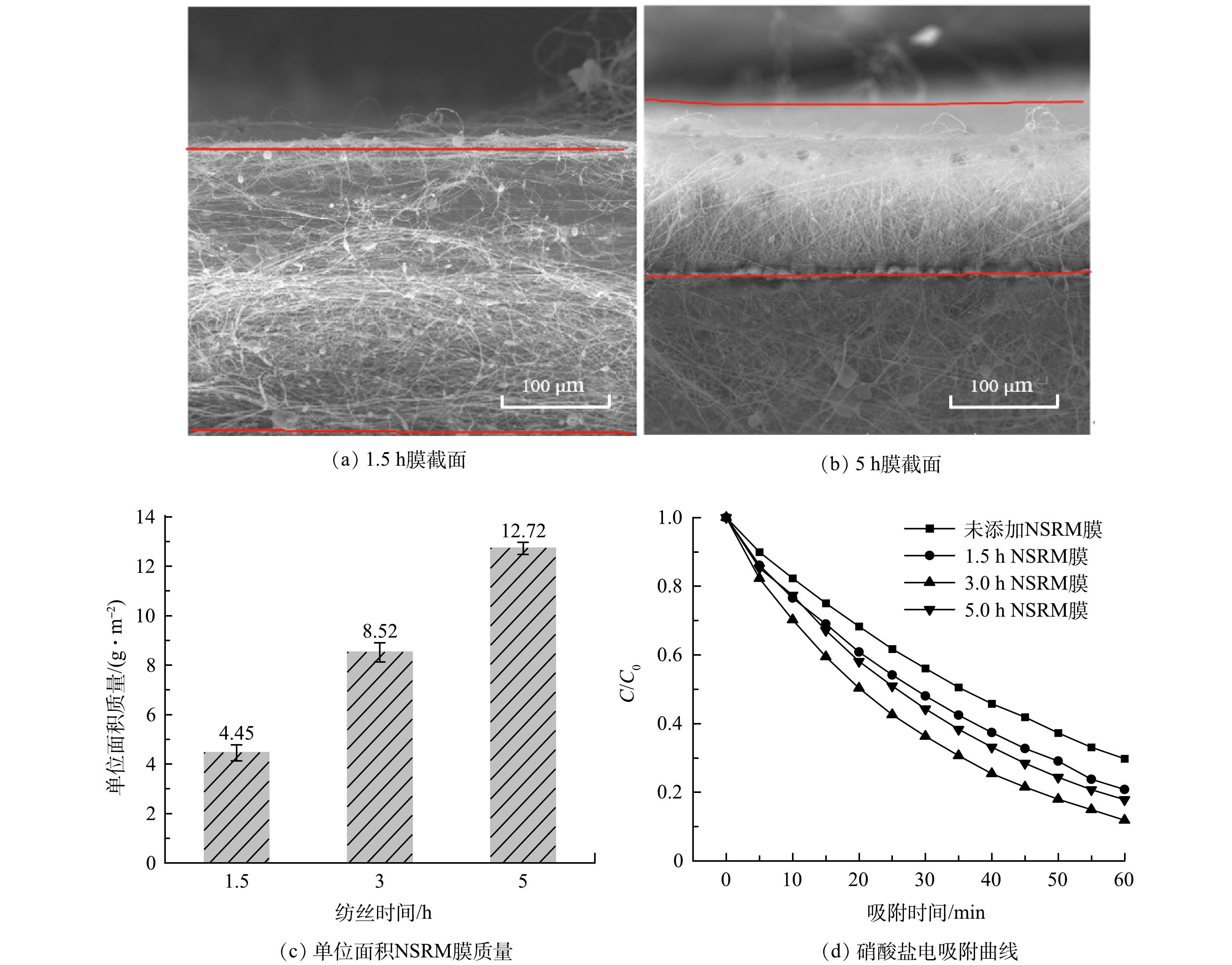
图6反映了NSRM膜制备工艺中纺丝时间对硝酸盐去除性能影响。图6(a)和图6(b)分别为纺丝时间为1.5 h和5 h时 NSRM膜的截面SEM表征结果。经测量其厚度均分别为235 μm和130 μm,随着纺丝时间的延长,膜的厚度反而变薄。实验同时采用称重法测量不同纺丝工艺NSRM膜单位面积质量,结果如图6(c)所示。可见,随着纺丝时间延长,其单位面积的膜质量有所增加。结合截面SEM结果,发现随着纺丝时间的延长,膜逐渐变得密实。图6(d)为添加不同纺丝时间制得的NSRM膜后MCDI装置对硝酸根离子的去除情况。由图6(d)可知,添加NSRM膜后MCDI装置对硝酸盐去除性能均有明显改善,其中添加3 h NSRM膜的MCDI装置对硝酸根离子的去除效果最佳,其去除率达88.17%,比未添加NSRM膜的装置提高了18%。由表1的计算结果可见,采用伪二级动力学方程进行描述,速率为0.015 3 g·(m2·min)−1。
由以上结果推测,3 h的膜去除率和去除速率比1.5 h和5 h更高的原因是随着时间延长,单位面积上有效官能团和IRA402树脂成分增加,但随着时间延长,单位面积膜质量增加,膜更密实,从而引起电荷传质效率降低。后续实验均采用纺丝时间为3 h的NSRM膜。
-
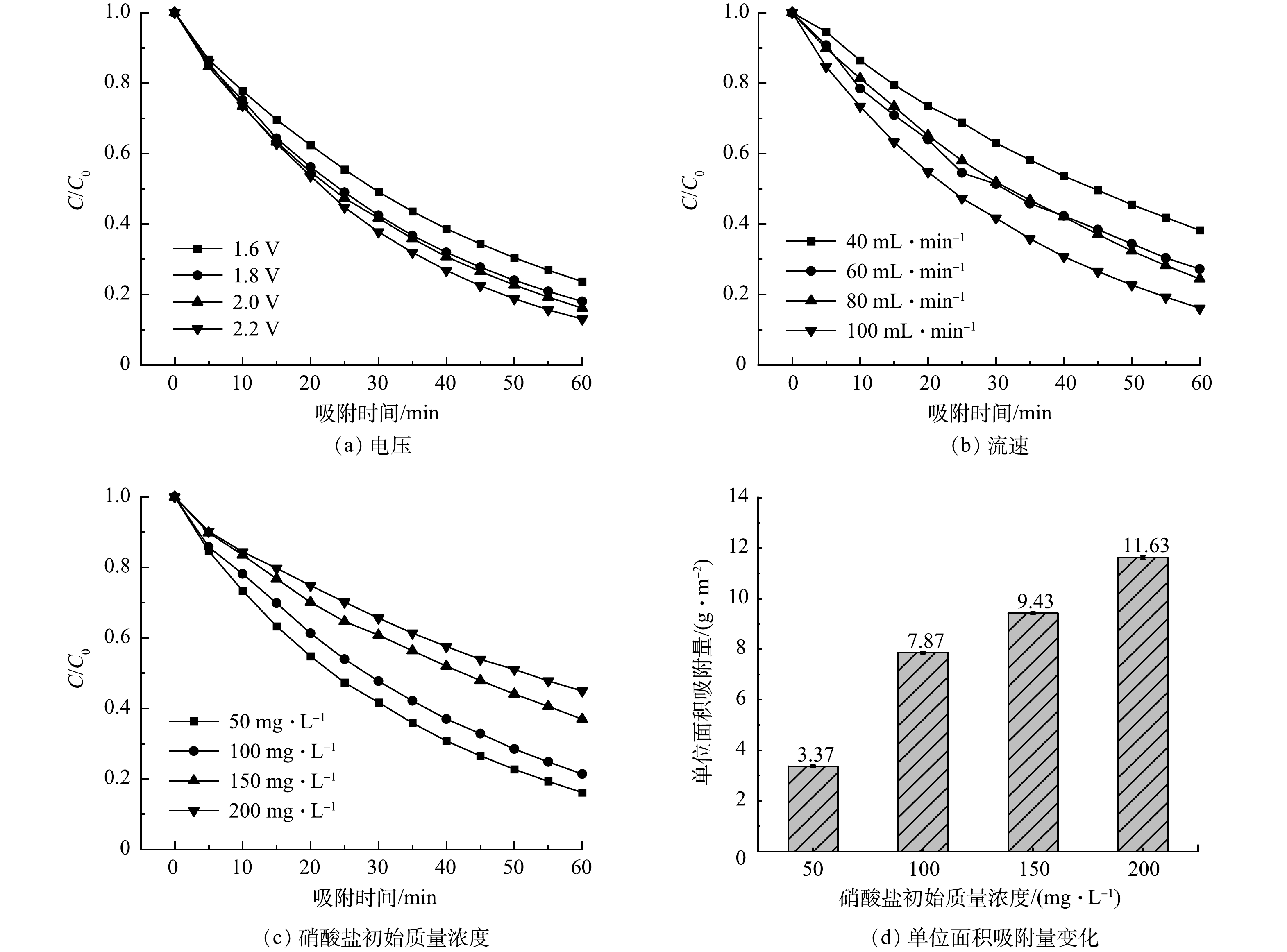
图7分别考察了装置操作电压、循环流速和硝酸盐初始质量浓度对MCDI去除硝酸盐性能的影响。由图7(a)可见,硝酸根离子的去除率随电压的增大而升高。这是因为随着电压的升高,极板间的电位差增大,对带电离子的吸附驱动力增强,但过高的电压会造成水的电解,进而降低电极寿命。电压为2.0 V时未产生明显气泡,溶液pH由6.94升至7.21,实验过程中未探测到亚硝酸盐等其他产物。同时,采用式(4)对MCDI电荷效率进行计算,在硝酸根初始质量浓度为50 mg·L−1、电压为2.0 V时电荷效率为69.98%;当电压为2.2 V时,电荷效率为60.10%。相比之下,2.0 V条件电荷效率更高,因此,后续实验中采用2.0 V操作电压。
由图7(b)可见,当硝酸根初始质量浓度为50 mg·L−1、电压为2.0 V、循环流速分别为40、60、80、100 mL·min−1时,60 min后硝酸根离子的去除率为61.76%、72.69%、75.58%和83.86%。即随着循环流速增加,单位时间内硝酸根离子的去除率有所升高。这是因为当流速较低时,水的循环率也较低,水体中离子与电极接触概率低;随着流速增加,大大提高了水体中离子与电极表面的接触概率,因此,硝酸根离子去除效率得到提高。
图7(c)是装置对硝酸盐初始质量浓度分别为50、100、150、200 mg·L−1 、电压为2.0 V、循环流速为100 mL·min−1时的电吸附变化曲线。可见,60 min后硝酸根离子的去除率分别为83.86%、78.66%、63.10%和55.05%。图7(d)是电压为2.0 V、循环流速为100 mL·min−1时,硝酸根离子的单位面积吸附量变化。可见,随着硝酸盐溶液初始质量浓度的增加,虽然硝酸根离子的单位面积吸附容量增加,但去除效率呈下降趋势。因为随着硝酸盐初始质量浓度增加,水体电导率增加,即电极表面和溶液间可形成更高的浓度梯度,从而降低离子的扩散阻力,进而提升电极的单位面积吸附容量。而去除率随着质量浓度增加降低是因为电极与溶液界面形成的双电层厚度与溶液的质量浓度呈反比[28],低浓度时双电层更厚,有更多吸附点,有利于有效地保持电荷,从而提高离子去除率。
综合考虑以上因素认为,当操作电压为2.0 V,溶液循环流速为100 mL·min−1时,MCDI系统对硝酸根离子有最优的吸附效果,故后续实验均在此条件下进行。
-
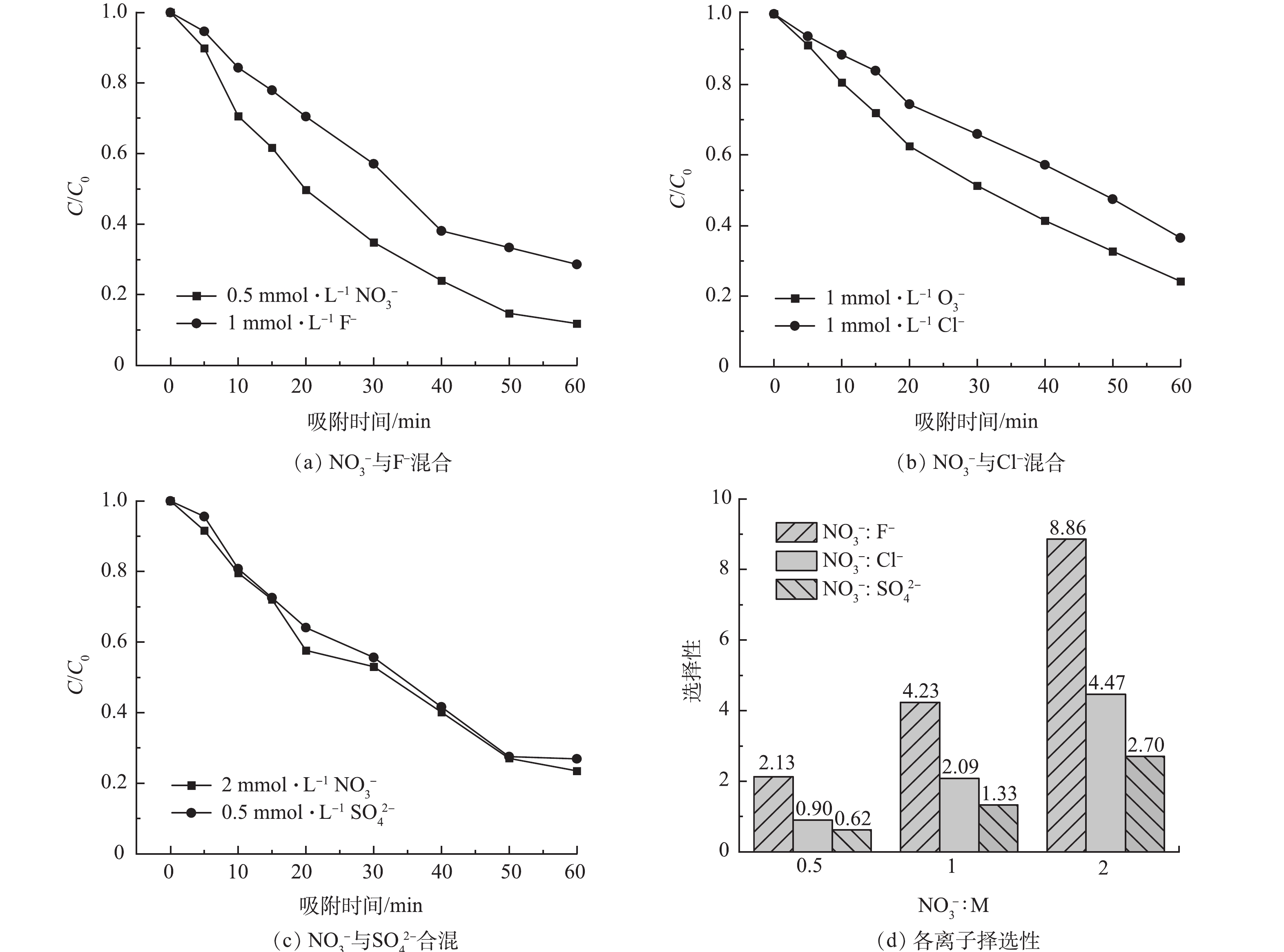
图8(a)~(c)为1 mmol·L−1 F−、1 mmol·L−1 Cl−、0.5 mmol·L−1 SO42−分别与不同浓度NO3−(0.5、1、2 mmol·L−1)混合时降解曲线。当以上3种离子与硝酸根电荷比分别为0.5:1、1:1和2:1时,硝酸根降解率达88.16%、75.8%和76.46%,相比F−、Cl−和SO42−降解率分别提高了16.74%、12.31%和3.37%。结合表2可见,在同价态情况下,NSRM膜对硝酸根离子的去除具有较强选择性。而当其与二价阴离子SO42−混合时,对其选择性略有不足,可能与离子的水合半径和在水中迁移的速率有关。有研究表明,较小水合半径[19]及较高价态的离子[29]更能够被有效去除。在相同外加电压下,离子在溶液中电迁移率越低,迁移速度越慢,而在理想溶液中,F−、NO3−、Cl−、SO42−的迁移率分别为5.74×10−8、7.40×10−8、7.92×10−8和8.27×10−8 m2·(s·V)−1[20],溶液中硫酸根离子快速迁移到电极附近,优先被吸附。由图8(d)可见,随硝酸根浓度增加,溶液中硝酸根离子数目增多,相对各离子的选择性也增加,在与硝酸根离子电荷数为1:1的情况下,F−选择性达4.23,Cl−可达2.09。
图9(a)、图9(b)、图9(c)分别为F−、Cl−、NO3− 3种离子按不同浓度比例混合时的降解曲线。可见,当F−:Cl−:NO3−分别为1:1:1、1:1:2和1:2:1时,硝酸根离子的去除率均为最高,达81.2%、81.1%和70.9%。根据图9(e)~(g)单位面积吸附量,硝酸根离子单位面积吸附量较其他离子高。结合表3中添加NSRM膜前后多离子混合去除率的变化可知,加入NSRM薄膜后,各离子的去除率均有所提高,但NO3−的去除率提升最为显著。表明NSRM膜添加后对硝酸盐具有较强选择性。图9(d)为水中不同价态阴离子等电荷共存时的动态降解曲线。可见,NO3−、SO42−、PO43−在同一混合液中的降解率分别为55%、59.7%和51.2%,无明显差异。该膜对于不同价态离子的选择去除效果有待进一步探究。
-
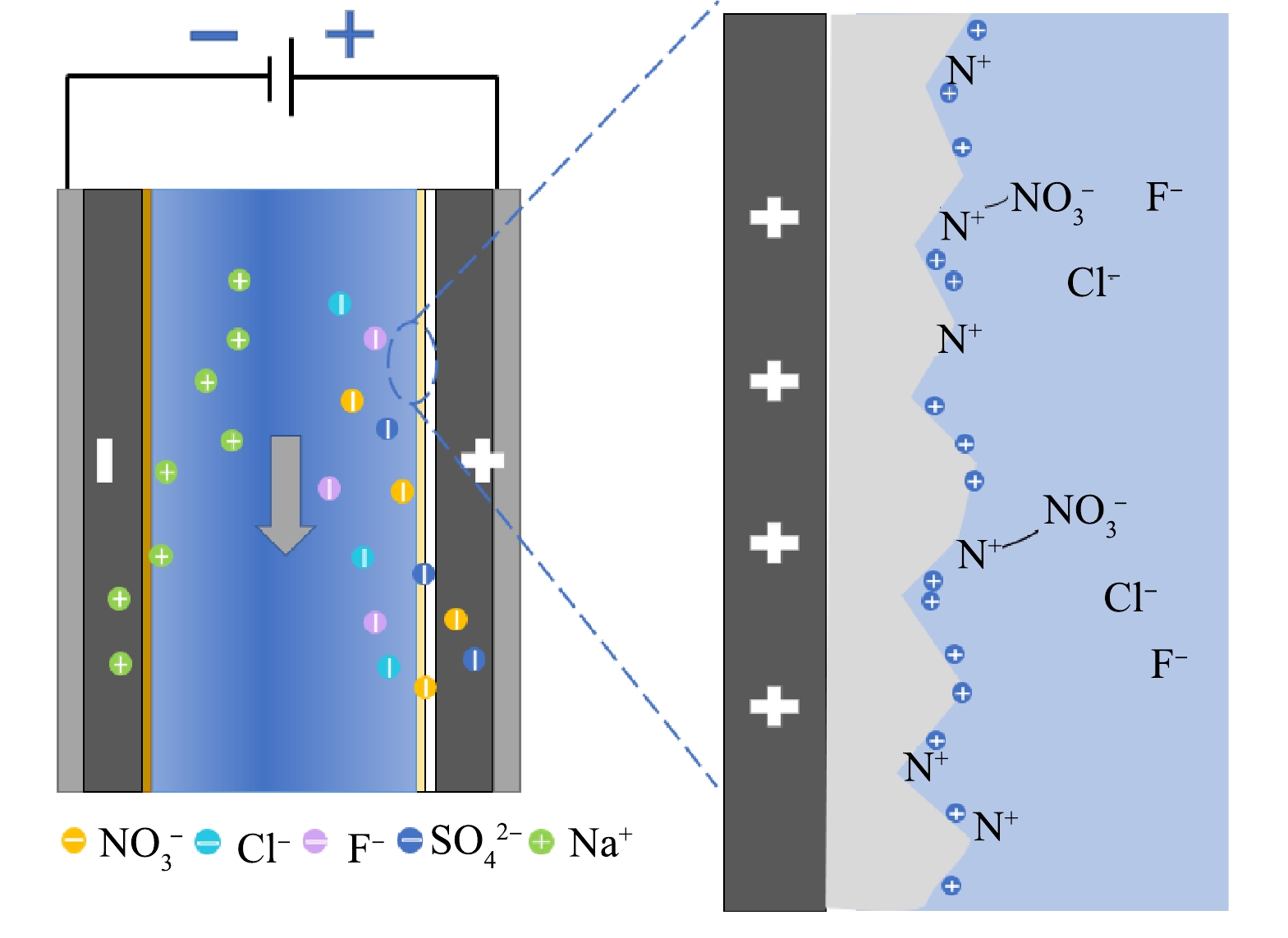
图10为硝酸盐吸附原理图。在两极电压驱动下,溶液中的阴阳离子分别聚集在阳极和阴极周围形成双电层。由图5(a)中Zeta电位可知,NSRM膜表面带正电,减少了阴离子吸附的排斥,同时提高了吸附驱动力,使两极间双电层更加稳定;结合FTIR图推测NSRM膜所带正电基团为胺基基团,根据共价键的量子理论,共价键的强弱取决于形成共价键的2个电子轨道相互交叠的程度,交叠越多,键能越大,系统能量越低,键越牢固,NO3−中心氮原子采用sp2杂化形式,杂化轨道为平面三角形。与胺基基团相似,故相对于F−和Cl−,两者结合更为稳定;另一方面通过查阅N—N、N—F和N—Cl原子之间结合能,发现N—N结合能更高,因而吸附稳定性更强,且在吸附过程中N—Cl结合能较低,因此,具有较高吸附稳定性的N—N会取代稳定性较差的N—Cl[30]。
-
1)采用静电纺丝法直接一体成型制备出对硝酸盐具有选择去除性能的NSRM薄膜,薄膜中树脂颗粒与纤维丝相互交缠形成多孔结构,膜表面含
$ \mathrm{C}\equiv \mathrm{N} $ 、C(O)NH、N—H基团,且表面电荷在较宽的pH范围内均带正电。2) NSRM薄膜电容去除硝酸盐性能的测试结果表明,纺丝时间为3 h制备得到的膜对硝酸根离子的去除效果最佳,去除率可达88.17%,通过拟合计算,采用伪二级动力学方程进行描述,速率为0.015 3 g·(m2·min)−1。当操作电压为2.0 V,溶液循环流速为100 mL·min−1时,MCDI系统对硝酸根离子有最优的吸附效果。
3) NSRM薄膜对F−、Cl−有较好的选择性,对硫酸根有一定选择性,对磷酸根选择性不佳,其中,当NO3−与F−、Cl−比值均为1:1时,硝酸的选择性分别为4.23和2.09。
选择性复合电极的制备及其对硝酸盐的去除性能
Preparation of selective composite electrode and its performance on nitrate removal
-
摘要: 以IRA 402强碱性阴离子交换树脂为原料,通过静电纺丝技术制备对硝酸盐具有选择去除性能的薄膜(nitrate selective removal membrane,NSRM),采用扫描电镜、傅里叶红外、Zeta电位分析仪等仪器对其进行分析和表征,考察了纺丝时间、操作电压、循环流速、硝酸盐初始浓度和共存离子等因素对NO3−去除性能的影响。表征结果表明,NSRM膜表面粗糙多孔且带正电,薄膜上C≡N、C(O)NH、N—H等基团的存在促进NO3−的优先吸附。电吸附结果表明,纺丝时间3 h制得的NSRM膜对NO3−去除率最优,在初始质量浓度为50 mg·L−1,操作电压为2.0 V,循环流速为100 mL·min−1时硝酸盐去除率达88.17%。多离子混合条件下,NSRM薄膜对F-、Cl-有较好的选择性,对SO42−有一定选择性,对PO43-选择性不佳,其中,当NO3−与F−、Cl−物质量浓度比值为1:1时,NO3−的选择性分别为4.23和2.09。Abstract: In this study, the strong base anion exchange resin(IRA 402) doped nitrate selective removal membrane (NSRM) was prepared through electrospinning. Scanning electron microscopy, Fourier transform infrared, and Zeta potentiometer are used for its analysis and characterization. The effects of spinning time, operating voltage, circulation flow rate, nitrate initial mass concentration and coexisting ions on the NO3− removal performance were investigated. The characterization results showed that the surface of the NSRM membrane was rough, porous, and positively charged. The presence of C≡N, C(O)NH and N-H groups on the membrane promoted the preferential adsorption of NO3−. The results of electrosorption experiments showed that the NSRM membrane produced with a spinning time of 3 h had a best performance on NO3− removal, with a nitrate removal efficiency of 88.17% at an initial mass concentration of 50 mg·L−1, an operating voltage of 2.0 V, and a circulation flow rate of 100 mL·min−1. Under the multi-ion mixing conditions, the NSRM membrane had a good selectivity for F− and Cl−, a partial selectivity for SO42−, and a poor selectivity for PO43−, where the selectivity for NO3− was 4.23 and 2.09 when the mass concentration ratios of NO3− to F− or Cl− substances were 1:1, respectively.
-
Key words:
- electrosorption /
- nitrate /
- composite electrode /
- selective removal
-

-
表 1 伪一级、伪二级吸附动力学参数模型
Table 1. Parameters of pseudo first-order and pseudo second-order model
纺丝时间/h 伪一级动力学方程 伪二级动力学方程 K1/min−1 R2 K2/(g·(m−2·min−1)) R2 未添加NSRM 0.020 0.998 6 7.5×10-3 0.997 9 1.5 0.022 0.999 2 9.0×10-3 0.999 6 3 0.030 0.999 4 0.015 3 0.999 9 5 0.027 0.999 1 0.012 4 0.999 4 表 2 不同混合条件下离子单位面积吸附量
Table 2. Adsorption capacity per unit area of ions under different mixing conditions
g·m−2 NO3−: M 与F−混合 与Cl−混合 与SO42−混合 NO3− F− NO3− Cl- NO3− SO42− 0.5 2.73 1.29 2.52 2.81 2.60 4.19 1 5.74 1.35 4.70 2.25 4.92 3.70 2 10.83 1.22 9.14 2.05 9.48 3.51 表 3 添加NSRM膜前后多离子混合去除率
Table 3. Removal efficiency of multi-ion mixing before and after adding NSRM membrane
% F−:Cl−:NO3− 空白 3 h NSRM F− Cl− NO3− F− Cl− NO3− 1:1:1 30.39 40.91 49.02 55.57 70.70 81.19 1:2:1 27.11 39.71 43.66 46.62 66.06 70.94 1:1:2 36.44 51.36 57.66 60.04 74.54 81.11 -
[1] 刘恒源, 杨彦韬, 鲍文达, 等. 电化学法去除地下水中硝酸盐的机理研究[J]. 赤峰学院学报(自然科学版), 2021, 37(7): 46-49. doi: 10.13398/j.cnki.issn1673-260x.2021.07.011 [2] 陈骞. 浅析地下水中的硝酸盐污染[J]. 2022, 3(2): 90-92. [3] 王诗绘, 马玉坤, 沈珍瑶. 氮氧稳定同位素技术用于水体中硝酸盐污染来源解析方面的研究进展[J]. 北京师范大学学报(自然科学版), 2021, 57(1): 36-42. [4] 厉彦梅. 地下水中硝酸盐的各种去除方法概述[J]. 城市地理, 2014(22): 90. doi: 10.3969/j.issn.1674-2508.2014.22.074 [5] 张懿文, 罗建中, 陈宇阳. 我国水体中硝酸盐的污染现状及危害[J]. 广东化工, 2015, 42(14): 99-100. doi: 10.3969/j.issn.1007-1865.2015.14.049 [6] 干方群, 吴珂, 马菲, 等. 水体硝酸盐检测方法的研究进展[J]. 中国无机分析化学, 2022, 12(1): 69-81. doi: 10.3969/j.issn.2095-1035.2022.01.012 [7] KUMAR P S, YAASHIKAA P R, RAMALINGAM S: Efficient removal of nitrate and phosphate using graphene nanocomposites[J]. Applications in Water Technology, 2019: 287-307. [8] QASIM M, BADRELZAMAN M, et al. Reverse osmosis desalination: A state-of-the-art review[J]. Desalination, 2019, 459: 59-104. doi: 10.1016/j.desal.2019.02.008 [9] HEKMATZADEH A A, KARIMI J A, et al. Modeling of nitrate removal for ion exchange resin in batch and fixed bed experiments[J]. Desalination, 2012, 284: 22-31. doi: 10.1016/j.desal.2011.08.033 [10] ALIASKARI M, SCHAFER A I. Nitrate, arsenic and fluoride removal by electrodialysis from brackish groundwater[J]. Water Research, 2021, 190: 116683. doi: 10.1016/j.watres.2020.116683 [11] PARK J Y, YOO Y J. Biological nitrate removal in industrial wastewater treatment: which electron donor we can choose[J]. Applied Microbiology and Biotechnology, 2009, 82(3): 415-29. doi: 10.1007/s00253-008-1799-1 [12] NIARAGH E K, MOGHADDAM M R A, et al. Evaluation of direct and alternating current on nitrate removal using a continuous electrocoagulation process: Economical and environmental approaches through RSM[J]. Journal of Environmental Management, 2019, 230: 245-254. [13] LEE J B, PARK K K, EUN H M, et al. Desalination of a thermal power plant wastewater by membrane capacitive deionization[J]. Desalination, 2006, 196(1/2/3): 125-134. [14] BIESHEUVEL P M. Membrane capacitive deionization[J]. Journal of Membrane Science, 2010, 346(2): 256-262. doi: 10.1016/j.memsci.2009.09.043 [15] LIU Y H, HSI H C, LI K C, et al. Electrodeposited manganese dioxide/activated carbon composite as a high-performance electrode material for capacitive deionization[J]. ACS Sustainable Chemistry & Engineering, 2016, 4(9): 4762-4770. [16] CHEN L, HE F, LI F. Denitrification enhancement by electro-adsorption/reduction in capacitive deionization (CDI) and membrane capacitive deionization (MCDI) with copper electrode[J]. Chemosphere, 2022, 291: 132732. doi: 10.1016/j.chemosphere.2021.132732 [17] BAO S, CHEN Q, ZHANG Y, et al. Optimization of preparation conditions of composite electrodes for selective adsorption of vanadium in CDI by response surface methodology[J]. Chemical Engineering Research and Design, 2021, 168: 37-45. doi: 10.1016/j.cherd.2021.01.032 [18] LI D, NING X A, YUN Y, et al. Ion-exchange polymers modified bacterial cellulose electrodes for the selective removal of nitrite ions from tail water of dyeing wastewater[J]. Journal of Environmental Sciences, 2020, 91: 62-72. doi: 10.1016/j.jes.2020.01.002 [19] KIM D I, GONZALES R R, DORJI P, et al. Efficient recovery of nitrate from municipal wastewater via MCDI using anion-exchange polymer coated electrode embedded with nitrate selective resin[J]. Desalination, 2020: 484. [20] GAN L, WU Y, SONG H, et al. Selective removal of nitrate ion using a novel activated carbon composite carbon electrode in capacitive deionization[J]. Separation and Purification Technology, 2019, 212: 728-736. doi: 10.1016/j.seppur.2018.11.081 [21] BULUT U, SAYIN V O. A flexible carbon nanofiber and conjugated polymer-based electrode for glucose sensing[J]. Microchemical Journal, 2023, 184: 108148. doi: 10.1016/j.microc.2022.108148 [22] ALTIN Y, BEDELOGLU A C. Polyacrylonitrile nanofiber optimization as precursor of carbon nanofibers for supercapacitors[J]. Journal of Innovative Science and Engineering (JISE), 2020, 4: 69-83. doi: 10.38088/jise.726792 [23] ABEYKOON N, BONSO J, FERRARIS J. Supercapacitor performance of carbon nanofiber electrodes derived from immiscible PAN/ PMMA polymer blends[J]. RSC Advances, 2015, 5: 19865-19873. doi: 10.1039/C4RA16594B [24] GUO L Y, LU H Q, RACKEMANN D, et al. Quaternary ammonium-functionalized magnetic chitosan microspheres as an effective green adsorbent to remove high-molecular-weight invert sugar alkaline degradation products (HISADPs)[J]. Chemical Engineering Journal, 2021, 416: 129084. doi: 10.1016/j.cej.2021.129084 [25] NORHAUATI A, MUHAMMAD R, KASSIM A A. Pre-evaluation of strong base anion exchange, Amberlite IRA 958-Cl resin for nitrate removal[J]. Materials Today:Proceedings, 2019, 17: 679-685. doi: 10.1016/j.matpr.2019.06.350 [26] SABANTINA L, BOTTJER R, WEHLAGE D, et al. Morphological study of stabilization and carbonization of polyacrylonitrile/TiO2 nanofiber mats[J]. Journal of Engineered Fibers and Fabrics, 2019, 14: 1558925019862242. [27] CIPRIANI E, ZANERRI M, BRACCO P, et al. Crosslinking and carbonization processes in PAN films and nanofibers[J]. Polymer Degradation and Stability, 2016, 123: 178-188. doi: 10.1016/j.polymdegradstab.2015.11.008 [28] BOHINC K, IGLIC A. Thickness of electrical double layer. Effect of ion size[J]. Electrochimica Acta, 2001, 46(19): 3033-3040. doi: 10.1016/S0013-4686(01)00525-4 [29] SEO S J, JEON H, LEE J K, et al. Investigation on removal of hardness ions by capacitive deionization (CDI) for water softening applications[J]. Water Research, 2010, 44(7): 2267-75. doi: 10.1016/j.watres.2009.10.020 [30] CEN B, YANG R, LI K, et al. Covalently-bonded quaternized activated carbon for selective removal of NO3– in capacitive deionization[J]. Chemical Engineering Journal, 2021: 425. -



 下载:
下载: