-
全氟辛烷磺酸(perfluorooctane sulfonate,PFOS)具有大量的C—F键,极性高、稳定性强,因其链端具有亲水性磺酸基(—SO3H)而能部分溶解于水。PFOS具有较低的水表面张力和优异的化学稳定性,通常被用作铬雾抑制剂广泛用于硬铬电镀中[1]。然而,PFOS具有持久性、生物累积性和生物放大性,会引发许多毒性作用[2-4]。因而,PFOS被列为持久性有机污染物(persistent organic pollutants, POPs),在环境水体中检出的质量浓度可达10~50 ng·L−1 [5]。全氟烷基醚磺酸盐(F-53B)是PFOS的一种替代品,在中国电镀行业使用了30多年,一直未受管控[5],导致大量F-53B释放到自然水体中,检出水平与PFOS接近,达到10~50 ng·L−1。虽然F-53B在自然水体中检出的质量浓度较低,但考虑到F-53B具有环境持久性和生物累积性[5],低浓度的F-53B可能造成的健康危害也需引起足够的重视。有研究表明,F-53B的毒性竟然相当于甚至高于PFOS[6-7]。赵楠等[8]的研究也表明大鼠暴露于F-53B后可能会影响血清的氧化应激反应,引发炎症、心血管疾病、糖尿病和高血压,引起神经毒性且抑制大脑发育。因此,消除F-53B对水环境污染势在必行。
对水中F-53B的去除技术主要有吸附、电化学氧化、光降解和机械化学降解等[9-12]。由于F-53B的稳定性较强,电化学氧化、光降解和机械化学降解等技术的脱氟率较低,并不能完全降解F-53B,而吸附法则被认为是去除水中F-53B的最有效策略之一。目前关于活性炭和离子交换树脂吸附F-53B的研究报道较多,这2种吸附剂均能有效吸附去除水中的F-53B [9, 13-14],但在实际应用过程中还存在运行成本较高和易受阴离子影响等问题。近年来研究表明金属-有机骨架(metal-organic frameworks, MOFs)是一种有潜力的环境处理材料[15-17],可用于污染修复和环境监测等场景。这种由金属离子或金属簇与有机配体进行配位组装而成的多孔晶体材料[18]具有高比表面积、高孔隙率及结构/功能可调等作为吸附剂的优势。有研究[19-23]表明,MOFs可高效去除水中的重金属、染料和抗生素等,在PFOS捕获方面也表现出优异性能[24-26],这主要归因于MOFs材料的比表面积大、活性反应位点多及孔隙结构的限域作用。
MOFs材料的金属中心是重要的活性反应位点,先前的研究主要集中在过渡金属(如Cr、Fe、Co、Zn、Zr等)和镧系/锕系金属作为MOFs材料的金属中心[24-25, 27],所需成本较高。采用Ni作为MOFs材料的金属中心具有价格低廉、反应活性高等优点[28],但目前对于Ni-MOFs材料吸附F-53B的研究还鲜有报道。此外,研究发现,较大的分子尺寸限制了大分子有机物在三维MOFs材料孔道中的传输,吸附受限于表面。二维MOFs材料具有更为发达的孔隙结构,可以有效降低分子尺寸对有机物传输的限制,加上二维MOFs片层结构的限域作用,可以很好地解决该问题[29-30]。因此,尝试制备二维Ni-MOFs有望提高水中F-53B的吸附效能。
本研究采用溶剂热法通过控制合成温度制备二维和三维Ni-MOFs材料,利用X射线衍射(XRD)、傅里叶变换红外光谱(FTIR)、扫描电子显微镜(SEM)、N2吸/脱附实验和热重分析(TG)对材料的结构形貌和热稳定性进行了表征分析,采用动力学和热力学实验以及对比分析,开展了对水中F-53B的吸附研究,并进一步结合理论计算探究了可能的吸附机理。
-
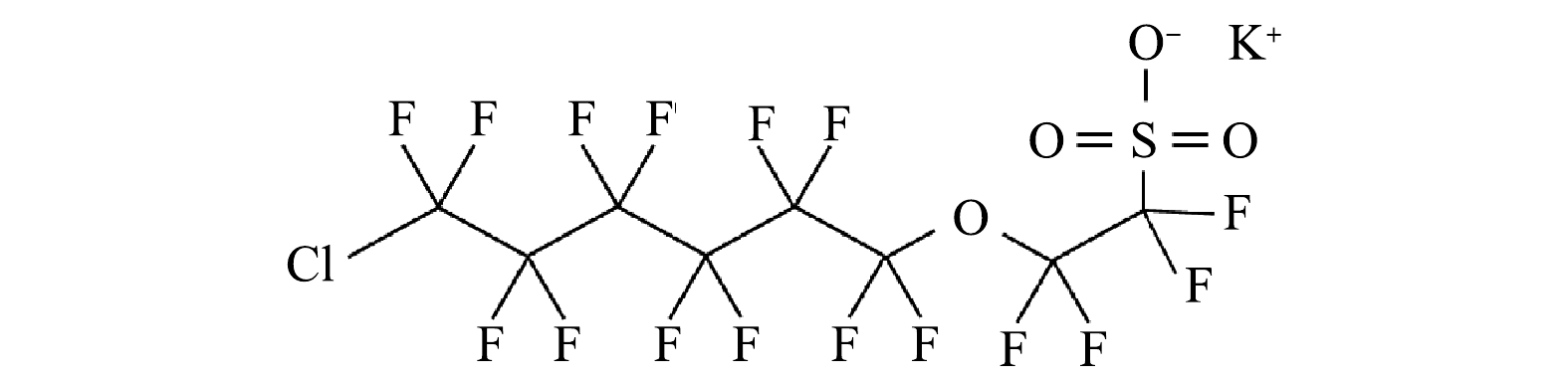
四水醋酸镍(Ni(OAc)2·4H2O)和对苯二甲酸(BDC)购自北京百灵威科技有限公司,N, N-二甲基乙酰胺(DMA)购自天津市大茂化学试剂厂,超纯水购自屈臣氏,F-53B(图1)购自上海江莱生物科技有限公司,去离子水为实验室自制。所有化学药品均为分析纯。
-
1) Ni-MOFs的制备方法。根据前人[31]的研究报道,通过改变合成温度可以获得不同形貌的MOFs,因此,本研究采用了不同的合成温度来制备二维和三维的Ni-MOFs。分别将1.2 mmol Ni(OAc)2·4H2O和0.5 mmol BDC溶于60 mL去离子水和60 mL DMA中,并将2种溶液混合,在150 ºC回流下搅拌3 h。随后抽滤、洗涤并真空干燥最后得到浅绿色粉末,记为二维Ni-MOFs。此外,将回流温度调整为100 ºC,制得产物记为三维Ni-MOFs。
2)表征与分析测试方法。采用德国布鲁克公司的D8型X射线衍射仪(XRD)分析材料的晶型结构。将材料研磨成粉末后装入样品载物台,用干净的玻璃片压平样品,随后上机测试。测试条件为:Kα 辐射,管压40 kV,管电流20 mA,扫描速度为10°·min−1,步长0.02°,扫描范围为5°~50°。采用德国布鲁克公司的Vector 33型傅里叶变换红外光谱仪(FTIR)分析样品的官能基团信息。取适量样品以质量比为1∶100 与溴化钾粉末混匀,于红外灯烘烤下研磨,压片成形后上机测试,扫描范围为4 000~400 cm−1,分辨率为4 cm−1,扫描次数为32。采用日本日立公司的SC8820型扫描电镜观察样品的形貌特征。将样品分散在贴有导电胶的样品托盘上,上机测试。在不同放大倍数下观测样品的微观形貌特征。采用美国麦克仪器公司的ASAP2460型比表面与孔隙度分析仪测试材料的孔道特征和比表面积,计算方法采用Barrtt Emmett Teller (BET)算法。采用美国TA公司的TGA 550型热重分析仪测试样品的失重曲线(TG)和失重速率(DTG)曲线。采用UPLC-MS/MS (液相为赛默飞UltiMate 3000型,质谱为赛默飞TSQ Vantage型)检测水样F-53B的质量浓度,色谱柱选用Agilent HPH-C18 (规格为100 mm×2.1 mm,2.7 µm),流动相为乙腈(A)和含0.05%乙酸的5 mmol·L−1乙酸铵(B),流动相A比例为60%,流动相B比例为40%,样品采集时间为2 min。
3)吸附实验方法。吸附实验操作如下:在100 mL锥形瓶中加入10 mg Ni-MOFs粉末,再加入50 mL超纯水配制的一定质量浓度的F-53B溶液,接着将锥形瓶置于立式恒温振荡培养箱中恒温振荡一定时间(25 °C,180 r·min−1),反应过程中pH维持在7.0±0.3。振荡后用注射器抽取溶液,通过0.22 µm滤膜,滤液置于2 mL色谱瓶中,随后测试F-53B的浓度
根据TI等[32]的研究报道,铬电镀厂周边污染水体中F-53B的质量浓度可达2.3 mg·L−1,并且考虑到本研究制备的Ni-MOFs对F-53B可能具有较大的吸附容量,因此,本研究采用质量浓度为mg·L−1级别的F-53B开展吸附动力学和热力学实验。吸附动力学实验在50 mg·L−1的F-53B溶液中进行,Ni-MOFs的投加量为0.2 g·L−1,实验24 h完成,每隔预定时间进行采样分析F-53B的质量浓度。吸附等温实验在5~150 mg·L−1的F-53B溶液中进行,Ni-MOFs的投加量为0.2 g·L−1,采样时间均为24 h以确保吸附达到平衡。
为探究共存无机离子对吸附F-53B的影响,将F-53B质量浓度设置为2 mg·L−1,Ni-MOFs添加量为0.3 g·L−1,采样时间均为24 h以确保吸附达到平衡。分别添加0、1、5、10和20 mmol·L−1的KCl、K2CO3、KNO3、K2SO4和K3PO4以考察共存无机离子对吸附性能的影响。
4)数据处理与理论计算方法分别采用准一级动力学(式(1))、准二级动力学(式(2))对吸附过程进行拟合,探究吸附过程的动力学规律。采用Langmuir(式(3))和Freundlich(式(4))等温线模型对实验数据进行拟合。Ni-MOFs对F-53B在t时刻的吸附量根据式(5)计算。
式中:qe和qt分别为平衡时和t时刻的吸附量(mg·g−1),t为吸附时间(min),k1为准一级动力吸附速率常数,k2为准二级动力吸附速率常数。
式中:Ce为平衡时溶液相的质量浓度,mg·L−1;qe和qm分别为平衡时和饱和时的吸附量,mg·g−1;KL为Langmuir平衡吸附常数,L·mg−1,KF为Freundlich平衡吸附常数,L·mg−1;n为Freundlich经验参数。
式中:qt为在t时刻的吸附量,mg·g−1;C0和Ct分别为F-53B的初始质量浓度和t时刻的质量浓度,mg·L−1;V为溶液体积,L;M为吸附剂的质量,g。
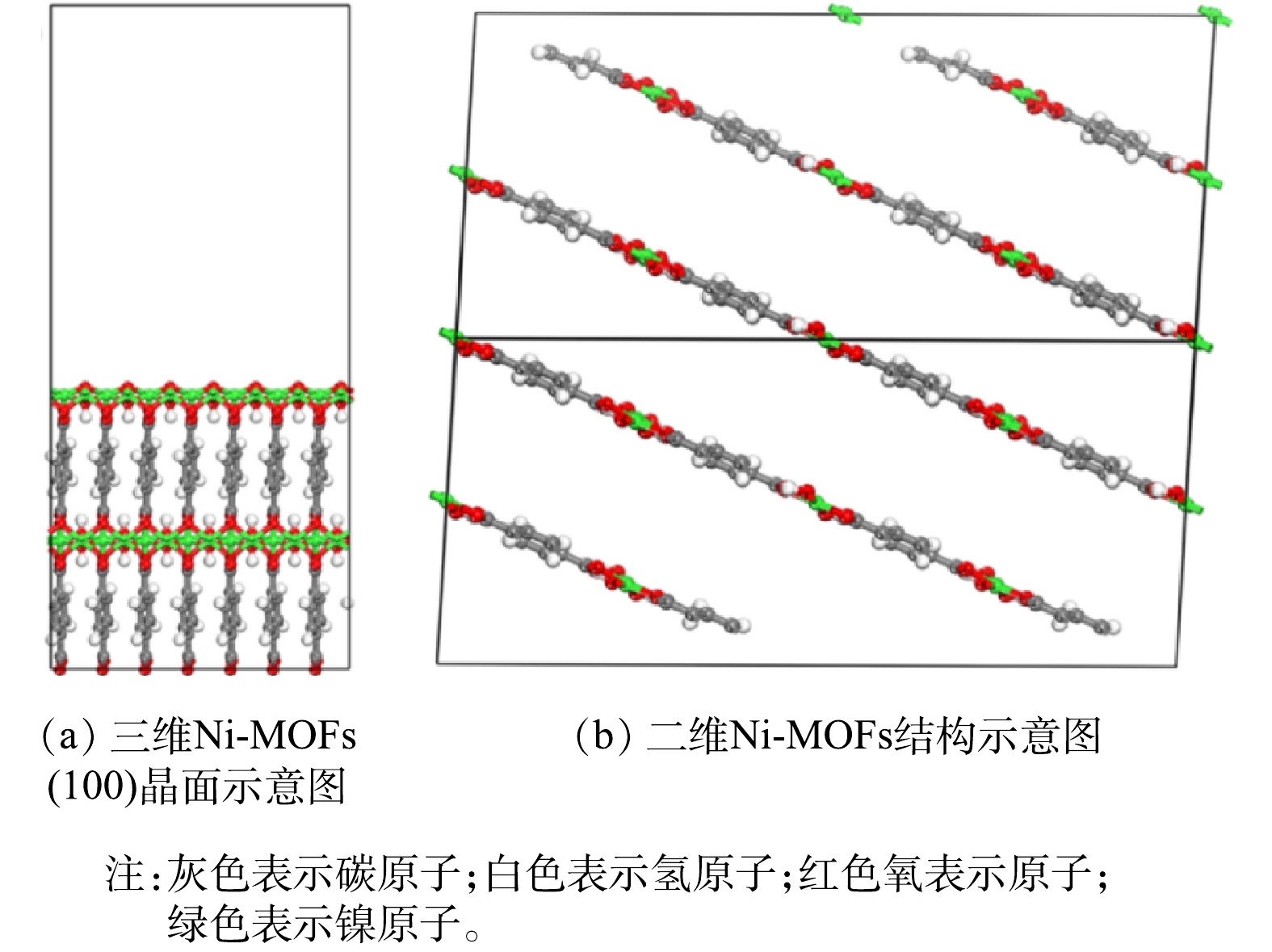
采用巨正则蒙特卡洛算法(grand canonical monte carlo, GCMC)结合密度泛函理论(density functional theory, DFT)计算描述两种Ni-MOFs材料对F-53B分子的吸附作用机理[33],所有的相关计算均在Materials Studio 7.0软件中进行。本文的三维Ni-MOFs晶体模型取自于剑桥晶体中心数据库[34],通过切除三维Ni-MOFs晶体的柱撑体得到二维Ni-MOFs晶体模型,并采用CASTEP模块对得到的两种MOFs模型进行结构优化,优化的精度为Fine,基组为GGA/PBE,通过Grimme对色散力进行矫正。基于优化后的三维模型,截取(100)晶面并在晶面上建立25 Å的真空层,相应的二维和三维Ni-MOFs结构如图2所示。采用Sorption模块中的Fixed loading任务计算MOFs骨架对F-53B分子的吸附作用能,吸附质-吸附剂和吸附质-吸附质之间的范德华相互作用由UFF力场[35]进行描述,而静电相互作用则采用EWALD方法计算,用于统计数据的初始和平衡总步长均为1.0×106步。利用Forcite模块中Dynamic任务的分子动力学方法计算了F-53B在骨架中的自由扩散系数,自由扩散系数根据式(6)计算,分子动力学计算采用NVT系综,步长为1 fs,用于统计均方分布(mean square distribution, MSD)数据的时间为5 ns。
式中:ri(t)是在t时刻下分子i的位置矢量。
另外,为定性比较三维Ni-MOFs表面和二维Ni-MOFs结构中不同吸附位点与吸附质分子中不同原子之间的相互作用,还计算了径向分布函数(radial distribution function, RDF)曲线。
-
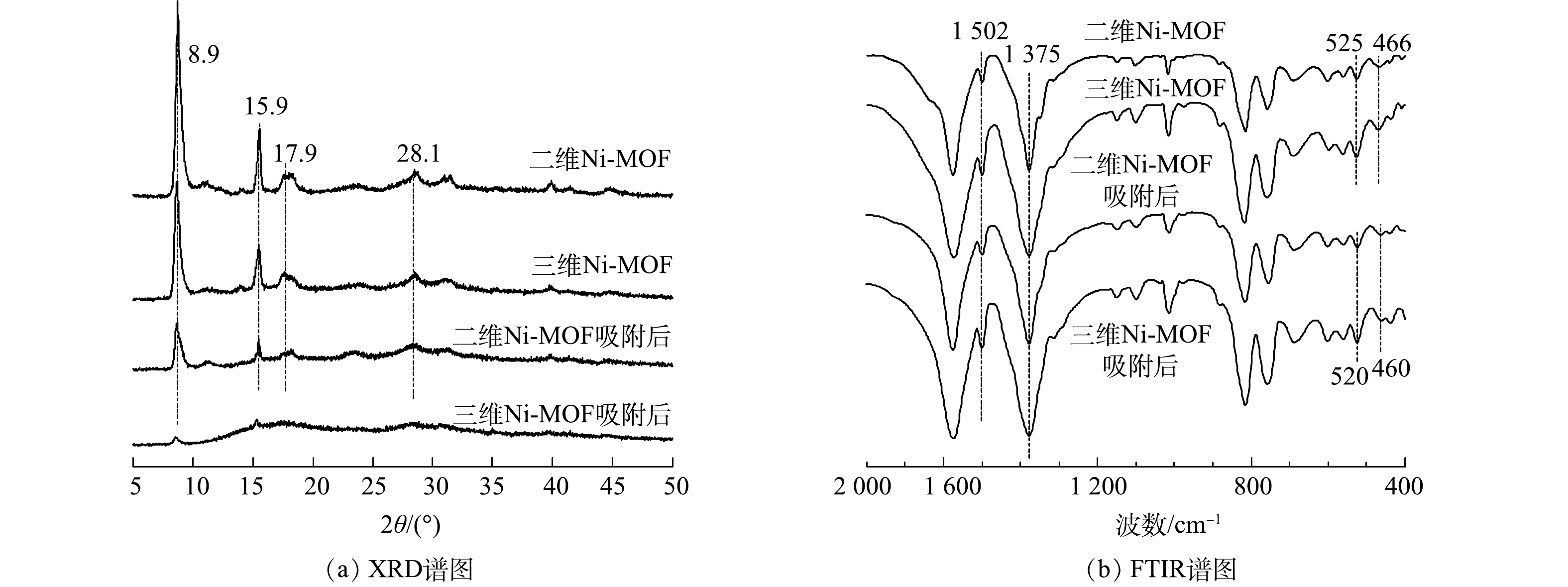
XRD对2种Ni-MOFs材料的晶型结构表征结果如图3(a)所示。二维Ni-MOFs和三维Ni-MOFs材料均在8.9o、15.9o、17. 9o和28.1o处具有明显的特征峰,分别对应于[Ni3(OH)2(C8H4O4)2(H2O)4]·2H2O (剑桥晶体数据中心,638866)的(100)、(101)、(210) 和(221)晶面,表明合成的材料确实为Ni-MOFs材料。图3(b)反映了2种Ni-MOFs材料的FTIR图谱。其中,位于466 cm−1和525 cm−1的特征峰为Ni—O伸缩振动峰[36],表明Ni成功与BDC配体结合。此外,位于1 375 cm−1和1 502 cm−1的特征峰分别为—COO−的对称和不对称拉伸振动峰,表明Ni与BDC配体以双齿配位模式进行结合[36]。
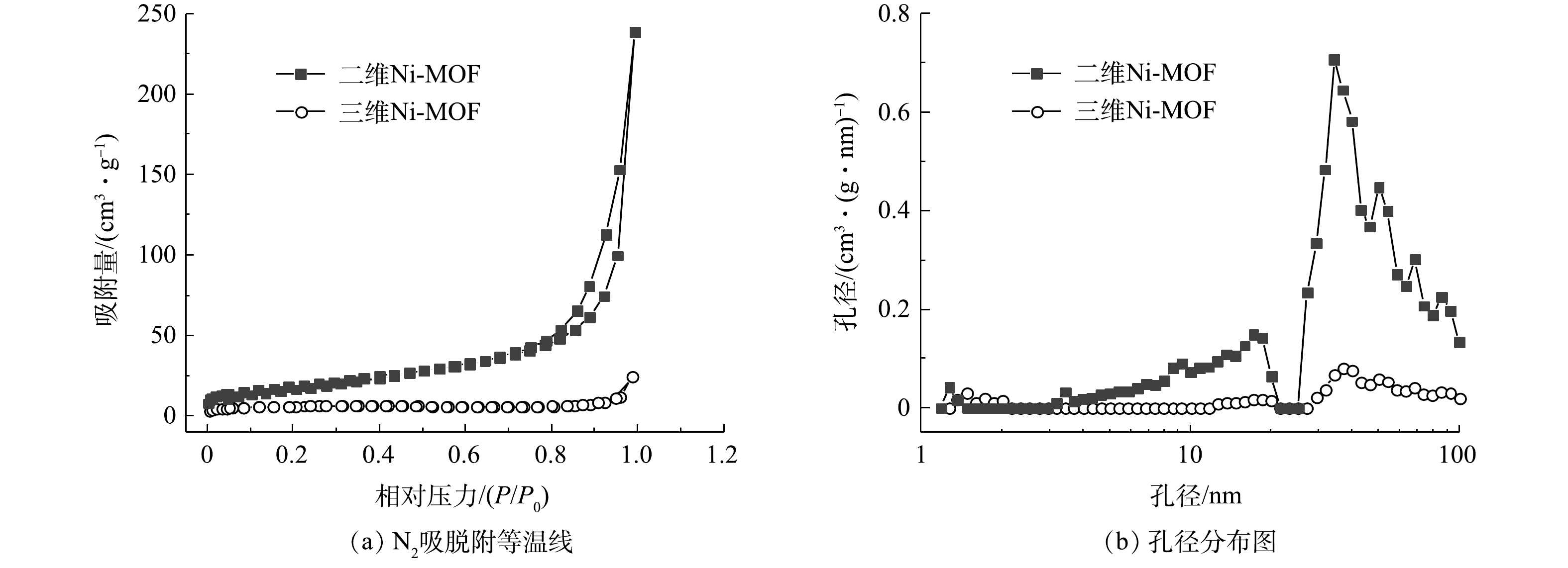
2种产物的SEM表征结果如图4所示。可见,三维Ni-MOFs材料呈现出明显的块状形貌,二维Ni-MOFs产物呈现较好的分散片层结构。这种片层结构使得活性位点充分暴露出来,有望在F-53B吸附测试中表现出优异的吸附性能。此外,这些二维Ni-MOFs微片的横向尺寸明显大于其厚度,从而产生弯曲或卷曲的形貌。这种弯曲或扭曲形成了明显的介孔结构,图5的孔径分布特征也证实了该结果。这些弯曲的微片结构为客体分子的传质提供足够的通道。由表1可见,二维Ni-MOFs的比表面积达到58.4 m2·g−1,明显高于三维Ni-MOFs的比表面积(21.1 m2·g−1)。由此可见,二维Ni-MOFs比三维Ni-MOFs可提供更多的吸附位点,从而有利于F-53B的吸附。
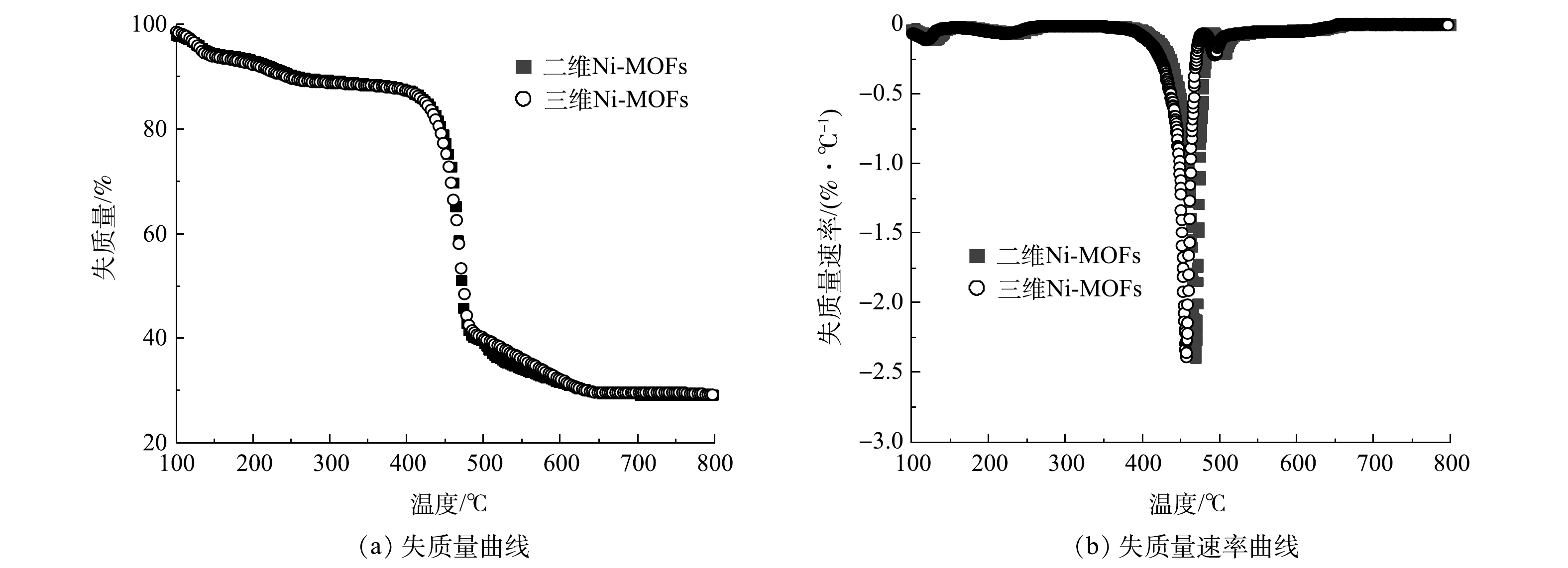
在N2氛围下,通过热重测试考察了Ni-MOFs的热稳定性。如图6所示,样品在40~800 °C内的质量损失主要发生在2个阶段。第1阶段的质量损失发生在100~250 °C,这是材料中的水分子(主要是自由水和结晶水)从孔道中释放的原因[37]。第2阶段的质量损失约开始于400 °C,此时质量损失曲线呈现出急剧下降的趋势,表明材料的金属-有机骨架开始坍塌,材料开始热分解,且该阶段的质量损失超过60%。DTG曲线进一步证实了这一点,在380~490 °C,DTG曲线表现出显著的响应,且二维Ni-MOFs的响应温度(468 °C)略高于三维Ni-MOFs的响应温度(456 °C)。由此可知,2种Ni-MOFs材料的热稳定性整体较好,且二维Ni-MOFs比三维Ni-MOFs材料具有更好的热稳定性。
-
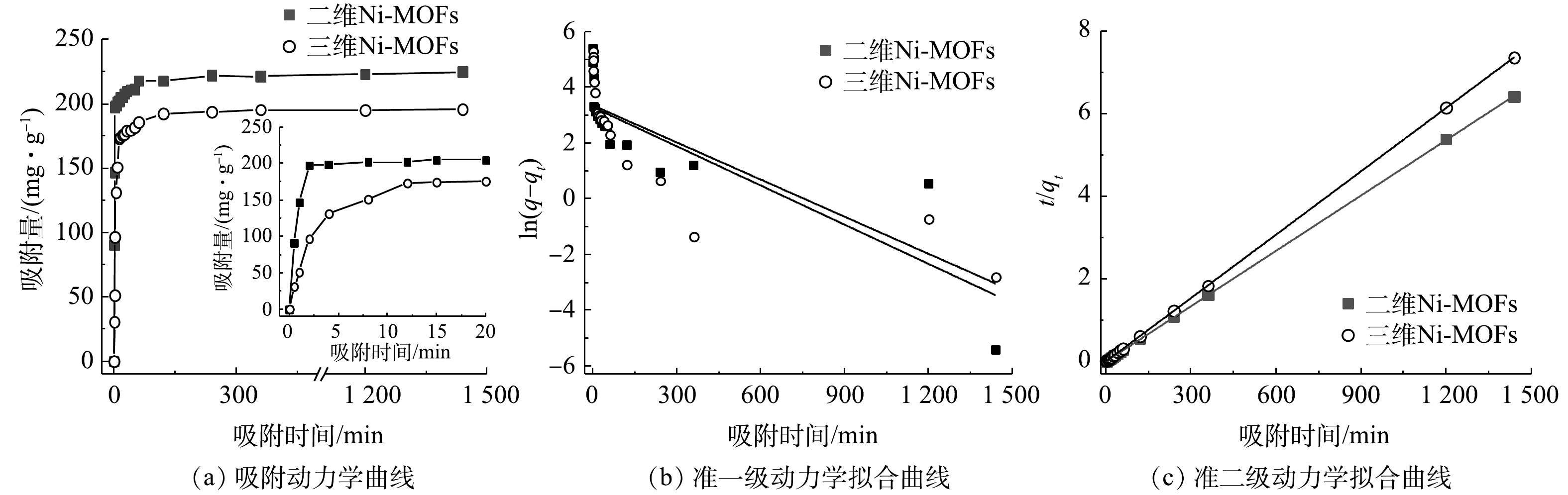
1)吸附动力学拟合与分析。Ni-MOFs吸附F-53B的动力学拟合结果如图7(a)所示。整个吸附过程可大致分为3个阶段:快速吸附阶段、缓慢吸附阶段和平衡阶段。对于三维Ni-MOFs材料,其快速吸附阶段发生在0~4 min,在此阶段,由于MOFs材料表面具有丰富的吸附活性位点,可以提供大量的键合位点,进而促进F-53B的吸附,因此,吸附速率较高;随后在4~12 min内吸附速率较慢,这是因为此时F-53B占据了大量的吸附位点,使吸附剂表面客体分子浓度增大,导致溶液与吸附剂表面的浓度差降低,传质推动力也随之降低,所以吸附量的增加变得缓慢;随着吸附时间的延长(超过 12 min后),吸附基本达到稳定,此时吸附达到平衡,平衡容量为195.6 mg·g−1。相比之下,二维Ni-MOFs在0~2 min内快速吸附F-53B,其吸附速率明显高于三维Ni-MOFs材料。这是因为二维结构比相应的三维结构暴露出更多的活性吸附位点(图4),加上限域作用的贡献,导致F-53B吸附速率明显增强。此外,该材料的吸附速率也快于大部分文献报道的吸附剂(活性炭[14]、离子交换树脂[9]和氧化铝[38]等)吸附速率。而且,二维Ni-MOFs在2 min即可接近吸附平衡,其平衡吸附量为224.6 mg·g−1,高于三维Ni-MOFs的平衡容量(195.6 mg·g−1)。由此可见,通过降维策略将三维MOFs转变为二维MOFs具有明显的优势,实现了对F-53B的高效快速吸附。
对图7(a)的结果采用准一级动力学、准二级动力学拟合,结果如图7(b)、图7(c)以及表2所示。相比于准一级动力学模型,准二级动力学模型具有更好的相关性(R2=0.999 9),且由该模型计算得到的qecal值也更接近实际测量值,这说明准二级动力学模型能够更准确地描述吸附过程,也进一步说明Ni-MOFs对F-53B的吸附过程存在化学吸附。GAO等[9]利用阴离子交换树脂吸附F-53B的结果也表明准二级动力学模型能更好地描述F-53B的吸附过程。
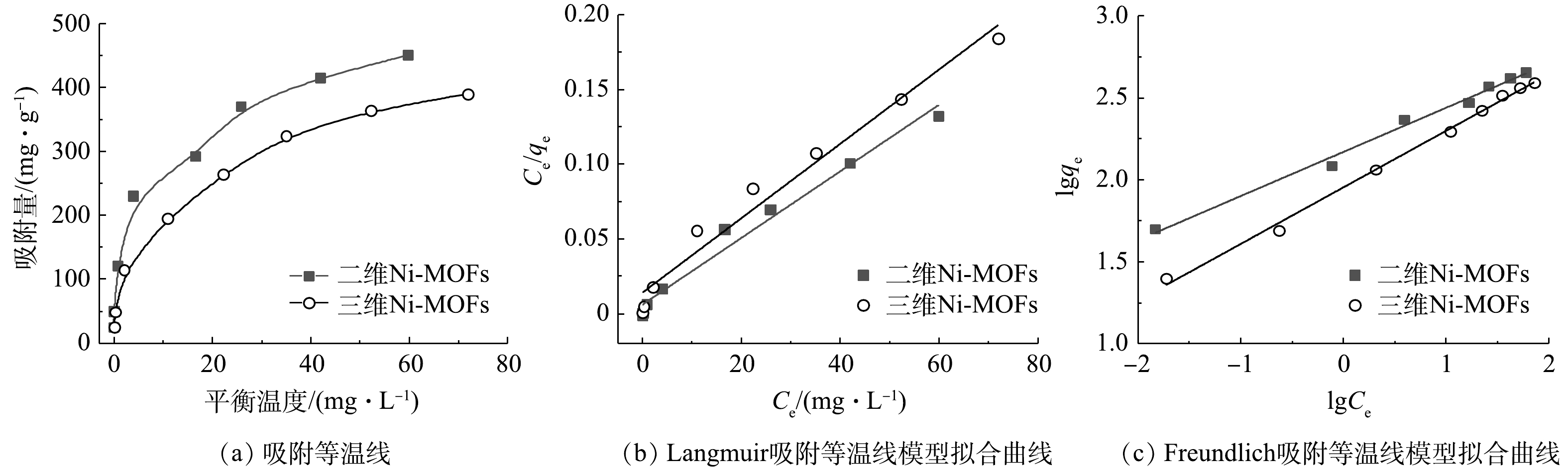
2) 吸附等温线。图8(a)为2种Ni-MOFs材料对F-53B的吸附等温线。2种Ni-MOFs材料对F-53B的吸附量随着平衡质量浓度的升高而升高,并且二维Ni-MOFs材料比三维Ni-MOFs材料对F-53B具有更高的吸附量。在F-53B初始质量浓度为150 mg·L−1时,二维Ni-MOFs材料的吸附容量达到451.2 mg·g−1,明显高于三维Ni-MOFs材料的吸附容量(390.4 mg·g−1)。 Langmuir和Freundlich模型对等温线数据的拟合结果如图8(b)和图8(c)以及表3所示。可见,Freundlich模型(R2超过0.98)能更精确描述F-53B在2种Ni-MOFs材料上的吸附过程,这与GAO等的研究结果一致[9]。以上结果表明F-53B在2种Ni-MOFs材料表面的吸附不是简单的单层吸附,而是存在非均质多层吸附。这可能是因为F-53B中的疏水性官能团C—F产生了疏水性聚集作用。
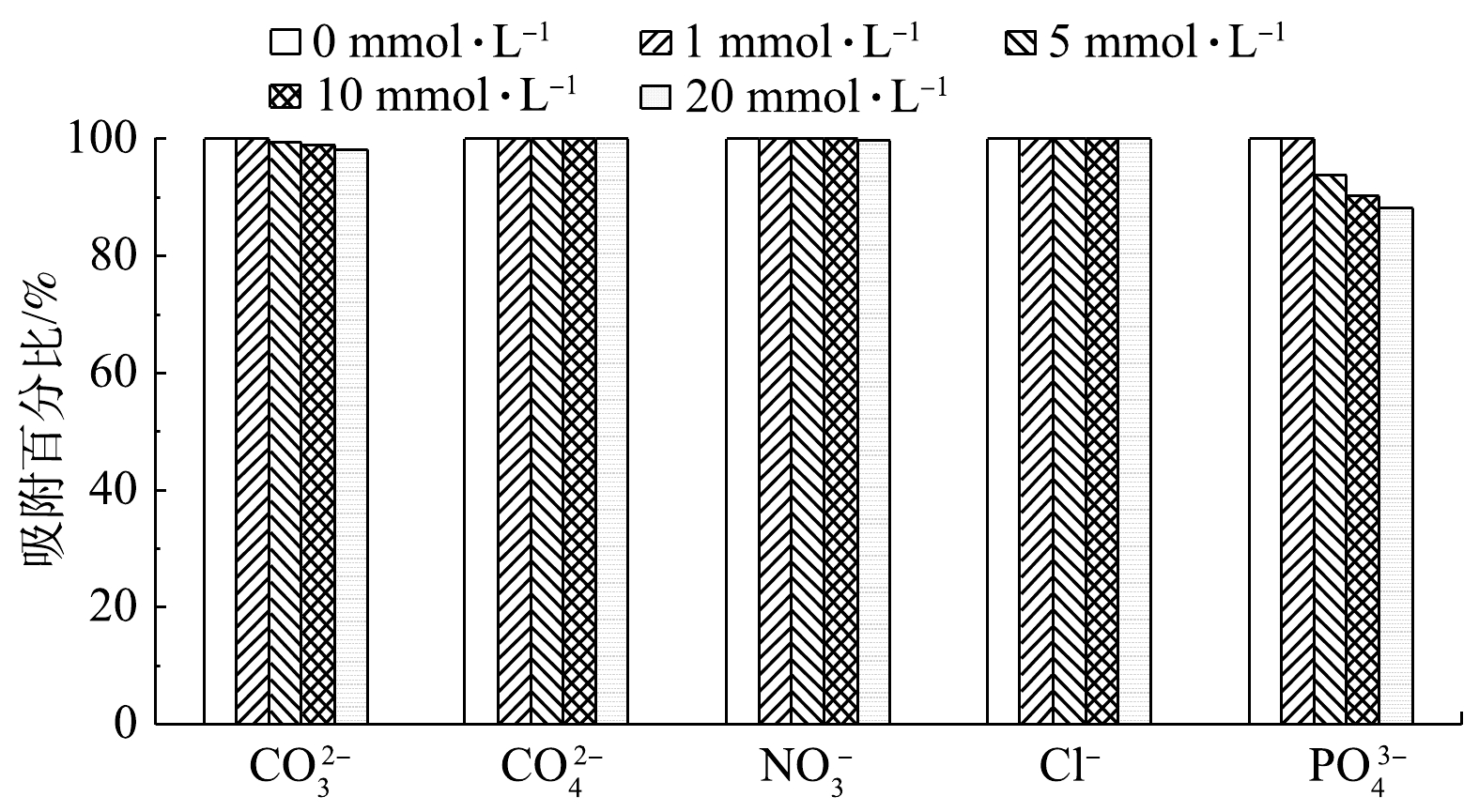
3)共存阴离子的影响。共存阴离子对吸附的影响结果如图9所示。可见,二维Ni-MOFs对F-53B的吸附百分比受共存PO43−的影响较大,二维Ni-MOFs对F-53B的吸附百分比随PO43−质量浓度的升高而降低。然而,二维Ni-MOFs对F-53B的吸附百分比受CO32−、SO42−、NO3−或Cl−的影响很小,几乎可以忽略不计。这可能是因为PO43−带有3个负电荷,其电负性强于带有2个负电荷的CO32−和SO42−以及带有1个负电荷的NO3−和Cl−,因此,PO43−更容易被带正电的二维Ni-MOFs吸附,竞争的F-53B的吸附点位,从而导致F-53B的吸附量略有降低。而CO32−、SO42−、NO3−和Cl−由于电负性较弱,且二维Ni-MOFs可提供较多的吸附位点,从而可降低这4种共存阴离子的影响。以上结果表明二维Ni-MOFs有望在复杂的实际水环境中对F-53B表现出较好的吸附能力。
-
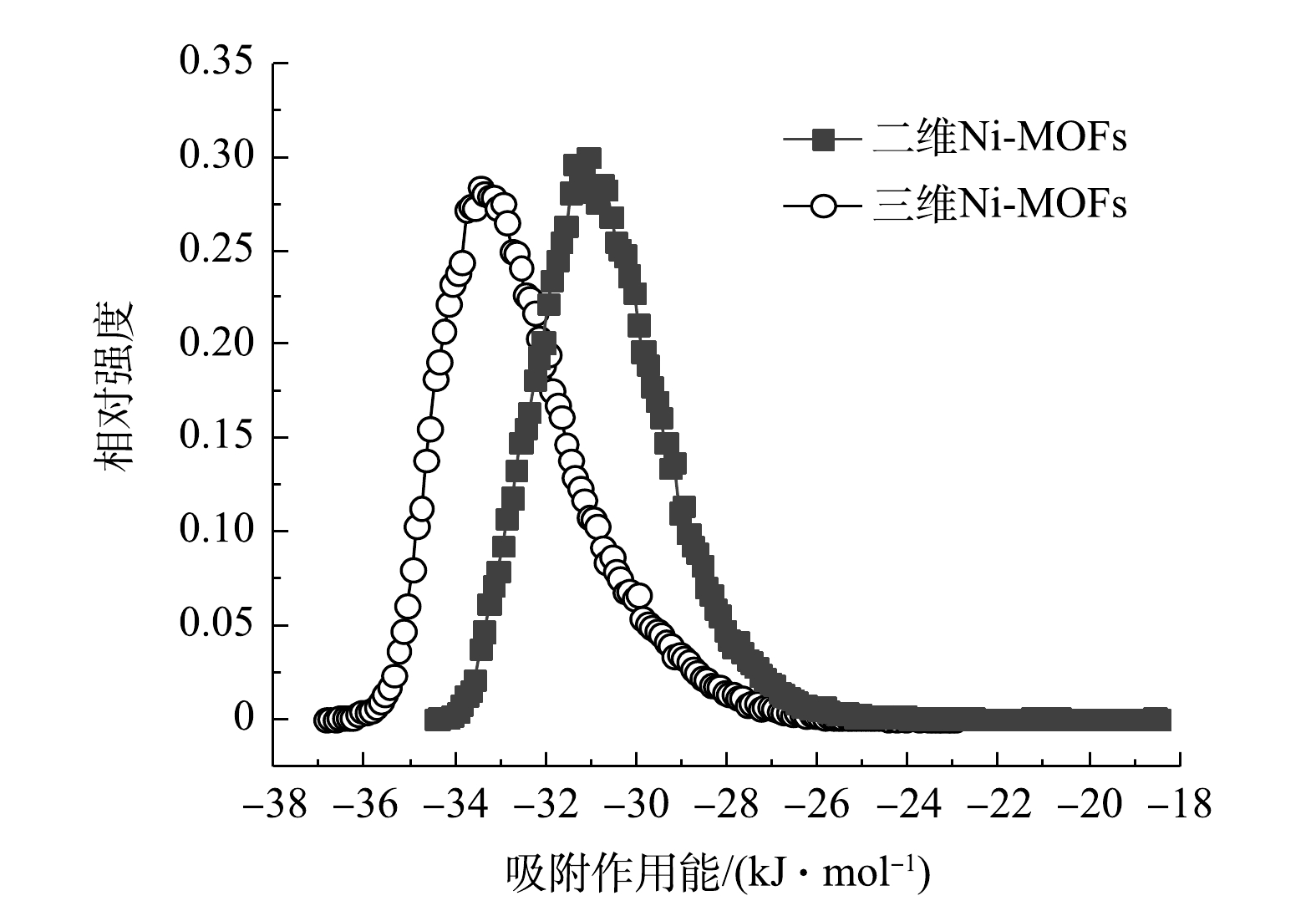
由XRD图谱(图3(a))可知,二维Ni-MOFs吸附F-53B后依然具有明显的特征峰,表明二维Ni-MOFs具有良好的稳定性,吸附F-53B后仍然具有良好的晶型结构;而三维Ni-FOMs吸附F-53B后部分特征峰消失,表明三维Ni-MOFs的稳定性较弱,吸附F-53B后可能出现了结构垮塌。由FTIR图谱(图3(b))可知,2种Ni-MOFs材料吸附F-53B后,原本位于466 cm−1和525 cm−1的Ni—O伸缩振动峰分别偏移到460 cm−1和520 cm−1处。这很可能是由于F-53B与2种Ni-MOFs材料之间的静电作用所致[39],表明静电作用是2种Ni-MOFs材料吸附F-53B的重要机理之一。本文进一步计算了单个F-53B分子与2种Ni-MOFs材料之间的吸附作用能曲线分布。由图10可见,2种Ni-MOFs材料的吸附作用能较小(低于83.68 kJ mol−1),表明这2种材料对F-53B的吸附主要为物理吸附(包括静电作用和范德华力),其次为化学吸附(直接键合形成络合物)。此外,三维Ni-MOFs对F-53B的吸附作用能略高于二维Ni-MOFs,这是由于三维Ni-MOFs的(100)晶面上暴露出大量的Ni原子和与之相连的O原子,使得在开始吸附时三维Ni-MOFs的表面与F-53B上的F和O位点之间的静电相互作用强于二维Ni-MOFs。虽然三维Ni-MOFs表现出对F-53B较强的吸附作用力,但是由于其表面的均一性,并且呈块状的形貌使得三维Ni-MOFs中的活性吸附位点数量有限,加上缺少不同片层之间的限域作用,使得F-53B在三维Ni-MOFs中的自由扩散系数(6.86×10−9 cm2·s−1)明显高于在二维Ni-MOFs中的自由扩散系数(1.18×10−10 cm2·s−1),从而造成F-53B难以被稳固吸附于三维Ni-MOFs表面,这也是二维Ni-MOFs的平衡吸附常数大于三维Ni-MOFs的主要原因。
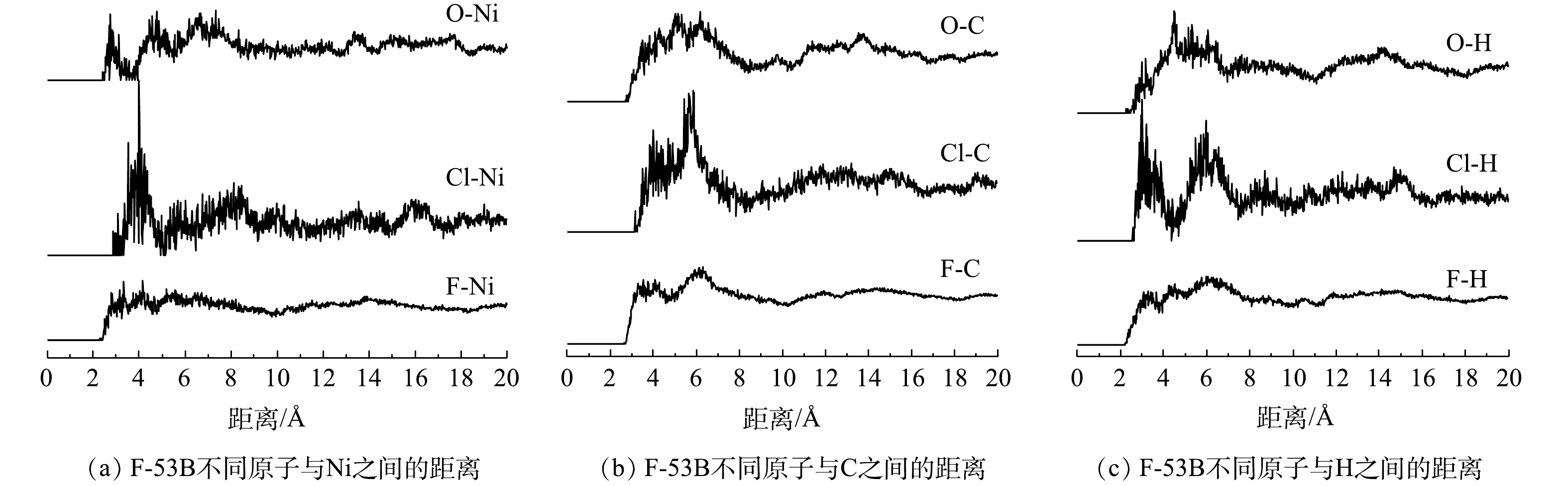
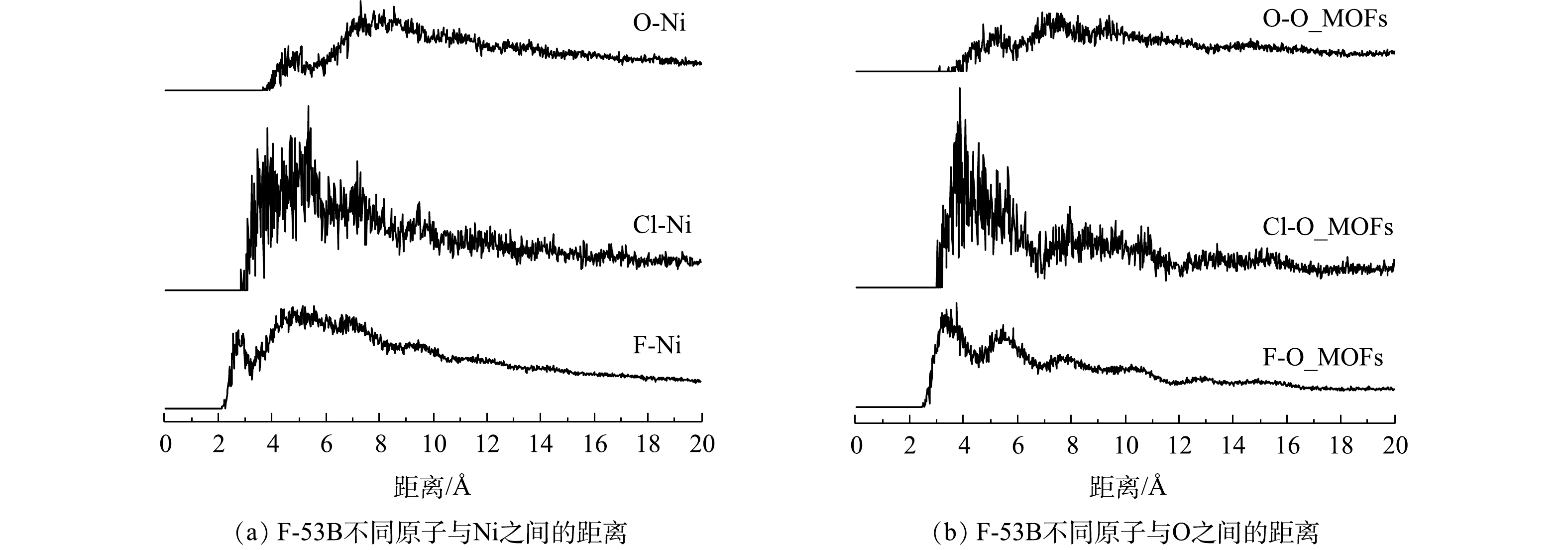
F-53B分子中不同原子(F、Cl、O)与Ni-MOFs骨架中不同吸附位点(Ni、C、H、O_MOFs)之间的RDF曲线如图11和图12所示。对于二维Ni-MOFs而言(图11),F-53B中不同原子与Ni之间的最短距离顺序为O(2.69 Å)<F(2.79 Å)<Cl(3.07 Å),与C的最短距离顺序为O(3.51 Å)<F(3.53 Å)<Cl(3.97 Å),与H的最短距离顺序为O(3.01 Å)<F(3.07 Å)<Cl(3.17 Å)。该结果表明,F-53B与二维Ni-MOFs之间的作用主要为带负电的F、Cl和O与Ni之间的静电作用,其次为F-53B与有机配体中之间的范德华作用,而F和O与骨架之间的作用明显强于Cl,原因为F的电负性强于Cl,而S=O键能够提供较丰富的电子云。由动力学结果可知,吸附过程存在一定的化学吸附,结合以上结果可推测,Ni可能与F-53B的O直接键合形成了络合物,但整体上该化学吸附作用对F-53B吸附的贡献相对较小。对于三维Ni-MOFs而言(图12),F-53B中不同原子与Ni之间的最短距离顺序为F(2.55 Å)<Cl(3.23 Å)<O(4.49 Å),与O之间的最短距离顺序为F (3.39 Å)<Cl (3.87 Å)<O (4.75 Å),证明S=O键上丰富的电子云使得F-53B中的O受到O_MOFs的排斥而远离O_MOFs,同时远离Ni,导致F和Cl与Ni之间的距离明显短于O与Ni之间的距离。总体而言,三维Ni-MOFs吸附F-53B也是以静电作用为主,而吸附过程涉及的化学作用可能是Ni与F直接键合形成络合物,但该作用的相对贡献较小。综上所述,二维和三维Ni-MOFs吸附F-53B的过程是以物理作用为主、化学作用为辅,其中物理作用主要表现为静电作用,其次为范德华力,而化学作用表现为络合反应。
-
1)二维Ni-MOFs和三维Ni-MOFs均具有明显的晶型,其中Ni是以双齿配位模式与有机骨架进行结合,二维Ni-MOFs呈现出分散片层结构,三维Ni-MOFs则呈现出块状结构,二维Ni-MOFs比三维Ni-MOFs具有更高的比表面积、孔隙率和热稳定性。
2) 2种材料对F-53B的吸附均符合准二级动力学方程和Freundlich等温方程,二维Ni-MOFs的吸附速率常数和饱和吸附容量分别达到0.002 4 g·(min·mg)−1和451.2 mg·g−1,分别比三维Ni-MOFs高出20%和16%,吸附过程以非均质多层吸附为主,PO43−的存在会抑制二维Ni-MOFs对F-53B的吸附,而CO32−、SO42−、NO3−或Cl−这些无机阴离子的影响则很小。
3)呈片层结构的二维Ni-MOFs比呈块状结构的三维Ni-MOFs具有更多的活性吸附位点,加上片层结构的限域作用导致二维Ni-MOFs具有更强的F-53B吸附能力,吸附过程主要由静电作用控制。
Ni-MOFs对水中全氟烷基醚磺酸盐的吸附性能及机理
Adsorption performance and mechanisms of Ni-MOFs towards chlorinated polyfluoroalkyl ether sulfonic acid in aqueous phase
-
摘要: 为有效去除水中的全氟烷基醚磺酸盐(F-53B),采用溶剂热法合成二维Ni-MOFs和三维Ni-MOFs材料。采用XRD、FTIR、SEM、N2吸/脱附实验和热重分析对材料的结构形貌和热稳定性进行了表征,采用动力学和热力学实验以及对比分析,考察了Ni-MOFs对水中F-53B的吸附性能,并进一步结合理论计算探究了可能的吸附机理。结果表明,2种材料均具有明显的晶型,其中Ni是以双齿配位模式与有机骨架进行结合,二维Ni-MOFs呈现分散片层结构,三维Ni-MOFs则呈现出块状结构,二维Ni-MOFs比三维Ni-MOFs具有更高的比表面积、孔隙率和热稳定性。2种材料对F-53B的吸附均符合准二级动力学方程和Freundlich等温方程,二维Ni-MOFs的吸附速率常数和吸附容量分别达到0.002 4 g·(min·mg)−1和451.2 mg·g−1,比三维Ni-MOFs分别高出20%和16%,吸附过程以非均质多层吸附为主,受共存阴离子的影响较小。对F-53B的吸附性能与2种材料的构效有关,活性吸附位点和限域作用决定了二维Ni-MOFs比三维Ni-MOFs更优的吸附特性,吸附过程主要受静电作用控制。以上研究结果表明二维Ni-MOFs对F-53B的吸附性能较好,具有良好的应用前景。Abstract: To effectively remove chlorinated polyfluoroalkyl ether sulfonic acid (F-53B) in aqueous phase, two metal-organic framework materials (2D Ni-MOFs and 3D Ni-MOFs) were prepared via solvothermal method. Multiple technologies, including X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), scanning electron microscope (SEM), N2 adsorption/desorption, and thermogravimetry (TG), were used to characterize the structure, morphology, and thermal stability of Ni-MOFs samples. The kinetic and thermodynamic experiments and comparative analysis were used to investigate the performance of Ni-MOFs towards of F-53B in aqueous phase. Moreover, the theoretical calculation was combined to further investigate the adsorption mechanism. Results showed that 2D and 3D Ni-MOFs were crystal materials, of which Ni bonded the organic framework with the bidentate complex form. 2D Ni-MOFs presented a lamellar structure, and 3D Ni-MOFs exhibited a massive structure. 2D Ni-MOFs had a higher specific surface area, porosity and thermal stability than those of 3D Ni-MOFs. The adsorption kinetics and isotherms of F-53B were in line with the pseudo-second-order model and Freundlich model, respectively. The adsorption rate constant and adsorption capacity of 2D Ni-MOFs reached 0.002 4 g·(min·mg)−1 and 451.2 mg·g−1, respectively, which were 20% and 16% higher than those of 3D Ni-MOFs. Moreover, the adsorption process was dominated by the heterogeneous multilayer adsorption, and the co-existing anions had negligible impacts on the adsorption of F-53B on 2D Ni-MOFs. The adsorption performance of Ni-MOFs to F-53B was dependent on their structure-effect. The abundant active adsorption sites and confinement effect determined the better adsorption performance of 2D Ni-MOFs than 3D Ni-MOFs, and the adsorption process was mainly controlled by electrostatic interaction. The results of this study showed that 2D Ni-MOFs is an excellent adsorbent for F-53B and has a good application prospect.
-

-
表 1 二维和三维Ni-MOFs的比表面积、孔容和孔径
Table 1. Specific surface area, pore volume and pore size of 2D Ni-MOFs and 3D Ni-MOFs
样品 BET比表面积/(m2·g−1) 总孔容/(cm3·g−1) 平均孔径/nm 二维Ni-MOFs 58.38 0.336 23.01 三维Ni-MOFs 21.12 0.037 6.98 表 2 2种Ni-MOFs材料吸附F-53B的动力学参数
Table 2. Kinetic parameters for F-53B adsorption on two Ni-MOFs samples
Ni-MOFs q实/(mg·g−1) 准一级动力学 准二级动力学 qecal/(mg·g−1) k1/min−1 R2 qecal/(mg·g−1) k2/(g·(min·mg)−1) R2 二维Ni-MOFs 224.6 26.7 0.004 7 0.714 5 224.2 0.002 4 0.999 9 三维Ni-MOFs 195.6 28.5 0.004 4 0.647 5 195.7 0.002 0 0.999 9 表 3 2种Ni-MOFs材料吸附F-53B的等温线拟合参数
Table 3. Adsorption isotherm parameters for F-53B by two Ni-MOFs samples
Ni-MOFs Langmuir模型 Freundlich模型 qm/(mg·g−1) KL/(L·mg−1) R2 n-1 KF/(L·mg−1) R2 二维Ni-MOFs 448.4 0.35 0.976 6 0.269 148.1 0.989 6 三维Ni-MOFs 401.6 0.17 0.969 7 0.345 90.0 0.995 6 -
[1] PILAT M J, PEGNAM R C. Particle emissions from chrome plating[J]. Aerosol Science and Technology, 2006, 40(8): 639-648. doi: 10.1080/02786820600763020 [2] 陆宏, 周锦阳, 杨帆, 等. 基于Meta分析的全氟化合物对鱼类生态毒性效应[J]. 环境科学,DOI:10.13227/j.hjkx.202209239. [3] FUJII S, POLPRASERT C, TANAKA S, et al. New POPs in the water environment: distribution, bioaccumulation and treatment of perfluorinated compounds: A review paper[J]. Journal of Water Supply:Research and Technology-Aqua, 2007, 56(5): 313-326. doi: 10.2166/aqua.2007.005 [4] MUNOZ G, LIU J, VO DUY S, et al. Analysis of F-53B, Gen-X, ADONA, and emerging fluoroalkylether substances in environmental and biomonitoring samples: A review[J]. Trends in Environmental Analytical Chemistry, 2019, 23: e00066. doi: 10.1016/j.teac.2019.e00066 [5] WANG S, HUANG J, YANG Y, et al. First report of a Chinese PFOS alternative overlooked for 30 years: Its toxicity, persistence and presence in the environment[J]. Environmental Science & Technology, 2013, 47(18): 10163-10170. [6] LI C H, REN X M, RUAN T, et al. Chlorinated polyfluorinated ether sulfonates exhibit higher activity toward peroxisome proliferator-activated receptors signaling pathways than perfluorooctanesulfonate[J]. Environmental Science & Technology, 2018, 52(5): 3232-9. [7] XIN Y, REN X M, RUAN T, et al. Chlorinated polyfluoroalkylether sulfonates exhibit similar binding potency and activity to thyroid hormone transport proteins and nuclear receptors as perfluorooctanesulfonate[J]. Environmental Science & Technology, 2018, 52(16): 9412-9418. [8] 赵楠, 孔媛, 张莹莹, 等. 基于1H NMR的代谢组学方法研究F-53B暴露对大鼠血清代谢表型的影响[J]. 环境化学, 2023, 42(1): 11-19. [9] GAO Y, DENG S, DU Z, et al. Adsorptive removal of emerging polyfluoroalky substances F-53B and PFOS by anion-exchange resin: A comparative study[J]. Journal of Hazardous Materials, 2017, 323: 550-557. doi: 10.1016/j.jhazmat.2016.04.069 [10] ZHUO Q, WANG J, NIU J, et al. Electrochemical oxidation of perfluorooctane sulfonate (PFOS) substitute by modified boron doped diamond (BDD) anodes[J]. Chemical Engineering Journal, 2020, 379: 122280. doi: 10.1016/j.cej.2019.122280 [11] CAO H, ZHANG W, WANG C, et al. Photodegradation of F-53B in aqueous solutions through an UV/Iodide system[J]. Chemosphere, 2022, 292: 133436. doi: 10.1016/j.chemosphere.2021.133436 [12] ZHANG K, CAO Z, HUANG J, et al. Mechanochemical destruction of Chinese PFOS alternative F-53B[J]. Chemical Engineering Journal, 2016, 286: 387-393. doi: 10.1016/j.cej.2015.10.103 [13] ATEIA M, ARIFUZZAMAN M, PELLIZZERI S, et al. Cationic polymer for selective removal of GenX and short-chain PFAS from surface waters and wastewaters at ng/L levels[J]. Water Research, 2019, 163: 114874. doi: 10.1016/j.watres.2019.114874 [14] DU Z, DENG S, LIU D, et al. Efficient adsorption of PFOS and F53B from chrome plating wastewater and their subsequent degradation in the regeneration process[J]. Chemical Engineering Journal, 2016, 290: 405-413. doi: 10.1016/j.cej.2016.01.077 [15] 兰馨, 高生军, 樊佳铜, 等. 氨修饰NiMg-MOF-74材料共吸附硫硝碳效能及其机理[J]. 环境工程学报, 2023, 17(1): 142-155. [16] HE Y, WANG Z, WANG H, et al. Metal-organic framework-derived nanomaterials in environment related fields: Fundamentals, properties and applications[J]. Coordination Chemistry Reviews, 2021, 429: 213618. doi: 10.1016/j.ccr.2020.213618 [17] DUAN C, YU Y, XIAO J, et al. Recent advancements in metal-organic frameworks for green applications[J]. Green Energy & Environment, 2021, 6(1): 33-49. [18] FURUKAWA H, MüLLER U, YAGHI O M. “Heterogeneity within Order” in Metal-Organic Frameworks[J]. Angewandte Chemie International Edition, 2015, 54(11): 3417-3430. doi: 10.1002/anie.201410252 [19] 岳琳, 张迎, 张文丽, 等. Sn-MOF对染料废水中酸性大红3R的吸附特性[J]. 环境工程学报, 2019, 13(11): 2553-2561. doi: 10.12030/j.cjee.201812100 [20] 万红友, 阎靖炜, 郭丛, 等. Cu/Fe-MOF复合材料在水处理过程应用研究进展[J]. 水处理技术, 2022, 48(11): 1-7. [21] XU G-R, AN Z-H, XU K, et al. Metal organic framework (MOF)-based micro/nanoscaled materials for heavy metal ions removal: The cutting-edge study on designs, synthesis and applications[J]. Coordination Chemistry Reviews, 2021, 427: 213554. doi: 10.1016/j.ccr.2020.213554 [22] KALAJ M, BENTZ K C, AYALA S, JR. , et al. MOF-polymer hybrid materials: From simple composites to tailored architectures[J]. Chemical Reviews, 2020, 120(16): 8267-8302. doi: 10.1021/acs.chemrev.9b00575 [23] WANG B, LV X-L, FENG D, et al. Highly stable Zr(IV)-based metal–organic frameworks for the detection and removal of antibiotics and organic explosives in water[J]. Journal of the American Chemical Society, 2016, 138(19): 6204-6216. doi: 10.1021/jacs.6b01663 [24] LI Y, YANG Z, WANG Y, et al. A mesoporous cationic thorium-organic framework that rapidly traps anionic persistent organic pollutants[J]. Nature Communication, 2017, 8(1): 1354. doi: 10.1038/s41467-017-01208-w [25] BARPAGA D, ZHENG J, HAN K S, et al. Probing the sorption of perfluorooctanesulfonate using mesoporous metal–organic frameworks from aqueous solutions[J]. Inorganic Chemistry, 2019, 58(13): 8339-8346. doi: 10.1021/acs.inorgchem.9b00380 [26] LI X, WANG B, CAO Y, et al. Water contaminant elimination based on metal-organic frameworks and perspective on their industrial applications[J]. ACS Sustainable Chemistry & Engineering, 2019, 7(5): 4548-4563. [27] CHANG P-H, CHEN C-Y, MUKHOPADHYAY R. et al. Novel MOF-808 metal–organic framework as highly efficient adsorbent of perfluorooctane sulfonate in water[J]. Journal of Colloid and Interface Science, 2022, 623: 627-636. doi: 10.1016/j.jcis.2022.05.050 [28] VENTURA K, ARRIETA R, MARCOS-HERNANDEZ M. et al. Superparamagnetic MOF@GO Ni and Co based hybrid nanocomposites as efficient water pollutant adsorbents[J]. Science of the Total Environment, 2020, 738: 139213. doi: 10.1016/j.scitotenv.2020.139213 [29] CHAKRABORTY G, PARK I-H, MEDISHETTY R, et al. Two-dimensional metal-organic framework materials: Synthesis, structures, properties and applications[J]. Chemical Reviews, 2021, 121(7): 3751-3891. doi: 10.1021/acs.chemrev.0c01049 [30] ZHAO M, HUANG Y, PENG Y, et al. Two-dimensional metal-organic framework nanosheets: Synthesis and applications[J]. Chemical Society Reviews, 2018, 47(16): 6267-6295. doi: 10.1039/C8CS00268A [31] XUE F, KUMAR P, XU, W, et al. , Direct synthesis of 7 nm-thick zinc(ii)-benzimidazole-acetate metal-organic framework nanosheets[J]. Chemistry of Materials, 2018, 30(1): 69-73. doi: 10.1021/acs.chemmater.7b04083 [32] TI B, LI L, LIU J. , et al. Global distribution potential and regional environmental risk of F-53B[J]. Science of the Total Environment, 2018, 640-641, 1365-1371. [33] 柳泽伟. 金属-有机骨架材料吸附分离性能的功能化改性计算化学研究及高通量筛选[D]. 广州: 华南理工大学, 2019. [34] ALLEN F H, BELLARD S, BRICE M, et al. The Cambridge Crystallographic Data Centre: computer-based search, retrieval, analysis and display of information[J]. Acta Crystallographica Section B:Structural Crystallography and Crystal Chemistry, 1979, 35(10): 2331-2339. doi: 10.1107/S0567740879009249 [35] RAPPé A K, CASEWIT C J, COLWELL K, et al. UFF, a full periodic table force field for molecular mechanics and molecular dynamics simulations[J]. Journal of American Chemical Society, 1992, 114(25): 10024-10035. doi: 10.1021/ja00051a040 [36] LI X, HOU M, QU X, et al. Electric‐field assisted hydrolysis-oxidation of MOFs: Hierarchical ternary (oxy) hydroxide micro-flowers for efficient electrocatalytic oxygen evolution[J]. Small, 2022, 18(6): 2104863. doi: 10.1002/smll.202104863 [37] 惠远峰. 新型金属有机骨架材料的合成及吸附染料废水的性能研究[J]. 功能材料, 2018, 49(7): 7188-7191. [38] ZHAO S, LIU S, WANG F, et al. Sorption behavior of 6: 2 chlorinated polyfluorinated ether sulfonate (F-53B) on four kinds of nano-materials[J]. Science of the Total Environment, 2021, 757: 144064. doi: 10.1016/j.scitotenv.2020.144064 [39] QIAN J, SHEN M, WANG P, et al. Perfluorooctane sulfonate adsorption on powder activated carbon: Effect of phosphate (P) competition, pH, and temperature[J]. Chemosphere, 2017, 182: 215-222. doi: 10.1016/j.chemosphere.2017.05.033 -



 下载:
下载: