-
新疆地处内陆干旱区,降雨少、蒸发大,淡水资源匮乏,严重制约了新疆地区社会经济发展[1-3]。南疆地区拥有丰富的咸水资源[4-5],但并未被有效利用,反而对灌区排水载体造成严重污染[6]。因此,南疆地区咸水的开发利用刻不容缓。
咸水淡化作为减轻水体污染、缓解淡水资源紧张的有效途径,被众多学者所关注[7-11]。咸水淡化技术通常有反渗透法、电渗析法、冷冻法。段作山等[12]以地下水为淡化原水,采用了反渗透除氟除氨氮技术对水质进行提标改造,结果表明,反渗透工艺对氟化物与氨氮的去除率分别为97%和88%。且反渗透膜对
$ \mathrm{SO}_{4}^{2-}$ 、$\mathrm{PO}_{4}^{3-} $ 均有很高的截留性能,达到99.20%以上[13]。潘俊杰等[14]采用高效低耗的频繁倒极电渗析(EDR)技术去除地下水源中的硝酸盐,结果显示产水中硝酸盐含量完全符合国家饮用水标准,整套工艺设备运行稳定、能耗低、对原水含盐量变化适应性强等优点。因此在咸水淡化过程中,掌握其盐分及离子运移规律具有重要意义。冷冻法具有能耗低、预处理简单、腐蚀轻等优点[15],是一种高效的咸水淡化技术,在淡化效率[16]、经济效应[17]、能量损耗[18]等方面做出了深入研究,但冷冻法在不同冷冻温度条件下,结冰与融冰过程中盐分及离子迁移规律尚不明晰。因此,基于目前所取得的研究成果,需要开展以南疆咸水为淡化原水的冷冻法室内模拟试验,探索咸水结冰与融化过程中盐分与离子的迁移规律。基于此,本文分析了不同冷冻温度下,南疆农田排水结冰与融化过程中,矿化度及离子浓度的分布规律;探讨了基于多级冷冻与重力脱盐条件下,盐分及Cl−、
$ \mathrm{SO}_{4}^{2-}$ 、K+、Na+的迁移规律成因,以期为南疆咸水冷冻法淡化技术的推广应用提供依据。 -
试验原水取自塔南总排渠,取水点位于210省道192 km处(40°32′32″N,081°34′45″N),系阿拉尔灌区标志性总排渠,因此,供试农田排水水质具有一定代表性,水质指标如表1所示。
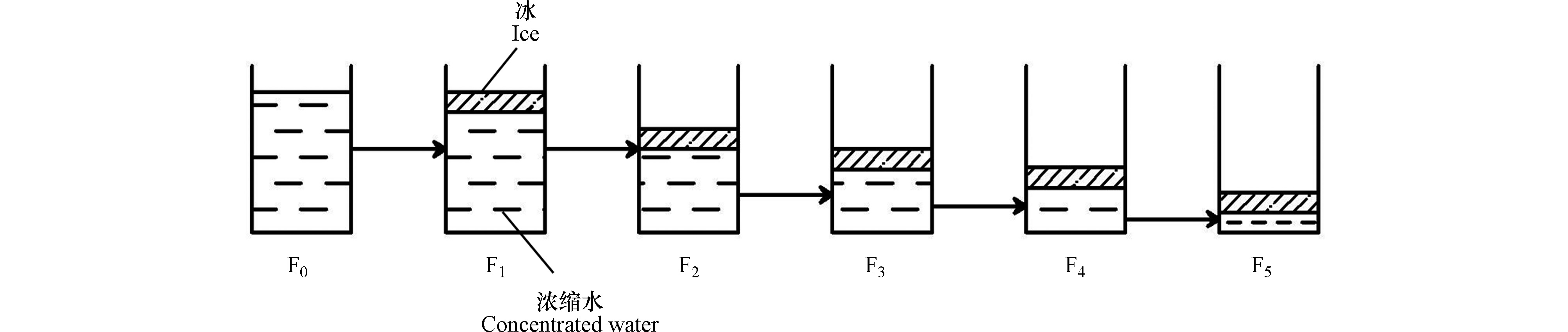
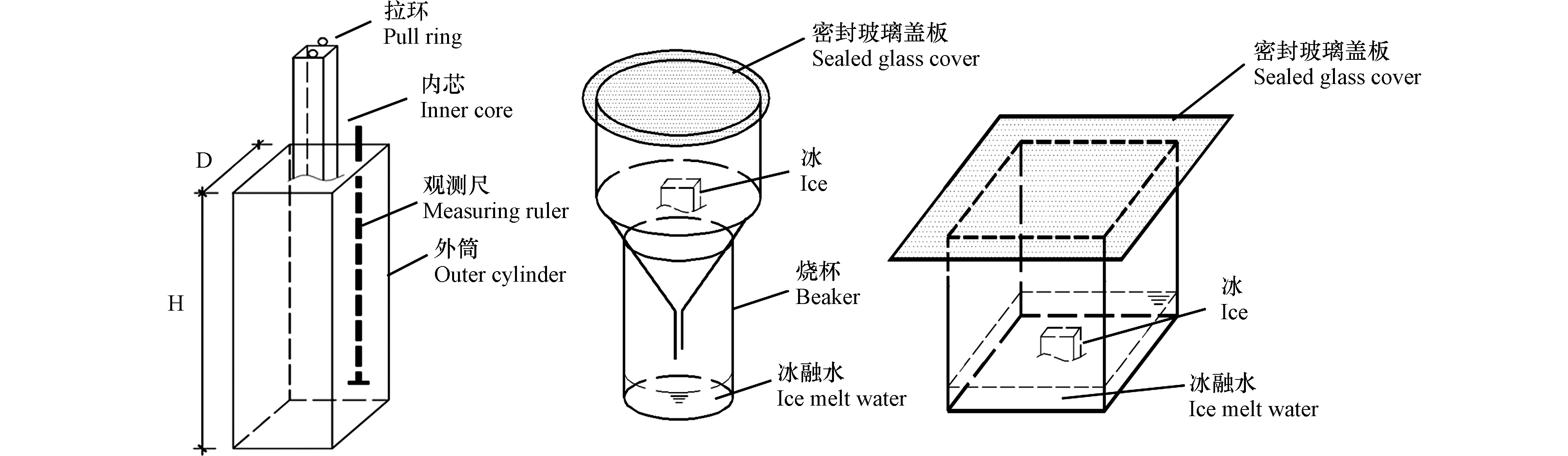
冷冻容器和融冰装置如图1所示。冷冻容器由外筒与内芯两部分(不锈钢材质)组成,其中,外筒高度H为35 cm,横截面为正方形,边长D为30 cm;内芯边长为25 cm与外筒高度一致,底部设有过水小孔,内筒上方拉环用于冷冻结束后内芯与外筒的分离,观测尺上刻有刻度,底部焊有钢片,每隔6 h对观测尺进行向上提拉,即可测得冰层厚度。为模拟自然结冰原理,冷冻容器四周及底部用保温材料(苯板)包裹,装置简图如图1所示。将供试水样放入冷冻容器后,置于恒温冰箱(BD/BC−768DKEMA)中,并以此恒温冰箱作为冷冻场。
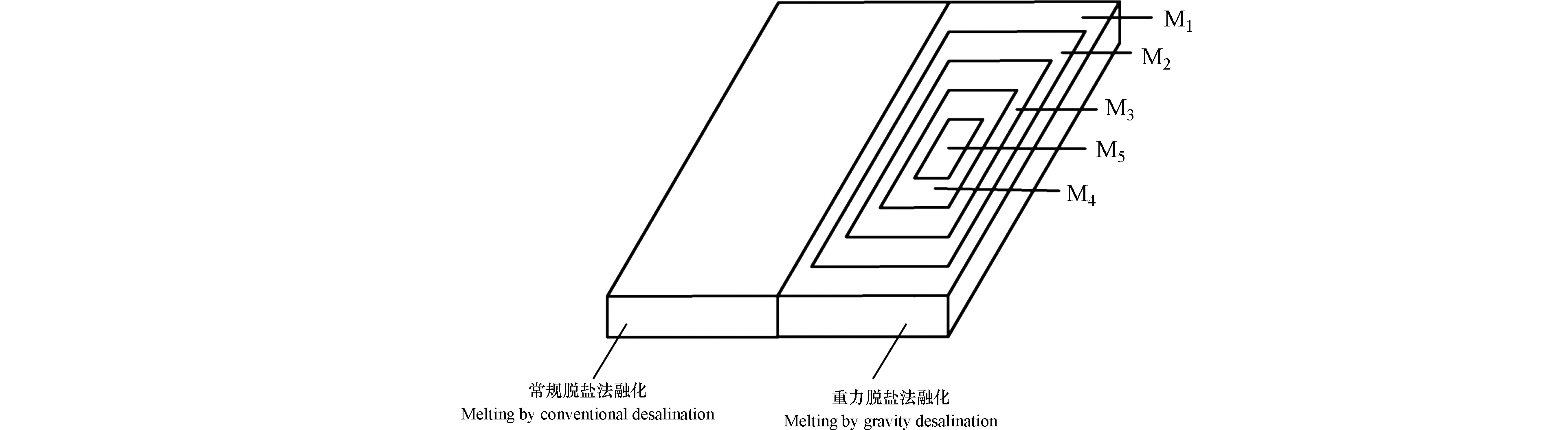
融冰装置:(a)为重力脱盐装置,由不锈钢漏斗、玻璃烧杯、密封玻璃盖板组成,将待融冰放在不锈钢漏斗内,漏斗上方加盖玻璃板,下方放置玻璃烧杯,用以接收冰融水;(b)常规解冻装置由玻璃容器及玻璃盖板组成,将待融冰放置于玻璃容器内融化并收集冰融水。
-
本试验设置冷冻温度与融冰方式2个影响因素,冷冻温度设置−6 ℃、−12 ℃、−18 ℃;融冰方式为重力脱盐融化与常规解冻融化。测定原水水质指标并标记为F0。
(1)结冰试验
结冰(Freeze)过程中,当冰层厚度达到3 cm时(V=1875 cm3),停止冷冻并进行冰水分离,以剩余浓缩水为原水,继续结冰,上述过程重复5次,按顺序标记为F1、F2、F3、F4、F5。结冰试验流程如图2所示。
(2)融冰试验
分离出的冰平分为2份,1份利用常规解冻法,收集冰融水,并测定水样指标;另外1份采用重力脱盐法融化,融化(Melt)过程如图3所示,每收集150 mL融水,记录融冰时间,收集5次,按顺序标记为M1、M2、M3、M4、M5。
-
离子:采用火焰光度计测定Na+、K+离子浓度;Cl−、
$\mathrm{SO}_{4}^{2-} $ 含量使用全自动化学分析仪(Smart chem 200)测定。矿化度:冰融水全盐体积质量用电导法测定(DDB-303A),采用15个样本。用重量法(GB 11901—89)标定矿化度与电导率的关系:
式中,Y为水样矿化度,mg·L−1;EC为电导率,µS·cm−1。
-
离子结冰比率定义为每级冰体中离子浓度与冰体中离子总浓度之比[19],即:
式中,Ci为每级冰体的离子浓度,mg·L−1,i为F1、F2、F3、F4、F5;Cice为冰体中离子的总浓度,mg·L−1。
离子融出比例定义为每份融水中离子浓度与总融水中离子浓度之比,即:
式中,Mi为每份融水中离子浓度,mg·L−1,i为M1、M2、M3、M4、M5;Mice为总融水中离子浓度,mg·L−1。
-
采用Origin2018、Auto CAD2018进行制图、Excel 2019对数据进行统计分析,使用SPSS 22.0对数据进行单因素方差分析(ANOVA)。
-
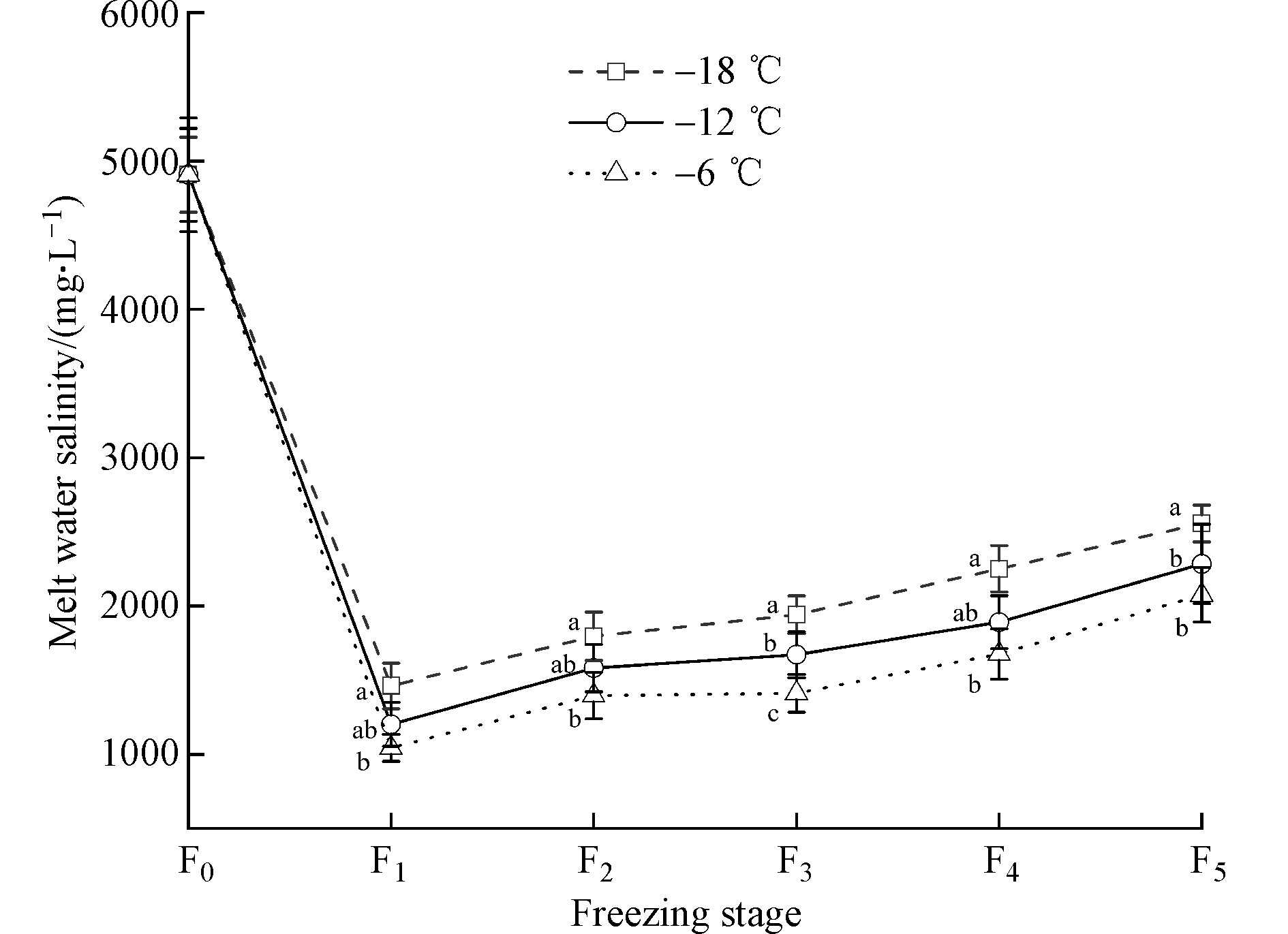
为探究不同温度对结冰过程中盐分及离子迁移的影响,采用常规脱盐冰融水进行分析。由图4可知,相同冷冻级数时,冰融水矿化度随温度升高而显著降低(P<0.05),冷冻级数为F1、F2、F3、F4、F5时,−18 ℃较−12 ℃冰融水矿化度升高17.79%、11.97%、13.91%、15.99%、10.65%,较−6 ℃升高28.60%、22.27%、27.30%、25.54%、18.80%,−12 ℃较−6 ℃冰融水矿化度升高13.15%、11.70%、15.56%、11.36%、9.11%,说明−18 ℃淡化效果最差,−6 ℃淡化效果最好。−18 ℃时冰融水矿化度由1461.24 mg·L−1(F1)上升至2555.82 mg·L−1(F5),−12 ℃时冰融水矿化度由1201.26 mg·L−1(F1)上升至2283.51 mg·L−1(F5),−6 ℃时冰融水矿化度由1043.27 mg·L−1(F1)上升至2075.37 mg·L−1(F5),说明同一冷冻温度条件下,冰融水矿化度随冷冻级数的增加而升高。综上所述,冷冻温度对冰融水矿化度具有显著影响,冰融水矿化度随冷冻温度的降低而上升。
-
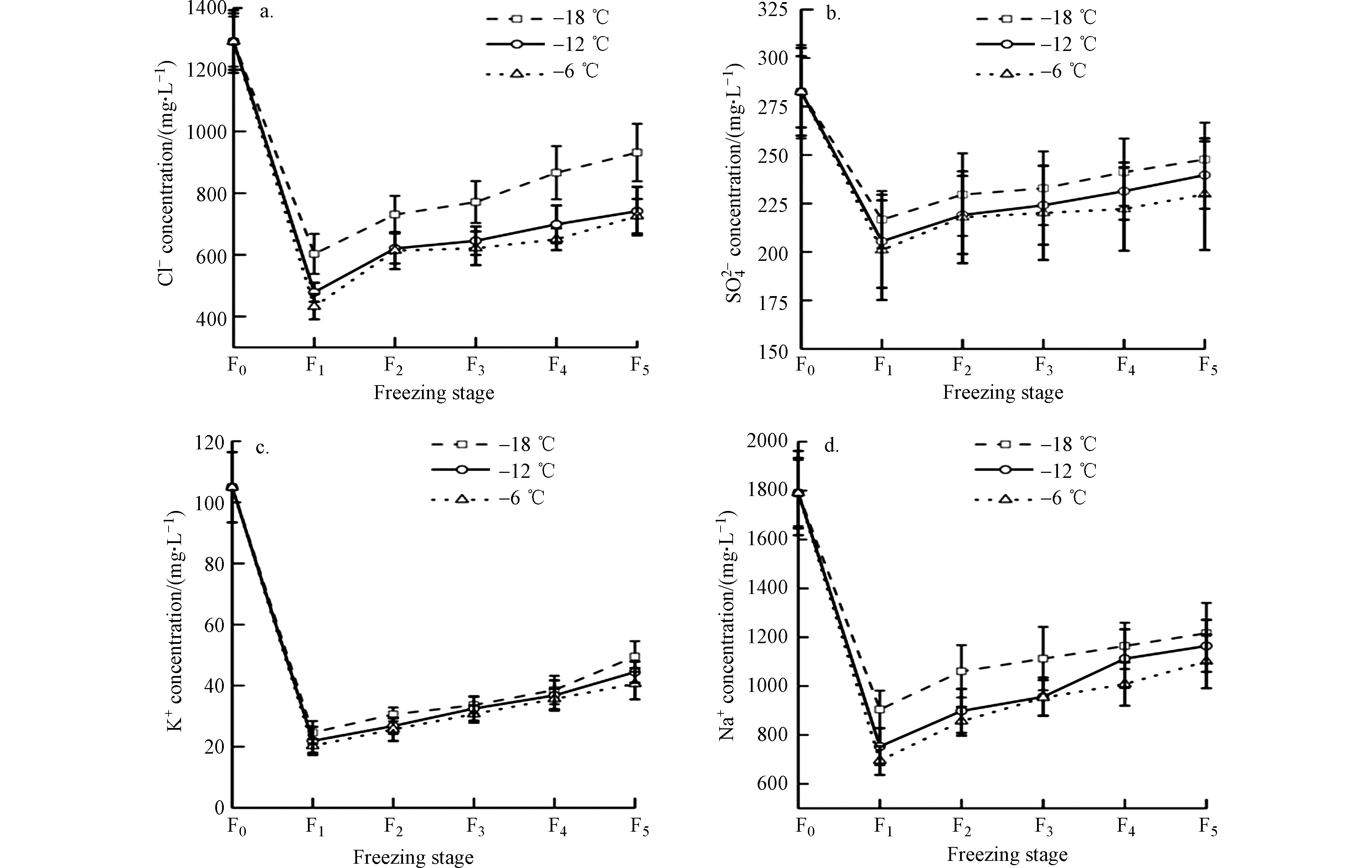
由图5可知,冷冻级数由F1增加到F5时,−6 ℃处理下Cl−、
$\mathrm{SO}_{4}^{2-} $ 、K+、Na+浓度提高40.47 %、12.57%、50.09%、36.87 %,−12 ℃时分别提高35.46 %、14.26%、50.66%、35.36 %,−18 ℃时分别提高35.30%、12.53%、50.27%、25.69%,各离子浓度随冷冻级数的增加而上升。相同冷冻级数下,各离子浓度随冷冻温度的降低而上升,冷冻级数为F1时,−18 ℃较−12 ℃冰融水中Cl−、$\mathrm{SO}_{4}^{2-} $ 、K+、Na+浓度上升20.60%、5.16%、10.99%、16.74%;−12 ℃较−6 ℃冰融水中Cl−、$\mathrm{SO}_{4}^{2-} $ 、K+、Na+浓度上升9.80%、2.23%、7.55%、7.67%。说明−18 ℃处理下冰融水中离子含量最高,离子去除效果最差;−6 ℃处理下冰融水中离子含量最低,离子去除效果最好。综上所述,不同温度处理下,各级冰融水矿化度及离子浓度较于初始水样(F0)显著降低(P<0.05),这是由于盐分与离子在冰中在水中的分配系数比在冰中分配系数大1—2个数量级的性质[20-21],因此结冰过程具有排斥盐分与离子的现象,盐分与离子会从上层冰体中迁移至下层溶液[22-23],从而导致上层冰体中矿化度和离子浓度较低。相同冷冻级数下,冰融水矿化度及离子含量随温度升高而显著降低(P<0.05)。是由于冰生长速率较低时,盐分及离子排出效果较好,冰生长速率较快,单位体积内形成迁移通道密度较大,被俘获在冰体中的盐分与离子较多[24];从分子化学的角度分析,水中氢键易受温度的影响,所以当环境温度相对较低的时候,氢键的缔结速度远大于断裂速度,水分子的热运动相对较弱,离子不易从冰体内逃脱[25]。因此,相同冷冻级数时,温度越低、盐分及离子去除效果越差。
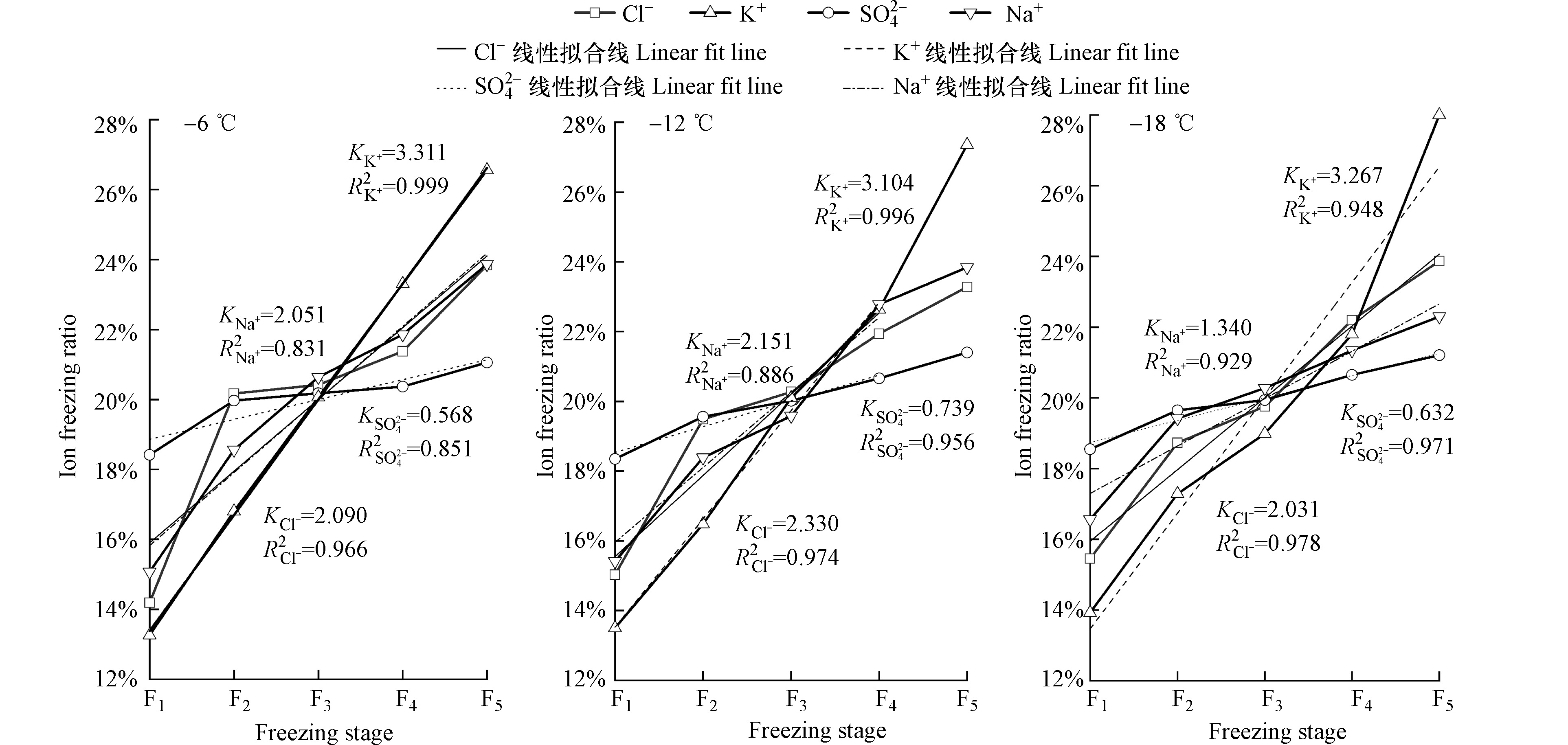
如图6所示,不同温度处理下,各离子变化趋势相似,皆随冷冻级数的增加而上升。冷冻级数为F1时,各离子结冰比率呈
$\mathrm{SO}_{4}^{2-} $ >Na+>Cl−>K+的分布规律,各温度处理下$\mathrm{SO}_{4}^{2-} $ 结冰比率高于其他离子,达18.35%—18.55%;Cl−、Na+结冰比率为14.19%—16.56%,K+结冰比率最低,达13.26%—13.92%。结冰过程中各离子迁移速率存在差异,这与离子自身性质(离子半径、离子带电荷数)有关,在冷冻温度、初始浓度、离子电荷量一致的条件下,离子半径越大、作用力越弱、与H2O的作用距离越长、与冰的结合能越小[26]。因此,相同温度处理下,F1冷冻级数冰中Na+结冰比率大于K+结冰比率,而$\mathrm{SO}_{4}^{2-} $ 结冰比率大于Cl−结冰比率,可能是由于$\mathrm{SO}_{4}^{2-} $ 为负二价离子,与氢键缔合的结合能较Cl−更大。F5时K+结冰比率达到最高,较−6 ℃处理的Cl−、
$\mathrm{SO}_{4}^{2-} $ 、Na+结冰比率提高 2.72%、5.50%、2.69%,较−12 ℃时提高4.08%、5.96 %、3.52 %,较−18 ℃时提高4.13 %、6.79 %、5.70%。不同温度处理下,由F1至F5过程中,各离子结冰比率拟合线斜率呈K+>Cl−>Na+>$\mathrm{SO}_{4}^{2-} $ 的分布规律,说明K+结冰比率上升趋势最为显著,$\mathrm{SO}_{4}^{2-} $ 最在各级冰体中占比差异最小。综上所述,不同离子在冷冻过程中进入冰体的数量存在差异。 -
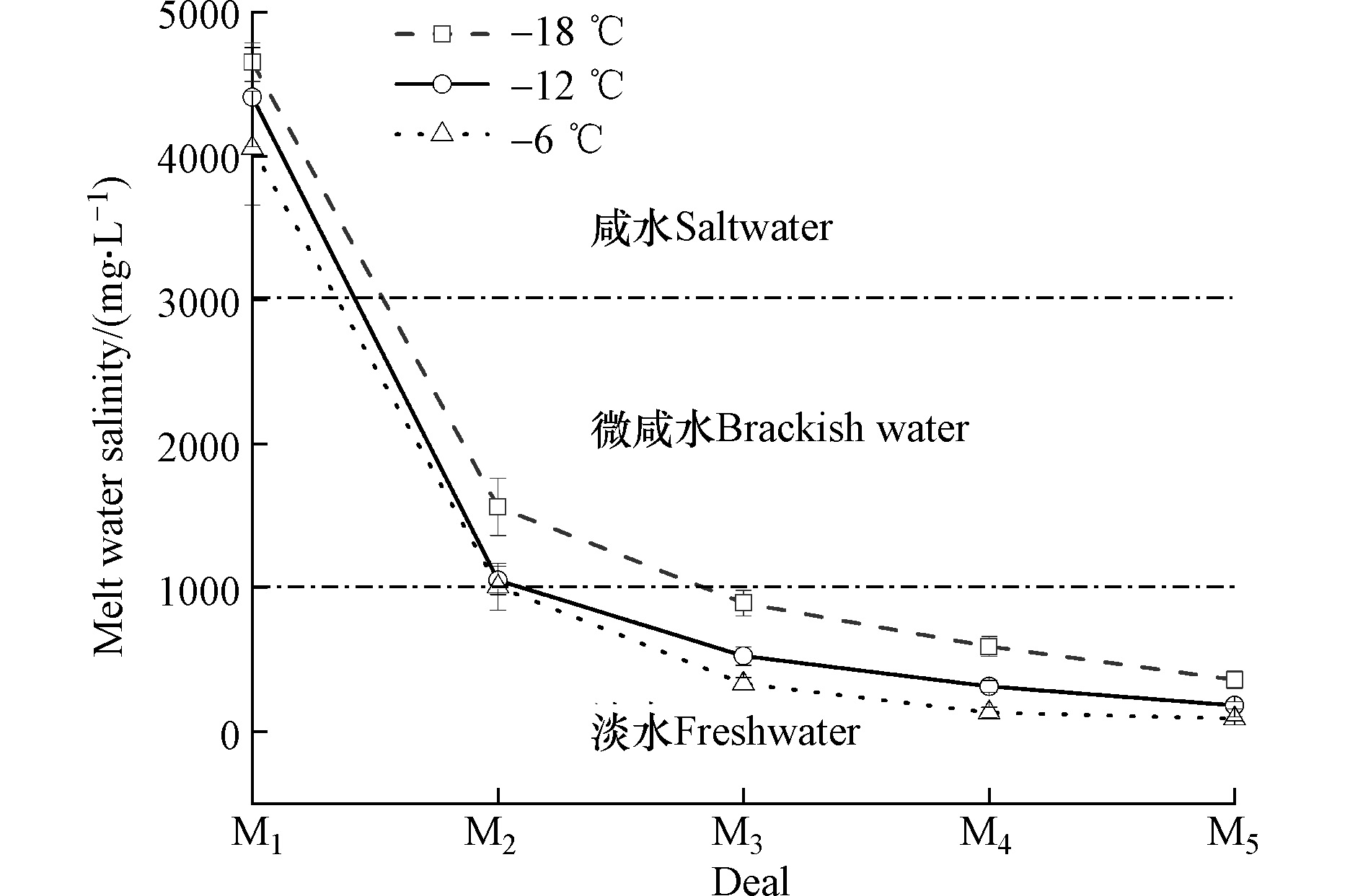
为保证初始原水矿化度一致,取F1级重力脱盐冰融水进行分析。融冰试验中冰融水矿化度变化可分为3个阶段:第一阶段(M1—M2)、第二阶段(M2—M4)、第三阶段(M4—M5)。分析图7可知,由M1至M5,冰融水矿化度呈逐渐降低趋势。−18 ℃、−12 ℃、−6 ℃时,第一阶段冰融水矿化度由4651.24、4409.26、4055.27 mg·L−1下降至1561.20、1051.23、1006.46 mg·L−1,第二阶段冰融水矿化度由1561.20、1051.23、1006.46 mg·L−1下降至591.56、313.47、131.72 mg·L−1,第三阶段冰融水矿化度由591.56、313.47、131.72 mg·L−1下降至361.23、182.26、89.34 mg·L−1;其中,第二阶段冰融水矿化度变化较第一阶段明显变缓,第三阶段冰融水矿化度变化趋于平缓,−18 ℃时,M1的后半段至M3的前半段为微咸水(1000—3000 mg·L−1),M3至M5为淡水(<1000 mg·L−1);−12 ℃与−6 ℃时,M1至M2后半段为微咸水,M2至M5为淡水,表明M1至M5冰融水矿化度逐渐降低,融冰过程呈现高浓度咸水先融,低浓度咸水后融的规律。
-
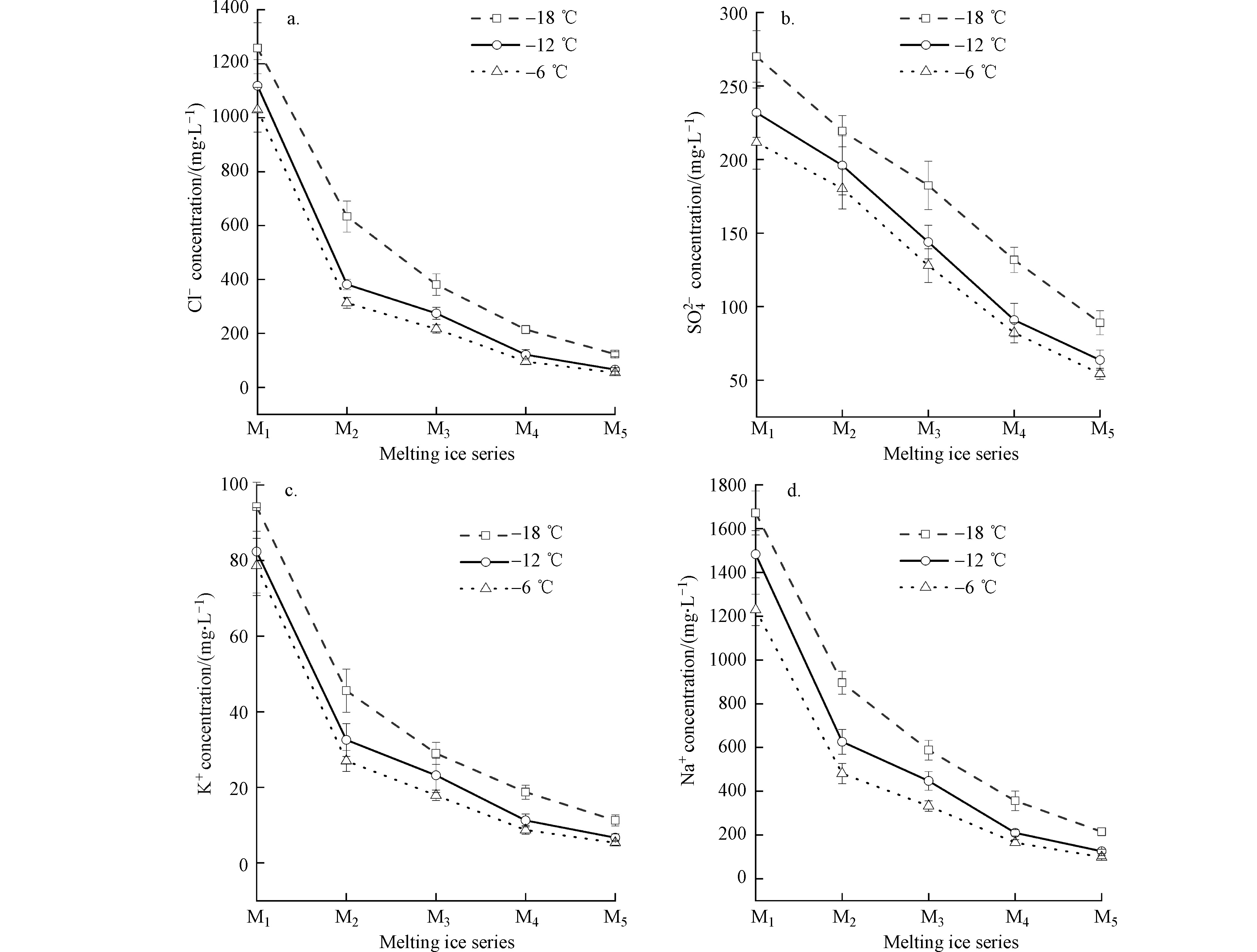
由图8可知,不同温度处理下,各离子浓度由M1至M5显著降低(P<0.05);Cl−、Na+、K+含量变化趋势与冰融水矿化度变化趋势相似,均呈现先快速下降、后缓慢、最终趋于平稳的变化趋势,
$\mathrm{SO}_{4}^{2-} $ 浓度下降趋势平稳。综上所述,不同温度处理下,由M1至M5冰融水矿化度、Cl−、K+、Na+浓度均呈现先快速下降、后缓慢、最终趋于平稳的变化趋势。由离子在冰体中的分布规律可知,离子多存在于冰体的表面或冰晶界面;冰体融化初期主要为表面冰的相变化,因此融出离子浓度较大;后期融出离子多存在于晶体内部,含量相对较少[27]。由冰的融化机理可知,离子以液袋的形式被捕获于冰晶中,在冰的融化初期,冰体逐渐融化,液袋发生向下迁移,冰体内部形成排水通道,离子沿排水通道融出,所以各离子呈现前期集中释放,中后期相对平稳均匀释放的趋势[19],与本实验结果相似。咸水结冰融化入渗土壤过程,高离子浓度咸水先入渗,此时,对土壤盐分与离子主要起交换和淋洗作用,后期融水离子浓度较低,对土壤盐分与离子主要起淋洗作用[28],使其向下迁移,进入土壤深层,从而促进土壤表层脱盐,减轻对植物根系的作用[29]。
由表2可知,各温度处理下,M1中离子融出比例呈Cl−>K+>Na+>
$\mathrm{SO}_{4}^{2-} $ 的分布规律,其中,Cl−融出比例均为最高,占Cl−总浓度的48.19%—60.23%;K+、Na+融出比例为44.86%—57.17%;$\mathrm{SO}_{4}^{2-} $ 融出比例最低,达30.25%—32.24%;由M1至M5过程中,Cl−、K+及Na+含量占离子总浓度的比例下降,其中在−6 ℃时降幅最大,降低了14.91%;相反地,$\mathrm{SO}_{4}^{2-} $ 含量占离子总含量的比例上升,−6 ℃时增幅最大,为17.28%,这与离子特性有关[19],说明不同离子在融化过程中融出比例存在差异,冰融水由最初的Na-Cl型咸水,逐渐转变成SO4·Cl-Na型的微咸水,融冰过程也是离子重新分配的过程。 -
(1)结冰试验中,冰融水矿化度与离子浓度随冷冻温度的降低而上升。不同温度处理下,各级冰融水矿化度与离子浓度相较于初始水样(F0)均显著降低(P<0.05),F1级到F5级冰融水矿化度与离子浓度逐渐升高。各离子进入冰晶的迁移速率存在差异,离子结冰比率拟合线斜率呈K+>Cl−>Na+>
$\mathrm{SO}_{4}^{2-} $ 的分布规律。(2)融冰试验中,由M1至M5冰融水矿化度、Cl−、K+、Na+浓度呈现先快速下降、后缓慢下降、最终趋于平稳的变化趋势;
$\mathrm{SO}_{4}^{2-} $ 浓度下降趋势平稳。冰融水由最初的Na−Cl型咸水,逐渐转变成SO4·Cl−Na型的微咸水。
南疆咸水结冰与融化过程中盐分及离子的变化
Changes of salt and ions in the process of salt water freezing and melting in Southern Xinjiang
-
摘要: 咸水淡化是缓解淡水资源紧张,减轻水体污染的有效途径。本试验分析了南疆农田排水不同冷冻温度下,结冰与融化过程中矿化度及离子浓度的分布规律;探讨了基于多级冷冻与重力脱盐条件下,盐分及Cl−、
$\mathrm{SO}_{4}^{2-} $ 、K+、Na+的迁移规律成因。试验结果表明,冰融水矿化度与离子浓度随冷冻温度的降低而上升;不同温度处理下,各级冰融水矿化度与离子浓度相较于初始水样(F0)均显著降低(P<0.05),冰晶逐级冷冻过程中,冰融水矿化度与离子浓度逐渐升高;各离子进入冰晶的迁移速率存在差异,离子结冰比率拟合线斜率呈K+> Cl−>Na+>$\mathrm{SO}_{4}^{2-} $ 的分布规律。冰晶逐级融化过程中,冰融水矿化度、Cl−、K+、Na+浓度呈现先快速下降、后缓慢下降、最终趋于平稳的变化趋势;$\mathrm{SO}_{4}^{2-} $ 浓度下降趋势平稳。冰融水由最初的Na−Cl型咸水,逐渐转变成SO4·Cl−Na型的微咸水。本文结论可为南疆咸水冷冻法淡化技术应用提供依据Abstract: Salt water desalination is an effective way to relieve the shortage of fresh water resources and water pollution. This article analyses the distribution of salinity and ion concentration during freezing and melting of farmland drainage in Southern Xinjiang under different freezing temperatures; which explains the migration reason of salt, Cl−,$\mathrm{SO}_{4}^{2-} $ , K+, and Na+ under the condition of multistage freezing and gravity desalting. The results show that the salinity and ion concentration of ice melt water increase with the decrease of freezing temperature. Under the various temperatures, compared with the initial water sample (F0), the salinity and ion concentration of ice melt water significantly decreased (P < 0.05), during the gradual freezing of ice crystals, the salinity and ion concentration of ice melt water gradually increased. The migration rate of different ions getting into ice crystals is different, and the slope of fitting line of ion freezing ratio is K+>Cl−>Na+>$\mathrm{SO}_{4}^{2-} $ . In the process of melting ice crystals step by step the salinity of ice melt water, concentrations of Cl−, K+ and Na+ decrease rapidly first, then decrease slowly, and finally get stabilized. The concentration of$\mathrm{SO}_{4}^{2-} $ declines steadily. The ice melt water gradually changes from Na−Cl salt water to SO4·Cl−Na brackish water. The conclusions of this article can provide a basis for the application of salt water freezing desalination technology in Southern Xinjiang.-
Key words:
- desalination of salt water /
- salinity /
- ion migration /
- icing process /
- melting process
-

-
表 1 供试原水水质指标(mg·L−1)
Table 1. Test raw water quality index(mg·L−1)
矿化度
SalinitypH Cl− $ \mathrm{SO}_{4}^{2-}$ Na+ K+ 4906.77 8.36 1291.10 282.59 1789.58 104.91 表 2 咸水冰M1—M5离子变化
Table 2. Salt water ice M1—M5 ion change
冷冻温度/℃
Freezing temperature融水
Melt waterCl−/
(mg·L−1)$\mathrm{SO}_{4}^{2-} $
(mg·L−1)K+/
(mg·L−)Na+/
(mg·L−1)−6 M1 1029.23±83.11a 211.67±18.21a 78.68±7.24a 1229.14±71.61a M2 313.03±19.85b 180.26±13.76b 27.00±2.75b 480.56±45.71b M3 216.47±15.37b 127.92±11.51c 17.88±1.40c 332.54±23.54c M4 95.83±10.40c 82.25±6.83d 8.73±1.16d 164.95±13.13d M5 54.17 ±6.90c 54.40±3.74d 5.53±0.64d 98.78±6.90d −12 M1 1118.23±96.94a 231.91±16.76a 82.38±11.60a 1483.32±108.14a M2 381.34±19.29b 196.03±20.07b 32.54±4.35b 625.83±56.64b M3 274.50±22.23b 144.01±11.47c 23.17±4.56bc 436.95±42.77c M4 120.97±18.95c 90.92±11.32d 11.24±1.70cd 209.69±15.30d M5 65.73±6.99c 63.69±6.79d 6.68±1.02d 125.60±9.80d −18 M1 1257.73±94.81a 270.04±17.52a 94.26±6.51a 1671.67±100.56a M2 633.77±57.89b 219.30±10.66b 45.61±5.72b 896.18±52.69b M3 381.10±40.01c 182.42±16.46c 28.97±2.29c 587.65±45.48c M4 214.27±15.64d 131.83±8.68d 16.72±1.85d 356.21±44.61cd M5 123.10±9.21d 89.07±8.15e 10.05±1.44d 214.86±18.66d 注:表中数据为平均值±标准差;同列不同小写字母表示差异性显著(P<0.05).
Note: The data in the table are the mean ± standard deviation; different lowercase letters in the same column indicate significant differences(P<0.05). -
[1] 王新. 新疆水安全现状与形势浅析 [J]. 水利规划与设计, 2021(3): 38-42,76. doi: 10.3969/j.issn.1672-2469.2021.03.010 WANG X. Analysis of current situation and situation of water security in Xinjiang [J]. Water Resources Planning and Design, 2021(3): 38-42,76(in Chinese). doi: 10.3969/j.issn.1672-2469.2021.03.010
[2] 李江, 龙爱华. 近60年新疆水资源变化及可持续利用思考 [J]. 水利规划与设计, 2021(7): 1-5,72. doi: 10.3969/j.issn.1672-2469.2021.07.001 LI J, LONG A H. Thoughts on water resources change and sustainable utilization in Xinjiang in recent 60 years [J]. Water Resources Planning and Design, 2021(7): 1-5,72(in Chinese). doi: 10.3969/j.issn.1672-2469.2021.07.001
[3] 蔺卿. 新疆水生态文明建设的水资源保护利用策略研究 [J]. 干旱区地理, 2021, 44(5): 1483-1488. doi: 10.12118/j.issn.10006060.2021.05.28 LIN Q. Water resources protection and utilization strategy for water ecological civilization construction in Xinjiang [J]. Arid Land Geography, 2021, 44(5): 1483-1488(in Chinese). doi: 10.12118/j.issn.10006060.2021.05.28
[4] 吴琼, 梁伊, 高凡, 等. 新疆阿拉尔市苦咸水水化学特征、分布及成因分析 [J]. 环境化学, 2021, 40(3): 737-745. doi: 10.7524/j.issn.0254-6108.2020082202 WU Q, LIANG Y, GAO F, et al. Analysis of chemical characteristics, distribution and cause of formation of brackish water in Alar City, Xinjiang [J]. Environmental Chemistry, 2021, 40(3): 737-745(in Chinese). doi: 10.7524/j.issn.0254-6108.2020082202
[5] 曾妍妍, 周殷竹, 周金龙, 等. 新疆南部典型地区地下水重金属空间分布特征 [J]. 环境化学, 2015, 34(12): 2310-2312. doi: 10.7524/j.issn.0254-6108.2015.12.2015073002 ZENG Y Y, ZHOU Y Z, ZHOU J L, et al. Spatial distribution characteristics of heavy metals in groundwater in typical areas of southern Xinjiang [J]. Environmental Chemistry, 2015, 34(12): 2310-2312(in Chinese). doi: 10.7524/j.issn.0254-6108.2015.12.2015073002
[6] 董晓梅, 杨玉辉, 李朝阳, 等. 阿拉尔灌区农田排水水质水量变化规律分析 [J]. 水资源与水工程学报, 2017, 28(5): 250-254. doi: 10.11705/j.issn.1672-643X.2017.05.42 DONG X M, YANG Y H, LI Z Y, et al. Variation law analysis of Alar irrigated area farmland drainage quality and quantity [J]. Journal of Water Resources and Water Engineering, 2017, 28(5): 250-254(in Chinese). doi: 10.11705/j.issn.1672-643X.2017.05.42
[7] 杨玉辉, 王兴鹏, 李朝阳, 等. 基于单极冷冻法的南疆咸水淡化及应用 [J]. 环境工程, 2019, 37(12): 126-131,136. YANG Y H, WANG X P, LI Z Y, et al. Desalination and application of saline water in southern Xinjiang based on unidirectional freezing [J]. Environmental Engineering, 2019, 37(12): 126-131,136(in Chinese).
[8] 张璐, 杨帆, 王志春. 咸水结冰灌溉融水淋盐改良盐渍土研究进展 [J]. 土壤与作物, 2021, 10(2): 202-212. doi: 10.11689/j.issn.2095-2961.2021.02.009 ZHANG L, YANG F, WANG Z C. Research advances of saline soil reclamation by freezing saline water irrigation and meltwater leaching [J]. Soils and Crops, 2021, 10(2): 202-212(in Chinese). doi: 10.11689/j.issn.2095-2961.2021.02.009
[9] 郭凯, 刘小京, 封晓辉, 等. 冬季咸水结冰灌溉对河套重盐碱地改良效果研究 [J]. 中国生态农业学报(中英文), 2021, 29(4): 640-648. GUO K, LIU X J, FENG X H, et al. Reclamation effect of freezing saline water irrigation in winter season on the heavy saline-alkali soil in Hetao Irrigation District [J]. Chinese Journal of Eco-Agriculture, 2021, 29(4): 640-648(in Chinese).
[10] 马中昇, 谭军利, 魏童. 中国微咸水利用的地区和作物适应性研究进展 [J]. 灌溉排水学报, 2019, 38(3): 70-75. MA Z S, TAN J L, WEI T. The variation of salt-tolerance of crops in different regions irrigated with brackish water in China [J]. Journal of Irrigation and Drainage, 2019, 38(3): 70-75(in Chinese).
[11] 杨玉辉, 王兴鹏, 李朝阳, 等. 反渗透技术对南疆咸水淡化处理的适应性 [J]. 水处理技术, 2019, 45(10): 121-124. YANG Y H, WANG X P, LI Z Y, et al. Adaptability of reverse osmosis technology for salt water desalination in southern Xinjiang [J]. Technology of Water Treatment, 2019, 45(10): 121-124(in Chinese).
[12] 段作山, 王向举, 马小蕾, 等. 反渗透除氟除氨氮技术在饮用水中的应用 [J]. 净水技术, 2021, 40(3): 139-143. DUAN Z S, WANG X J, MA X L, et al. Application of enhanced fluoride and ammonia removal by reverse osmosis in drinking water [J]. Water Purification Technology, 2021, 40(3): 139-143(in Chinese).
[13] 刘茂举. 磷酸铁生产废水的反渗透膜处理及回收利用[D]. 南宁: 广西大学, 2020. LIU M J. Reverse osmosis membrane treatment and recycling utilization of waste water of FePO4[D]. Nanning: Guangxi University, 2020(in Chinese).
[14] 潘俊杰, 杨芃. 高效低耗电渗析技术去除地下水硝酸盐的试验研究[C]//第七届中国城镇水务发展国际研讨会论文集. 宁波, 2012: 4-12. [15] 郑智颖, 李凤臣, 李倩, 等. 海水淡化技术应用研究及发展现状 [J]. 科学通报, 2016, 61(21): 2344-2370. doi: 10.1360/N972015-00829 ZHENG Z Y, LI F C, LI Q, et al. State-of-the-art of R & D on seawater desalination technology [J]. Chinese Science Bulletin, 2016, 61(21): 2344-2370(in Chinese). doi: 10.1360/N972015-00829
[16] 杨晖, 李恒松, 张思健, 等. 基于冷冻过程的海水脱盐研究 [J]. 水处理技术, 2016, 42(7): 57-61. YANG H, LI H S, ZHANG S J, et al. Saline water desalination investigation on the basis of freezing process [J]. Technology of Water Treatment, 2016, 42(7): 57-61(in Chinese).
[17] 陈栋, 张朝升, 荣宏伟. 低成本冷冻法海水淡化技术 [J]. 净水技术, 2019, 38(1): 122-127. CHEN D, ZHANG C S, RONG H W. Technology of low cost freezing method for seawater desalination [J]. Water Purification Technology, 2019, 38(1): 122-127(in Chinese).
[18] 孙正阳. LNG冷能用于冷冻脱盐与区域供冷的研究[D]. 北京: 北京建筑大学, 2018. SUN Z Y. Study on LNG cold energy used in freezing desalination and district cooling[D]. Beijing: Beijing University of Civil Engineering and Architecture, 2018(in Chinese).
[19] 张岩, 任方云, 唐元庆, 等. 融冰过程中铁离子和锰离子的迁移规律 [J]. 中国环境科学, 2021, 41(5): 2391-2398. doi: 10.3969/j.issn.1000-6923.2021.05.047 ZHANG Y, REN F Y, TANG Y Q, et al. Migration of iron and manganese ions during ice melting [J]. China Environmental Science, 2021, 41(5): 2391-2398(in Chinese). doi: 10.3969/j.issn.1000-6923.2021.05.047
[20] SHANNON M C, GRIEVE C M. Options for using low-quality water for vegetable crops [J]. HortScience, 2000, 35(6): 1058-1062. doi: 10.21273/HORTSCI.35.6.1058 [21] ROOS A C, VERSCHUUR R J, SCHREURS B, et al. Development of a vacuum crystallizer for the freeze concentration of industrial waste water [J]. Chemical Engineering Research and Design, 2003, 81(8): 881-892. doi: 10.1205/026387603322482121 [22] 李晓洋, 崔康平, 席慕凡, 等. 多级冷冻工艺对高盐高浓度有机废水的处理效果及去除机理 [J]. 环境工程学报, 2020, 14(3): 652-661. doi: 10.12030/j.cjee.201905163 LI X Y, CUI K P, XI M F, et al. Treatment performance and mechanism of high-salt and high-concentration organic wastewater using multi-stage freezing technology [J]. Chinese Journal of Environmental Engineering, 2020, 14(3): 652-661(in Chinese). doi: 10.12030/j.cjee.201905163
[23] 张岩. 乌梁素海结冰过程中污染物迁移机理及其应用研究[D]. 呼和浩特: 内蒙古农业大学, 2012. ZHANG Y. Migration mechanism of pollutants and its application in ulansuhai lake in freezing process[D]. Hohhot: Inner Mongolia Agricultural University, 2012(in Chinese).
[24] SCHMIDT S, MOSKAL W, de MORA S J, et al. Limnological properties of Antarctic ponds during winter freezing [J]. Antarctic Science, 1991, 3(4): 379-388. doi: 10.1017/S0954102091000482 [25] 孙驰. 基于第一性原理的乌梁素海冰、水介质中重金属迁移特征研究[D]. 呼和浩特: 内蒙古农业大学, 2019. SUN C. Transport characteristics of heavy metals based on first principle in ice and water medium in the lake ulansuhai[D]. Hohhot: Inner Mongolia Agricultural University, 2019(in Chinese).
[26] 唐元庆. 水体结冰和融冰过程中铁、锰、钙、镁的迁移规律研究[D]. 烟台: 烟台大学, 2020. TANG Y Q. The migration law of iron, manganese, calcium and magnesium in water icing and melting processes[D]. Yantai, China: Yantai University, 2020(in Chinese).
[27] BRIMBLECOMBE P, CLEGG S L, DAVIES T D, et al. Observations of the preferential loss of major ions from melting snow and laboratory ice [J]. Water Research, 1987, 21(10): 1279-1286. doi: 10.1016/0043-1354(87)90181-3 [28] 潘洁, 肖辉, 王立艳, 等. 咸水冰融化与土壤入渗过程不同盐分离子迁移规律研究 [J]. 华北农学报, 2012, 27(1): 210-214. doi: 10.3969/j.issn.1000-7091.2012.01.039 PAN J, XIAO H, WANG L Y, et al. Study on migration of different salt ions in melting and infiltration processes of saline water ice [J]. Acta Agriculturae Boreali-Sinica, 2012, 27(1): 210-214(in Chinese). doi: 10.3969/j.issn.1000-7091.2012.01.039
[29] 车升国, 左余宝, 林治安, 等. 黄淮海地区地下微咸水资源农业灌溉模拟研究 [J]. 农业环境科学学报, 2011, 30(3): 611-612. CHE S G, ZUO Y B, LIN Z A, et al. Simulation study of agricultural irrigation with saline groundwater in the Huang-Huai-Hai region, China [J]. Journal of Agro-Environment Science, 2011, 30(3): 611-612(in Chinese).
-









 下载:
下载: