-
二氧化钛(TiO2)是最早被证明具有光催化效果的材料[1],由于其具有成本低、化学性质稳定、氧化还原能力强、抗腐蚀性能好、无毒环境友好和重复可用性高等特点[2],这使其成为降解有机污染物的首选光催化材料,但同时也存在光响应范围窄和电子-空穴对复合率高的缺点[3 − 4],导致对有机污染物的降解能力减弱,阻碍其实际应用[5]. 氧化锌(ZnO)是宽禁带的n型半导体材料[6],具有良好的光学性能和丰富的表面活性位置[7],并且由于氧化锌和二氧化钛具有相似的带隙能量和阶梯式带隙能级,因此ZnO常被用来改性TiO2[8 − 9],制备出性能更为优异的复合材料.
许多研究者对ZnO和TiO2进行复合掺杂改性,利用ZnO的四面体结构、极性面平整稳定和无再构现象的表面形貌优势,使两者产生界面耦合效应,吸收光谱红移,拓宽了光催化剂的光谱响应范围,提高对光照的利用率[10]. TiO2-ZnO复合光催化剂的能级差也有利于电子-空穴对的分离,从而减少了光生载流子(电子-空穴)的复合,极大地提升了光催化反应的效率[11],同时ZnO的掺杂不会改变晶格图案,可以很好地保持原有作用[12].
本文介绍了TiO2-ZnO光催化降解水中有机污染物的机理,系统综述了TiO2-ZnO复合材料的制备方法(溶胶-凝胶法、水热法、溶剂热法、沉淀法等)和进一步改性(掺杂改性、构造异质结和锚定优化)的研究,以期推动发展经济、可靠、性能优异的光催化技术.
-
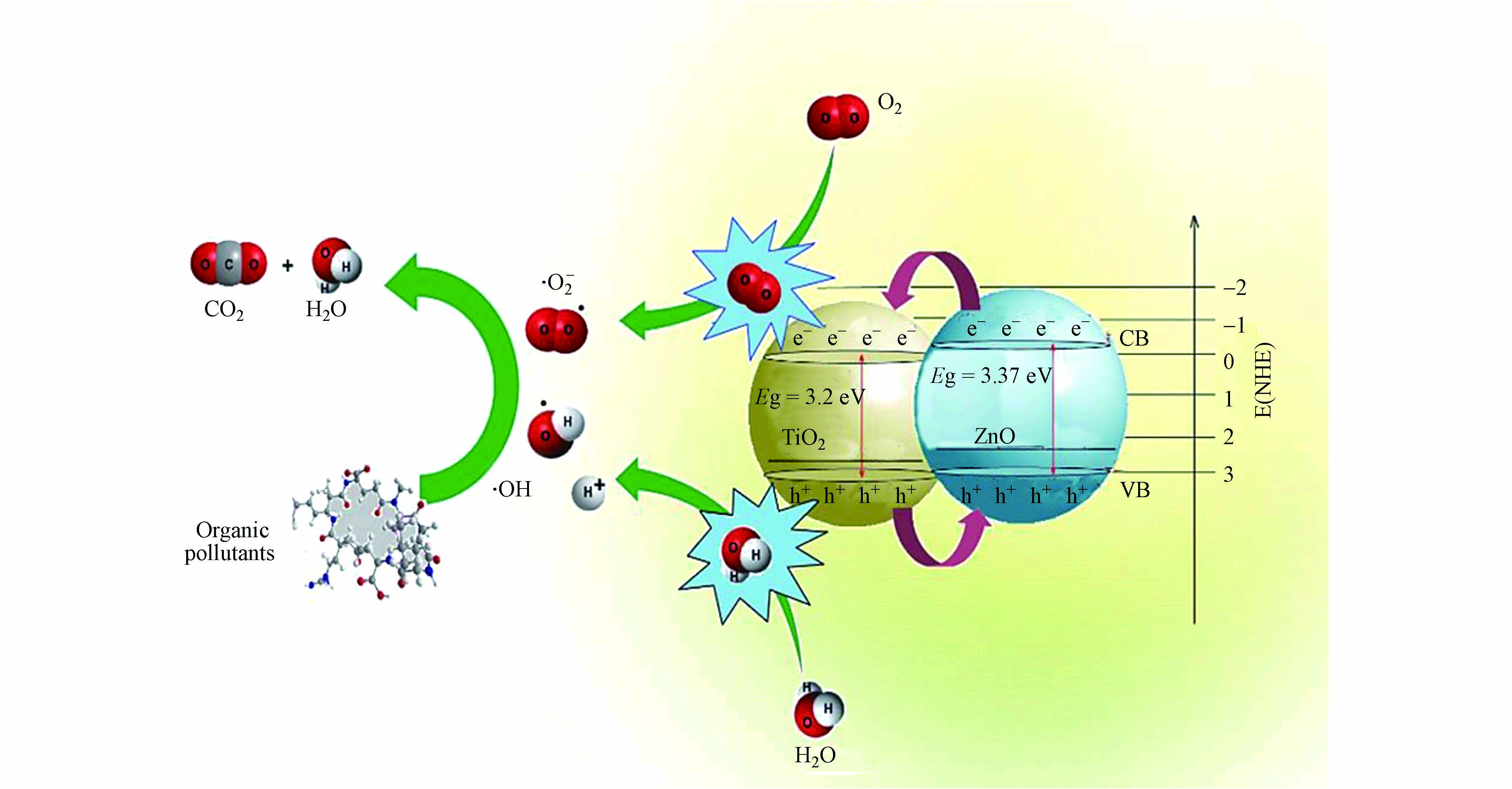
半导体光催化剂材料的能带结构通常是由一个充满电子的低能价带(VB)和一个空的高能导带(CB)组成,价带顶和导带底之间的能量差叫做禁带宽度(Eg)[13 − 14],二氧化钛的禁带宽度是3.2 eV(锐钛矿相)[15],氧化锌的禁带宽度是3.37 eV[16],光催化剂吸收光电子的能量后进入激发态[5],遵循能带理论,光催化反应机理如图1所示.
当光催化剂在受到大于或等于自身带隙能量的光子能量照射时,会激发价带上的电子跃迁到导带[18],在价带上留下空穴(h+),导带上出现等量的电子(e−),形成的电子-空穴对(式1),具有强氧化还原能力[19],由于ZnO的导带位置比TiO2高,部分电子(e−)从ZnO的导带转移到TiO2的导带,空穴(h+)从TiO2的价带转移到ZnO价带[20 − 21].
产生的电子和空穴迁移到光催化剂表面,可能会发生电子-空穴对复合,产生的能量以热能或辐射光的形式消散(式2)[22].
空穴具有氧化性,可与催化剂表面的H2O和OH−发生反应,生成氧化能力更强的羟基自由基(·OH)(式3、4),也可直接与催化剂表面吸附的有机物污染物发生氧化反应(式10),将污染物分解为CO2和H2O[23 − 24];电子具有还原性,可与吸附在催化剂表面的O2发生反应,生成具有强氧化能力的超氧根离子(·O2−)(式5)[25].
·O2−与溶液中的H2O反应生成具有强氧化能力的H2O2(式6、7),H2O2直接发生光解或者与光生电子反应生成·OH(式8、9)[26 − 27].
游离或者吸附在催化剂表面的有机污染物与·OH和·O2−发生氧化还原反应,将污染物矿化分解成CO2和H2O(式11、12)[28 − 29].
复合光催化剂光催化反应过程如下:
-
金属纳米复合材料制备方法有许多种,主要可分为固相法、气相法、液相法. 固相法是指将固态的原料经过研磨或煅烧等方式获得纳米颗粒物的方法[30],分为固相热分解法、高温固相反应法和室温固相反应法[31]. 固相法工艺简单、成本低廉,制得的产品粒度分布不均匀,形貌不规则,能源消耗大[32]. 气相法是指在真空条件下,将原料转变成气体,随后使其在气体状态下发生物理化学变化,再经过冷却凝聚得到纳米粒子的方法[30],该方法具有操作复杂,工艺成本高的缺点,难以推广应用[33]. 液相法是指一种或几种金属盐溶液经过一系列物理化学变化,最后析出纳米颗粒的方法[34],可分为溶胶-凝胶法、沉淀法、水/溶剂热法等. 液相法具有操作简便、纯度高、均匀性好、粒径可控等诸多优点[35],因此应用范围广. 本文介绍了几种制备TiO2-ZnO复合纳米材料的常见方法.
-
溶胶-凝胶法(Sol-Gel)可利用含钛或含锌的化合物经过水解和缩合反应形成分布均匀的溶胶,再通过陈化、聚合形成凝胶,凝胶经过干燥、煅烧等工艺得到目标纳米材料[36].
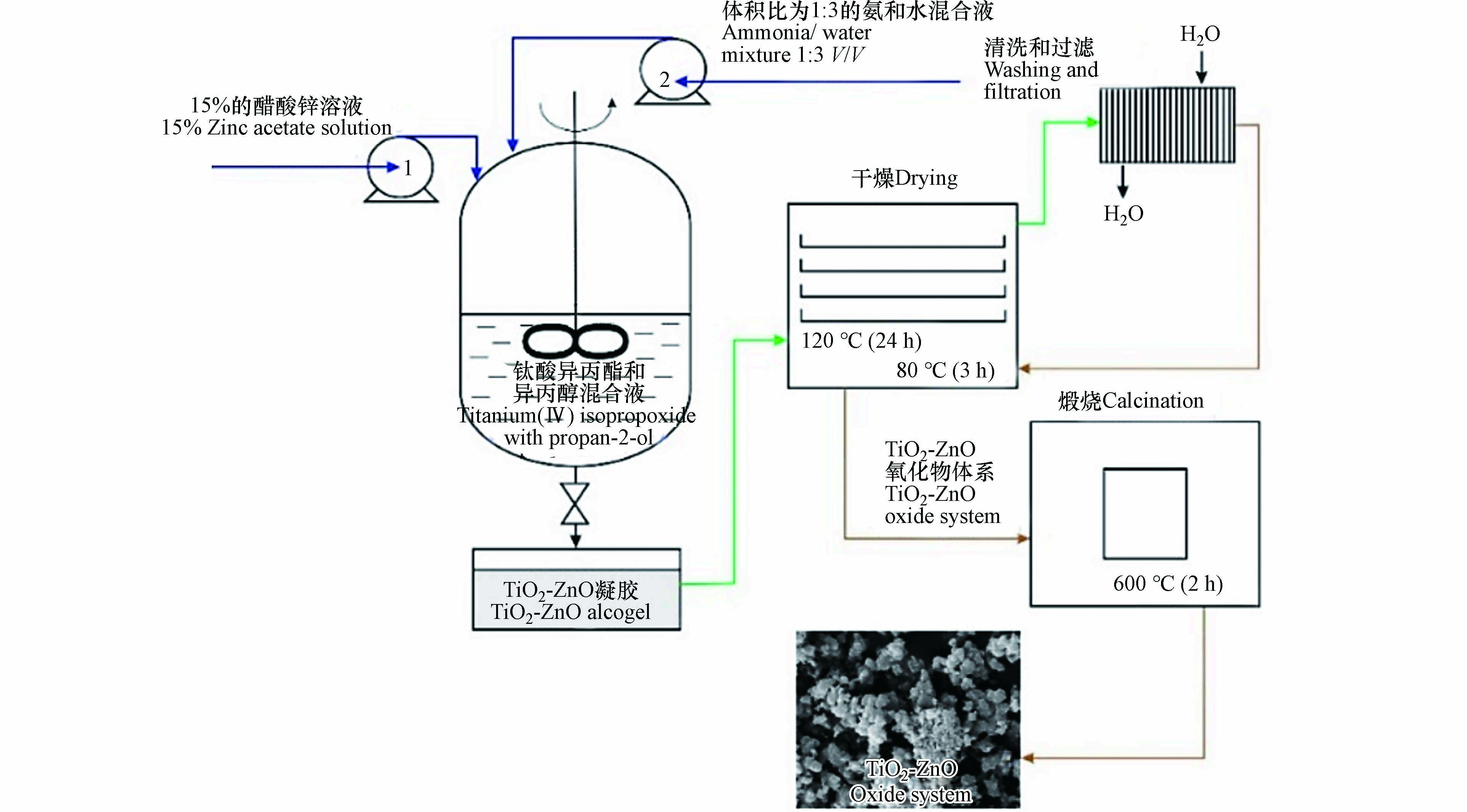
在溶胶-凝胶法中一般使用钛酸脂类(如钛酸异丙酯、钛酸四丁酯等)为钛源溶解在醇类溶液中,乙酸锌作为锌源溶解在去离子水中,表1总结了该方法制备TiO2-ZnO复合材料使用的原料. 如图2所示,Siwińska-Stefańska等[37]制得一系列物质的量比不同的ZnO/TiO2颗粒,制备出的复合材料均表现出高度团聚现象,最小粒径为255—615 nm(Ti/Zn=1/3),样品颗粒的比表面积随着TiO2摩尔数的增加而增加,而煅烧温度过高会降低样品的比表面积,当物质的量比为9:1且在600 ℃煅烧的情况下,ZnO/TiO2催化活性最高. Upadhyay等[38]制备出不同体积比ZnO/TiO2纳米复合粉末,制备出的ZnO和TiO2混合样品具有较高的结晶度,随着ZnO体积的增加,样品平均尺寸从18 nm增加42 nm.
溶胶-凝胶法具有工艺简单、均匀性高、纯度高和对实验室设备要求低等优点[39],但存在原料昂贵、目标产物团聚、制备时间长的缺点. 然而,已有研究表明通过热处理和加酸性物质可以改善团聚现象[40]. 溶胶-凝胶法是制备TiO2-ZnO复合材料最常用的方法,该方法制得的复合材料中TiO2一般是锐钛矿型,ZnO是六方纤锌矿型,煅烧温度、ZnO的含量等对材料性能的影响较大,未来可更加深入地研究操作条件对于光催化效果的影响.
-
水热法(hydrothermal method)指原料以水溶液为反应介质在高温高压的条件下(通常在高压釜中进行),使得在一般条件下难溶或者不溶的物质重新溶解反应并结晶,最终获得目标物质的方法[44].
图 3为水热法制备TiO2-ZnO复合材料的一般步骤:先制备ZnO或者TiO2中的一种物质,再在高压釜中将制备好的物质加入另一物质的前驱体液中,最后经过洗涤、干燥、煅烧等工艺得到目标产物. 表2总结了水热法制备TiO2-ZnO复合材料的研究. 通过向前驱体液中添加聚乙烯吡咯烷酮(PVP)、聚二烯丙基二甲基氯化铵(PDDA)等物质可以形成海胆状的ZnO/TiO2纳米复合材料,该形貌的复合材料有更多的活性点位和更大的比表面积,对有机污染物的降解效率也更高. Cheng等[45]测得海胆状ZnO/TiO2复合材料的比表面积为187.3 m2·g−1. Zhou等[46]使用海胆状的ZnO/TiO2对罗丹明B(RhB)进行了光降解实验,降解率高达95.5%,催化效果良好,并且表明pH值和钛源浓度对复合材料的形貌也存在影响.
该方法具有反应条件温和、工艺设备简单、前驱物费用低、目标物质产率高、纯度高的优点,但缺点是反应能耗高,获得的纳米材料易团聚[47]. 有研究表明通过改变水热条件(温度、时间、压力等)、掺杂剂和前驱物种类可以控制合成材料的形态、掺杂水平和表面缺陷[48]. 水热法在制备TiO2-ZnO复合材料的过程中,可通过添加表面活性物质(PVP、十二烷基硫酸钠SDS、十六烷基三甲基溴化铵CTAB等)获得存在更多活性点位形貌的材料,未来可以系统研究表面活性物质对于材料形貌的影响.
-
溶剂热法(solvothermal method)与水热法类似,只不过是把反应介质由水变换成有机溶剂(如甲醇、乙醇胺、甲苯等). 因为有机溶剂的加入可以改变反应温度和压力,使得反应比水热法更容易进行.
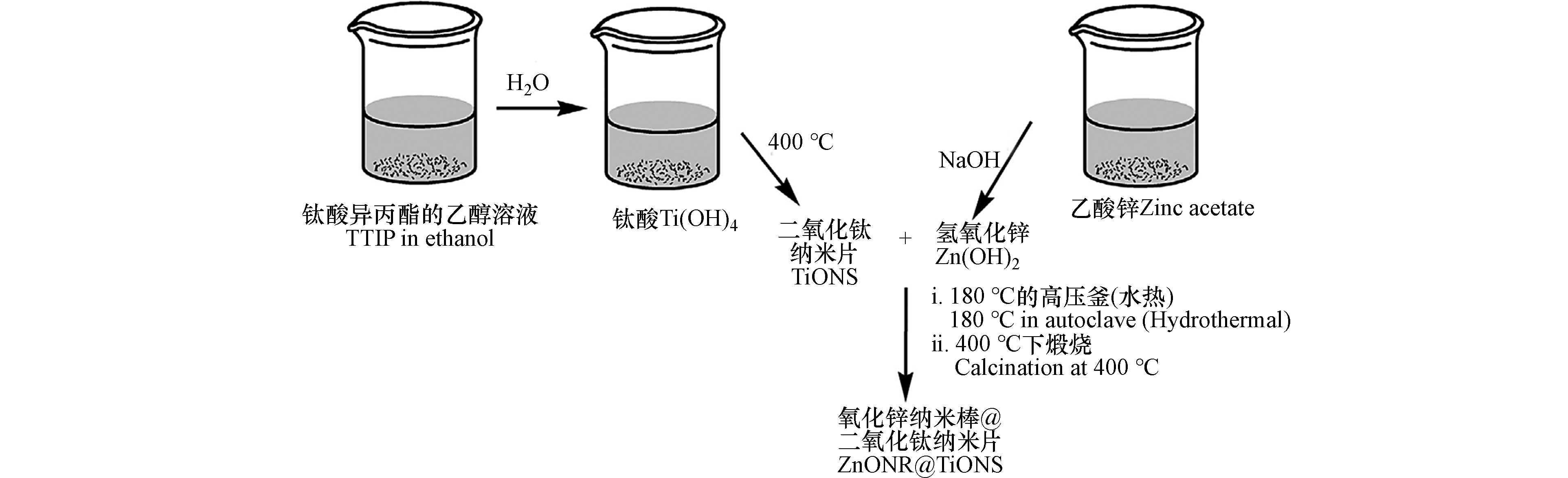
Zha等[51]以TiF4和Zn(Ac)2·2H2O分别作为钛源和锌源,无水乙醇为溶剂,制备出刺猬状和叶片状的TiO2/ZnO复合材料(如图4). 这两种形态的复合材料中TiO2为锐钛矿型,ZnO为六方纤锌矿型,刺猬状的复合材料对甲基橙染料(MO)的降解效果好. 研究者发现F−和柠檬酸钠分别对刺猬状、叶片状的复合材料形成有控制作用. Huo等[52]以钛酸四丁酯、乙酰丙酮锌为前驱体,异丙醇作为溶剂制备出TiO2/ZnO微球形态介孔的复合材料,探究了Ti/Zn比、反应时间对材料形态、催化效果的影响. Zhang等[53]采用溶剂热法制备了ZnO/TiO2双层薄膜,该薄膜在可见光范围内具有较高的光吸收强度,且电子-空穴对复合率降低.
溶剂热法具有工艺简单、结晶度高、纯度高、颗粒生长均匀的优点,但反应体系的压力相较于水热法更高,对实验室的操作条件更加苛刻[54]. 该方法与水热法类似,通过改变溶液的pH、温度、压力或者添加离子(如F−、Cl−、NH4+等)可以有效控制制备材料的尺寸和形貌,提高光催化能力.
-
沉淀法包括直接沉淀法、均匀沉淀法和共沉淀3种方法. 直接沉淀法是指溶液中Ti或者Zn离子立即与沉淀剂反应生成沉淀物,并对沉淀物进行一系列处理后得的纳米材料[55]. 均匀沉淀法是指沉淀剂缓慢加入含有Ti或者Zn离子的溶液中,缓慢而均匀地生成沉淀物[56 − 57]. 共沉淀是指沉淀剂加入含有Ti和Zn离子的溶液中,共同生成沉淀物[58].
Lachom等[59]以NH3OH为沉淀剂采用共沉淀法制备了TiO2/ZnO复合纳米颗粒,其中TiO2和ZnO分别是锐钛矿型和六方纤锌矿型,在紫外光下照射140 min后,该复合材料对甲基橙染料(MO)的降解率可达98.11%. Haghighatzadeh等[60]以NaOH为沉淀剂采用共沉淀法将ZnO包裹在TiO2中,制备出ZnO/TiO2核壳结构的复合材料,其中TiO2和ZnO分别是锐钛矿型和六方纤锌矿型,在紫外光照射下亚甲基蓝(MB)的最高降解率为59.56%,主要是由空穴转移到TiO2壳区价带引起的. Brooms等[61]先以ZnCl2溶液为锌源,NaOH为沉淀剂,制得白色ZnO粉末;后通过机械混合法将购买到的TiO2与制备的ZnO混合,最终获得TiO2/ZnO复合材料,该材料对对甲苯酚的降解率约为60%,但经过聚苯胺(PANI)改性的TiO2/ZnO复合材料(0.5:1:1)可使对甲苯酚的降解率达到99.3%. Arabnezhad等[62]以乙酸锌、四氯化钛作为原料,利用共沉淀法制备得到TiO2/ZnO复合材料,探究了不同Ti/Zn比对材料结构和催化效果的影响.
沉淀法具有原料来源广泛、成本低、反应温度低、生成物纯度高、易控制纳米颗粒尺寸的优点,但存在水洗困难和杂质难去除的缺点[63 − 64]. NaOH是该方法常用的沉淀剂,通过控制反应温度、pH、搅拌速度等因素,可以改善材料的颗粒尺寸、均匀性.
-
目前,除上述方法外,Kumari等[65]使用原子层沉积法(ALD)在导电多孔不锈钢衬底上制备了TiO2-ZnO复合材料. Zhou等[66]先通过水热法制得3D海胆状的ZnO纳米颗粒,后采用电化学沉积方法在聚二烯丙基二甲基氯化铵(PDDA)溶液中沉积纳米TiO2,最后成功获得海胆状ZnO/TiO2复合材料. Khan等[67]将TiO2粉末溶解于乙醇和二甘醇中获得二氧化钛溶液,将乙酸锌加入到2-甲氧基乙醇中获得氧化锌溶液,最后将两种溶液采用喷雾热解法在FTO(氟掺杂氧化锡)玻璃衬底上沉积TiO2/ZnO薄膜. Butalid等[68]采用三步法制得ZnO/TiO2纳米复合材料,首先,利用磁控溅射系统在玻璃衬底上生长锌膜,接下来通过热氧化将锌膜转化为ZnO,最后使用磁控溅射系统在ZnO表面沉积TiO2. 这些更具创新的方法为制备TiO2-ZnO复合材料提供了新的思路.
-
为了提升TiO2-ZnO复合材料的光催化性能,越来越多的研究者开始尝试对TiO2-ZnO进行进一步改性. 改性的方法大体上可分为3类:掺杂、构造异质结和锚定优化. 掺杂是指被掺杂物质直接引入TiO2-ZnO晶格内部;构造异质结是指一种或几种物质通过复合、沉积等手段与TiO2-ZnO结合,促进电子-空穴对的转移;锚定优化是指TiO2-ZnO复合材料通过与载体材料形成共价键,使得材料不易团聚、具有更大的机械强度[69].
-
掺杂元素会影响原材料的尺寸、形貌和晶体结构等特征,通常可掺杂金属或者非金属元素. 金属元素是通过改变原材料的结晶度或者引起材料的晶格缺陷,使得电子-空穴复合率降低,提高催化效果;非金属元素通过取代原材料中的氧元素,在原材料的带隙中产生新的能级,使禁带宽度变小,有效拓宽催化剂的光响应范围,改善对于太阳光的利用率. Ullah等[70]采用溶胶-凝胶法和回流技术合成Nd掺杂的TiO2-ZnO复合材料,掺杂后的复合材料禁带宽度降低到2.65 eV,有效地促进可见光吸收,最终提高光转换效率. Mofokeng等[71]使用溶胶-凝胶法制备了Dy3+掺杂的TiO2-ZnO复合材料,该材料光致发光(PL)强度有效降低,提高了电子-空穴的分离率,光催化活性得到进一步加强. Zhan等[72]溶胶-凝胶和浸渍法制得Mt掺杂的TiO2-ZnO复合材料,该材料的抑菌活性和润湿性能都明显增强.
表3总结了TiO2-ZnO元素掺杂改性的研究结果,从表3中可以看出,目前大多数研究者通过降解染料类有机污染物来表示掺杂元素后TiO2-ZnO复合材料的光催化效率,未来可研究掺杂后复合材料对于药物和个人护理品、全氟化合物类、多氯化萘等有机物污染物的降解效率,扩宽TiO2-ZnO光催化复合材料的适用范围.
-
在TiO2-ZnO复合材料表面沉积贵金属可以通过形成肖特基势垒异质结有效抑制电子-空穴对的复合,从而提高光催化活性. 常见的修饰内容包括Au、Ag、Pt等. Liang等[81]制备的TiO2-Au/ZnO三元异质结复合材料具有高效的光吸收能力,并且在光催化制氢和抗菌性能方面表现优异. Chen等[78]采用溶胶-凝胶法制备了Ag-TiO2/ZnO复合材料,该材料对MB染料的降解率可以达到83.65%. Xiang等[82]利用制备的Ag@TiO2@ZnO纳米材料,在紫外光照射下降解水中苯酚,TiO2/ZnO异质结和Ag/TiO2肖特基势垒的协同作用有效地增强了电子-空穴对的分离,提高了光催化活性,其反应速率常数是TiO2纳米颗粒的1.84倍和ZnO纳米颗粒的3.83倍. 贵金属沉积后TiO2-ZnO复合材料的催化效率可以显著增强,但贵金属价格较贵,因此在实际应用中很少被使用.
-
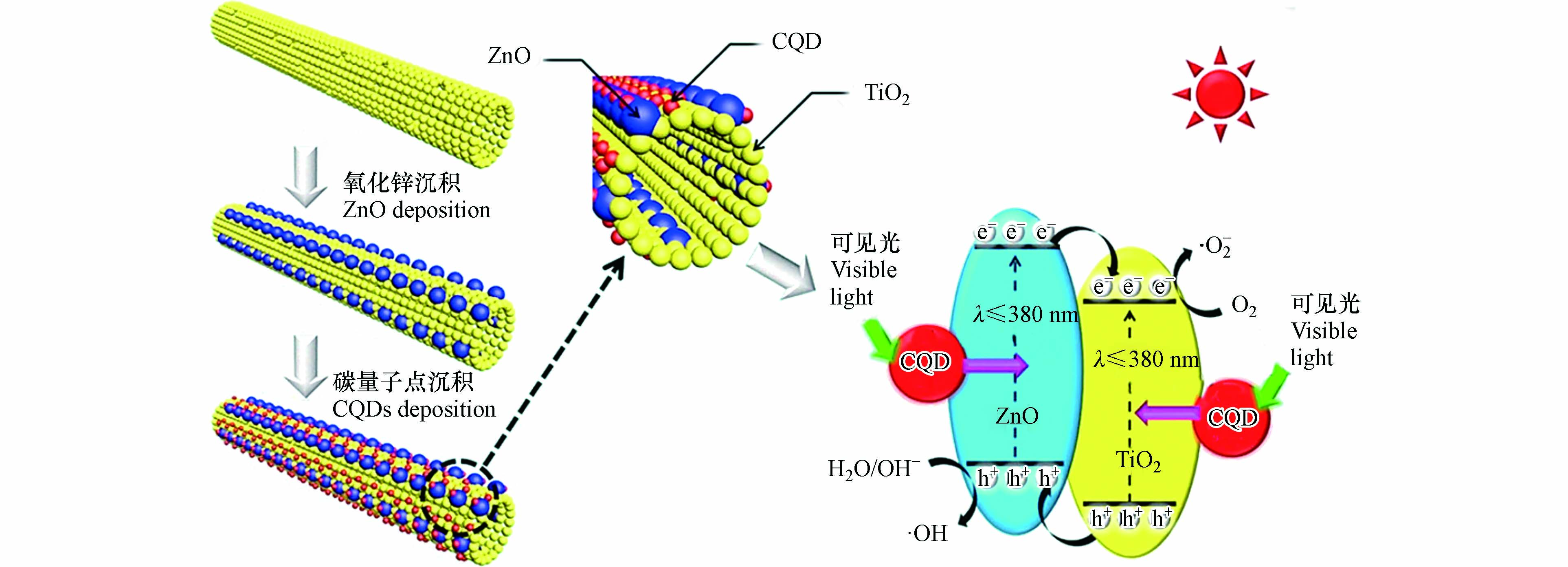
量子点(quantum dots,Qds)是指由元素周期表中Ⅱ—Ⅵ族、Ⅲ—Ⅴ族或Ⅳ—Ⅵ族双元素组成的零维半导体材料,颗粒尺寸通常等于或者小于其对应材料的激子波尔半径(一般为2—20 nm)[83 − 84]. 非碳量子点(如:CdS、PdS、ZnS等)具有高稳定性、易于表面修饰、优异的光学性能和环境友好等特点,与传统光催化剂结合后,可以形成异质结,提高光催化的效率[85];碳量子点(CQDs)具有可调节的可见光吸收和强的光致发光,与传统光催化剂结合后,可拓宽半导体催化剂的光吸收范围和促进电子-空穴对的分离[86]. Zhu等[87]通过电纺丝-水热法制备了碳量子点修饰的TiO2/ZnO复合材料,碳量子点本身没有催化效果,但与TiO2/ZnO复合材料结合后可以有效提高催化效果,对RhB染料的降解效率可达95%,图5是可见光下ZnO/TiO2复合材料在碳量子点修饰后的降解机理,碳量子点的引入,可将部分可见光转化为紫外-近紫外光,能够生成更多的电子-空穴对,提高光催化效果. Lee等[88]通过连续离子层吸附和反应(SILAR)法将PbS量子点沉积在ZnO/TiO2光催化剂上,制备出PbS/ZnO/TiO2光催化剂,由于PbS纳米粒子的量子尺寸效应以及载流子的高效转运,表现出较高的苯胺蓝降解效率(82%). 朱世从[89]采用溶胶-凝胶法制备出石墨烯量子点(GQDs)修饰的m-TiO2/ZnO(m-TiO2表示介孔TiO2)复合材料,GQDs可为光生电子提供无障碍通道,使得电子-空穴复合率降低,提高了光催化效果. An等[90]采用水热法用CdS量子点修饰三维有序空心球ZnTiO3-ZnO-TiO2复合材料,在模拟自然光下反应300 min后,对RhB染料的降解率在80%以上.
-
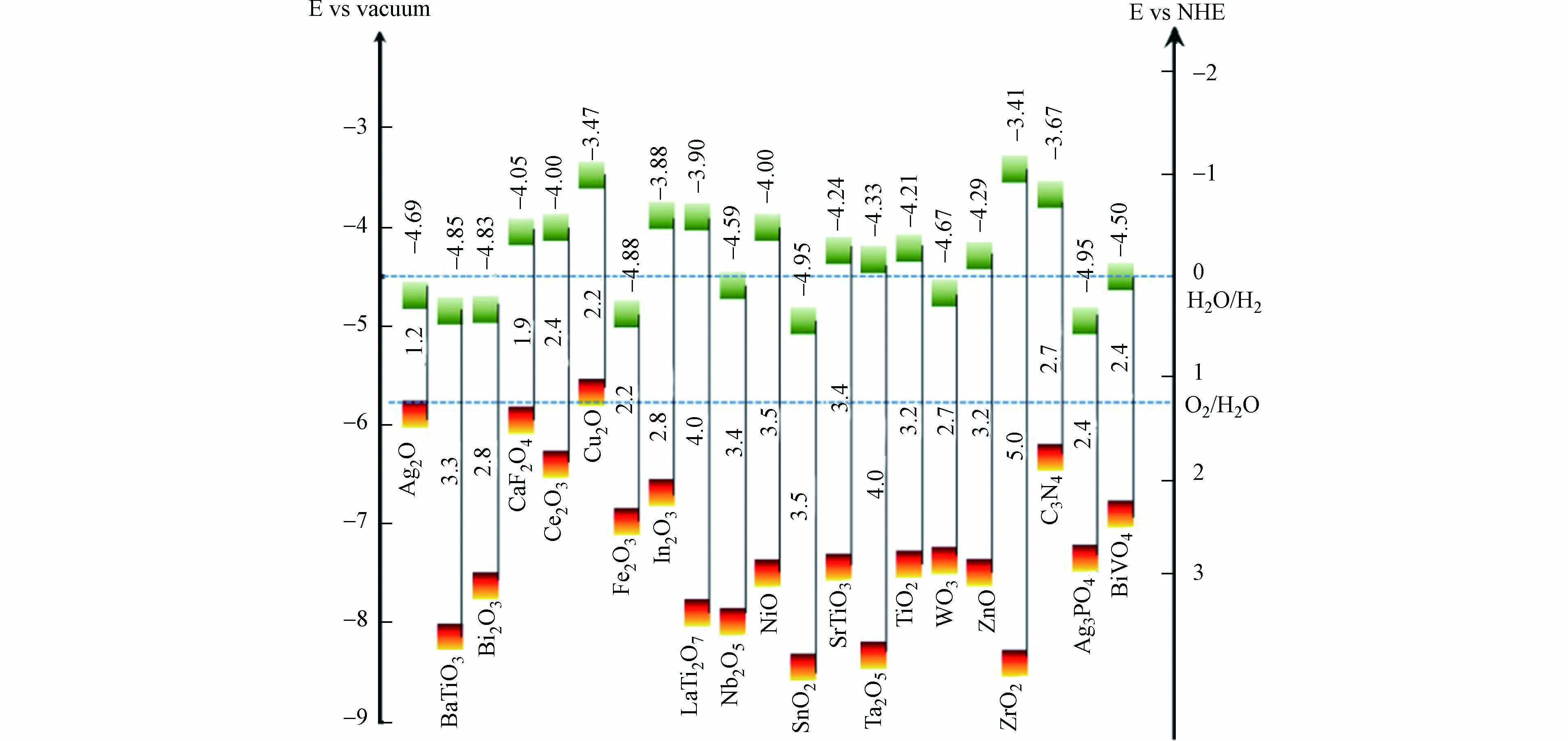
基于能带匹配原则,即不同半导体的禁带宽度不同,价带和导带之间存在差异[91],催化剂表面上复合材料的氧化还原电位需比催化剂本身的价带位置更负(能量更高),或者复合材料必须具有比催化剂本身的导带更正的氧化还原电位(能量更低)[92]. 在这种情况下,用其他半导体修饰TiO2-ZnO复合材料可形成异质结,有效阻止电子-空穴的复合,提高光催化效果,图6是常用半导体在pH=0时的禁带宽度及相对于真空和标准氢电势(NHE)的能级图(方块上面的数字表示相对于真空精确的导带能级,中间的数据表示带隙宽度)[93]. Wani等[25]在TiO2-ZnO中加入不同比例的Ag2O制备出三元复合材料,Ag2O的加入使催化剂的反应位点增加,对RhB染料表现出了很好的降解效率. Zhang等[94]采用水热法制备了ZnO-MoS2-TiO2光催化剂,分析结果表明复合材料的紫外和可见光吸收大大增强,电子和空穴的复合率降低,光催化效率提高. Liu等[95]合成的三元中空TiO2-ZnS@ZnO异质结,拓宽了可见光吸收范围,电子和空穴可以被有效分离,在可见光照射下对MB、RhB、二甲酚橙、酸性红18染料都具有良好的降解效果. Isac等[96]使用喷雾热解沉积制备了CuxS-ZnO-TiO2光活性异质结构,TiO2的价带低于ZnO和CuxS,从TiO2导带到CuxS导带的快速电子转移提高了异质结构电荷分离效率. 半导体异质结复合是复合材料改性常用的方法之一,但存在制备过程复杂、实际电荷载流子迁移途径不确定等问题,未来可在这两个方面对半导体异质结复合进行深入研究.
-
MXenes(二维过渡金属碳化物、氮化物或碳氮化物)材料和半导体结合能够形成异质结显著提高电子-空穴对的分离效率[97],并为光催化材料提供丰富的表面官能团,从而实现较高的光催化效果[98]. Ti3C2Tx是最常使用的MXenes材料,由于其具有表面积大、亲水性高、吸附效率高等特点,常用于废水处理[99]. Abbas等[100]使用NH4HF2刻蚀Ti3AlC2得到Ti3C2Tx MXene材料,接着采用水热法合成ZnO-TiO2-Ti3C2Tx复合材料,该材料在模拟太阳光对头孢曲松钠降解率可以达到99.4%,并且循环使用8次后降解率仍高达93%. Rdewi等[101]制备了ZnO-TiO2-Ti3C2Tx异质结构纳米光催化剂,在最佳条件下对卡马西平的去除率为99.6%,并且循环使用8次后依然具有95%以上的降解率. 目前对于MXenes材料复合TiO2-ZnO的研究较少,下一步可以继续利用MXenes材料优异的物理化学性质与TiO2-ZnO复合,进一步提高光催化效率.
-
将TiO2-ZnO复合光催化剂锚定在合适的载体上,能有效缓解催化材料易团聚、易流失等问题,可以增大催化剂与底物的接触面积,便于回收利用,提高光催化效率. 表4总结了近几年TiO2-ZnO复合催化剂锚定的材料及其主要结论,锚定材料的类型可分为无机物载体(如:活性炭、陶粒、碳纳米管等)和有机材料(如:聚氯乙烯、聚偏氟乙烯等),由于碳制材料具有来源广泛、价格低廉、制备简单等优点,因此常被用来作为TiO2-ZnO的锚定材料. 目前来说,对于有机材料作为TiO2-ZnO的锚定材料研究较少,但有机材料具有质地轻、柔韧性好、机械强度高、亲水性、比表面积大等特点[102 − 104],光催化材料与之结合后可以形成更大的活性表面,光氧化速率增加,传质降低,另外由于表面杂化的影响,拓宽了光催化剂的光响应范围,提高了对太阳光的利用率,光催化活性增强[105].
-
光催化是一种很有应用前景的污水处理技术,通过ZnO与TiO2复合及其进一步改性,可以显著降低电子与空穴的复合率,将光催化响应拓展到可见光区域,提高太阳光的利用率. 未来利用TiO2-ZnO复合材料光催化技术降解水中污染物的研究方向可集中在以下几个方面:
(1)TiO2-ZnO复合材料降解污染物的机理目前大多采用的是能带理论进行解释,但存在许多不足(如:电荷迁移机制不明确、改性方法的机理不具备普遍性等问题),未来可加强对于基础理论的研究,从而科学地制备出性能优异的光催化剂.
(2)当前对TiO2-ZnO复合材料的制备大多停留在传统方法中,今后的研究可加强计算机模拟技术(如第一性原理方法、密度泛函理论等)在该领域的应用,缩短材料从设计到应用的时间,节约人力、物力成本.
(3)TiO2-ZnO的锚定材料大多是无机材料(尤其是碳制材料),未来可以加强对于有机材料作为锚定材料的研究;可将药物和个人护理品、全氟化合物类、多氯化萘等有机污染物作为光催化降解的研究内容,拓宽TiO2-ZnO光催化剂的适用范围.
(4)目前大多TiO2-ZnO复合材料的制备都停留在实验室阶段,制备工艺相对复杂,后续可以优化制备方法,实现低成本、高产率的工业化.
(5)ZnO作为压电性的材料,具有良好的压电效应,可在外力作用下形成内建或者外建电场,有效地抑制电子-空穴的复合,因此今后可考虑在处理有机污水时加入超声波、搅拌等外力,将光催化技术与压电效应组合在一起形成“光-压电协同催化”,提高光催化效果.
TiO2-ZnO复合光催化剂制备与改性研究进展
Advances in synthesis and modification of TiO2-ZnO composite photocatalysts
-
摘要: 光催化作为一种绿色、高效的处理技术,在降解水中有机污染物方面有着巨大的优势. 二氧化钛(TiO2)是一种典型的光催化材料,与氧化锌(ZnO)复合后可以克服单一TiO2材料电子-空穴复合率高、可见光利用率低等缺点. 本文介绍了TiO2-ZnO光催化降解水中有机污染物的机理,重点系统综述了TiO2-ZnO复合材料的制备方法和进一步改性的研究,最后对未来TiO2-ZnO光催化技术的发展方向做出了展望.Abstract: As a green and efficient treatment technology, photocatalysis has great advantages in the degradation of pollutants in wastewater. Titanium dioxide (TiO2) as a typical photocatalytic material, its drawbacks such as high electron-hole recombination rate and low visible-light utilization can be overcome by the combination with zinc oxide (ZnO). In this review, the mechanism of TiO2-ZnO photocatalytic degradation of organic pollutants in wastewater was introduced. Synthesis methods and further modification of TiO2-ZnO composites were systematically summarized. Finally, the development direction of TiO2-ZnO photocatalytic technology in the future was prospected.
-
Key words:
- titanium dioxide /
- zinc oxide /
- photocatalysis /
- synthesis /
- modification.
-

-
表 1 溶胶-凝胶法制备TiO2-ZnO复合材料使用的原料
Table 1. Raw materials for TiO2-ZnO composites prepared by sol-gel method
表 2 水热法制备TiO2-ZnO复合材料
Table 2. Preparation of TiO2-ZnO composite by hydrothermal method
原料
Raw material高压釜的条件
Conditions of autoclave主要形貌
Main morphology文献References 钛酸异丙酯、乙酸锌、异丙醇、硝酸 180 ℃下24 h 棒状的ZnO和片状的TiO2复合,平均长度约为2.75 μm [49] 乙酸锌、硼氢化钠、聚乙烯吡咯烷酮、氨水、硫酸钛 120 ℃下3 h
150 ℃下16 h海胆状ZnO/TiO2复合材料,TiO2与ZnO之间形成直接的
Z-scheme(Z方案)异质结[46] 乙酸锌、钛酸异丙酯、乙酸、氢氧化钠、乙醇 150 ℃下18 h Zn/Ti为2:1时,样品颗粒高度团聚,平均粒径为8—11 nm [50] 表 3 TiO2/ZnO复合材料元素掺杂改性
Table 3. Element doping modification of TiO2-ZnO composites
掺杂元素
Doping element合成方法
Fabrication method污染物
Pollutants底物浓度(mg·L−1)
Substrate
concentration光源
Light source反应时间/min
Reaction time降解率/%
Degradation efficiency文献
ReferencesB 机械法和溶剂法 MB 15 太阳光 15 95 [73] N 溶胶-凝胶法 亚硫酸钠 0.38 氙灯(紫外光) 90 100 [74] C 水热法和煅烧法 RhB 10 氙灯(320- 1100 nm)30 94 [75] F 共沉淀法 MO 20 金卤灯(280-780 nm) 35 93.75 [76] Cu 溶胶-凝胶法 MO 20 可见光(>400 nm) 150 97 [77] Al 溶胶-凝胶法 MB 3 紫外灯(365 nm) 240 66.36 [78] Nd和Er 溶胶-凝胶法 苯酚 20 汞灯(紫外光) 180 90.88 [79] Gd 溶胶-凝胶法 MB 20 紫外灯(365 nm) 90 93 [80] MO 20 90 94.4 表 4 TiO2-ZnO锚定材料及其主要结论总结
Table 4. Summary of TiO2-ZnO anchored materials and their main conclusions
锚定材料
Anchoring
material制备方法
Fabrication
method污染物
Pollutants底物浓度(mg·L−1)
Substrate
concentration光源
Light source反应时间/min
Reaction
time降解率/%
Degradation
efficiency文献
References聚丙烯腈(PAN) 电弧放电法静电纺丝法 孔雀绿 3.65 紫外灯(254 nm) 240 100 [106] 聚氯乙烯 沉积法 百草枯 20 紫外灯 240 73 [107] 聚偏氟乙烯 原子层沉积 MB 3.2 氙灯(400—780 nm) 30 80 [108] 磁性Fe3O4 水热法 RhB 10 汞灯 120 98.11 [109] 多孔陶粒 水热法 MB 10 氙灯(200—400 nm) 180 96.3 [110] 碳纳米管CNF 静电纺丝法和水热工艺 MB 10 紫外灯(365 nm) 120 >90 [111] 碳纳米管CNF 溶剂热法 RhB 5 紫外-可见光 40 100 [112] 活性炭 涂覆法 二甲苯 100 紫外灯 110 95.5 [113] 活性炭 溶胶-凝胶法和浸涂法 砷 9.8 汞灯(365 nm) 287.6 99.9 [114] 活性炭 沉淀法和固定化 类黑色素 5000 紫外-可见光 60 97 [115] 还原氧化石墨烯(RGO) 溶剂热法 MB 0.3 氙灯(可见光) 120 92 [116] RGO 水热法 MO — 可见光 120 90 [117] 海泡石 溶胶-凝胶法和沉淀法 氟甲喹 10 紫外灯(260 nm) 240 85 [118] 羟基磷灰石 机械法 MB 0.32 卤素灯 120 95 [119] MO 0.33 卤素灯 120 45 三元壳聚糖 溶胶-凝胶法 四环素 20 紫外灯(365 nm) 180 97.2 [120] 硅藻土 沉淀法 气态甲苯 — 氙灯(可见光) 210 63.5 [121] 气态丙酮 210 53.3 MB 150 80.34 -
[1] FUJISHIMA A, HONDA K. Electrochemical photolysis of water at a semiconductor electrode[J]. Nature, 1972, 238(5358): 37-38. doi: 10.1038/238037a0 [2] VILD A, TEIXEIRA S, KÜHN K, et al. Orthogonal experimental design of titanium dioxide—Poly(methyl methacrylate) electrospun nanocomposite membranes for photocatalytic applications[J]. Journal of Environmental Chemical Engineering, 2016, 4(3): 3151-3158. doi: 10.1016/j.jece.2016.06.029 [3] 吴士杰. 二氧化钛/金属复合光催化剂在二氧化碳还原和固氮中的应用及催化机理研究[D]. 金华: 浙江师范大学, 2021. WU S J. Applications of titanium dioxide/metal composites in photocatalytic carbon dioxide reduction and nitrogen fixation and related mechanism studies[D]. Jinhua: Zhejiang Normal University, 2021 (in Chinese).
[4] CAGLAR A, AKTAS N, KIVRAK H. Photocatalytic glucose electrooxidation of titanium dioxide doped CdTe enhanced for a photocatalytic fuel cell[J]. Fuel, 2022, 330: 125653. doi: 10.1016/j.fuel.2022.125653 [5] 董德志. TiO2及ZnO-TiO2基地聚物光催化剂微球的制备及其对盐酸四环素降解性能的研究[D]. 南宁: 广西大学, 2022. DONG D Z. Preparation of TiO2 and ZnO-TiO2 based polymer photocatalyst microspheres and their degradation properties for tetracycline hydrochloride[D]. Nanning: Guangxi University, 2022 (in Chinese).
[6] ARAÚJO E S, DA COSTA B P, OLIVEIRA R A P, et al. TiO2/ZnO hierarchical heteronanostructures: Synthesis, characterization and application as photocatalysts[J]. Journal of Environmental Chemical Engineering, 2016, 4(3): 2820-2829. doi: 10.1016/j.jece.2016.05.021 [7] HUANG X J, ZENG X F, WANG J X, et al. Synthesis of monodispersed ZnO@SiO2 nanoparticles for anti-UV aging application in highly transparent polymer-based nanocomposites[J]. Journal of Materials Science, 2019, 54(11): 8581-8590. doi: 10.1007/s10853-019-03393-z [8] 马良. 水热法制备二氧化钛和其氧化锌纳米复合材料及光催化性能研究[D]. 西安: 西北大学, 2016. MA L. Hydrothermal synthesis and photocatalytic performance of TiO2 nanoparticles and TiO2/ZnO nanocomposites[D]. Xi'an: Northwest University, 2016 (in Chinese).
[9] VIET T Q Q, KHOI V H, THI HUONG GIANG N, et al. Statistical screening and optimization of photocatalytic degradation of methylene blue by ZnO–TiO2/rGO nanocomposite[J]. Colloids and Surfaces A:Physicochemical and Engineering Aspects, 2021, 629: 127464. doi: 10.1016/j.colsurfa.2021.127464 [10] 蒋银花, 邱琳, 张文莉, 等. ZnO掺杂TiO2复合空心球的制备方法及其应用: CN101905153A[P]. 2010-12-08. JIANG Y H, QIU L, ZHANG W L, et al. Preparation method and application of ZnO-doped TiO2 composite hollow sphere: CN101905153A[P]. 2010-12-08(in Chinese).
[11] SHAO D L, SUN H T, XIN G Q, et al. High quality ZnO-TiO2 core-shell nanowires for efficient ultraviolet sensing[J]. Applied Surface Science, 2014, 314: 872-876. doi: 10.1016/j.apsusc.2014.06.182 [12] WILLIAM C, MATTHEW P, JUSTIN C, et al. Tunable atmospheric microplasma synthesis of nitrogen-doped zinc oxide and titanium dioxide nanostructures[J]. Nano-Structures & Nano-Objects, 2022, 30: 100866. [13] 杭子清. 简析光催化技术及其研究现状[J]. 资源节约与环保, 2021(2): 122-123. doi: 10.3969/j.issn.1673-2251.2021.02.064 HANG Z Q. Brief analysis of photocatalytic technology and its research status[J]. Resources Economization & Environmental Protection, 2021(2): 122-123 (in Chinese). doi: 10.3969/j.issn.1673-2251.2021.02.064
[14] 王颖, 杨传玺, 王小宁, 等. 二维光催化材料研究进展[J]. 有色金属科学与工程, 2021, 12(2): 30-42. doi: 10.13264/j.cnki.ysjskx.2021.02.005 WANG Y, YANG C X, WANG X N, et al. Research advances on two-dimensional materials using as photocatalysts[J]. Nonferrous Metals Science and Engineering, 2021, 12(2): 30-42 (in Chinese). doi: 10.13264/j.cnki.ysjskx.2021.02.005
[15] MAHY J G, DESCHAMPS F, COLLARD V, et al. Acid acting as redispersing agent to form stable colloids from photoactive crystalline aqueous Sol–gel TiO2 powder[J]. Journal of Sol-Gel Science and Technology, 2018, 87(3): 568-583. doi: 10.1007/s10971-018-4751-6 [16] KUMAR J S, BOLIMERA U R, THANGADURAI P. Direct sunlight responsive ZnO photocatalyst: Highly efficient photodegradation of methylene blue[C]//DAE SOLID STATE PHYSICS SYMPOSIUM 2018, 2019. [17] ZANGENEH H, ZINATIZADEH A A, ZINADINI S, et al. Visible light-driven photoactive L-Methionine (CNS tripledoped)-TiO2/ZnO nanocomposite aiming for highly efficient photodegradation of xenobiotic compounds in wastewater[J]. Materials Research Bulletin, 2022, 150: 111783. doi: 10.1016/j.materresbull.2022.111783 [18] 崔舒. 氧化锌和二氧化钛材料的制备及其光催化、气敏性能研究[D]. 长春: 吉林大学, 2019. CUI S. Fabrication, photocatalytic and gas sensing properties of ZnO and TiO2 materials[D]. Changchun: Jilin University, 2019 (in Chinese).
[19] 崔义慧. 硒化镉敏化二氧化钛及氧化锌纳米光催化材料的制备及性能研究[D]. 沈阳: 沈阳师范大学, 2016. CUI Y H. Study on the preparation and properties of TiO2 and ZnO photocatalytic materials sensitized by CdSe[D]. Shenyang: Shenyang Normal University, 2016 (in Chinese).
[20] JOHRA F T, JUNG W G. RGO–TiO2–ZnO composites: Synthesis, characterization, and application to photocatalysis[J]. Applied Catalysis A:General, 2015, 491: 52-57. doi: 10.1016/j.apcata.2014.11.036 [21] HOU X B, STANLEY S L, ZHAO M, et al. MOF-based C-doped coupled TiO2/ZnO nanofibrous membrane with crossed network connection for enhanced photocatalytic activity[J]. Journal of Alloys and Compounds, 2019, 777: 982-990. doi: 10.1016/j.jallcom.2018.10.174 [22] EL-SHAZLY A N, RASHAD M M, ABDEL-AAL E A, et al. Nanostructured ZnO photocatalysts prepared via surfactant assisted Co-Precipitation method achieving enhanced photocatalytic activity for the degradation of methylene blue dyes[J]. Journal of Environmental Chemical Engineering, 2016, 4(3): 3177-3184. doi: 10.1016/j.jece.2016.06.018 [23] XU G, ZHANG Y, PENG D D, et al. Nitrogen-doped mixed-phase TiO2 with controllable phase junction as superior visible-light photocatalyst for selective oxidation of cyclohexane[J]. Applied Surface Science, 2021, 536: 147953. doi: 10.1016/j.apsusc.2020.147953 [24] 李江鸿, 梁发文, 陈必儒, 等. 非金属掺杂石墨氮化碳复合材料光催化协同活化过一硫酸盐降解有机污染物的研究进展[J]. 环境化学, 2022, 41(10): 3457-3468. doi: 10.7524/j.issn.0254-6108.2022050101 LI J H, LIANG F W, CHEN B R, et al. Research progress of nonmetallic doped graphite carbon nitride composites in photocatalytic activation of peroxymonosulfate system for organic pollutants removal[J]. Environmental Chemistry, 2022, 41(10): 3457-3468 (in Chinese). doi: 10.7524/j.issn.0254-6108.2022050101
[25] WANI S I, GANIE A S. Ag2O incorporated ZnO−TiO2 nanocomposite: Ionic conductivity and photocatalytic degradation of an organic dye[J]. Inorganic Chemistry Communications, 2021, 128: 108567. doi: 10.1016/j.inoche.2021.108567 [26] BAO H V, DAT N M, GIANG N T H, et al. Behavior of ZnO-doped TiO2/rGO nanocomposite for water treatment enhancement[J]. Surfaces and Interfaces, 2021, 23: 100950. doi: 10.1016/j.surfin.2021.100950 [27] 张冬梅. 可回收TiO2及ZnO纳米复合光催化剂的制备及其光催化降解性能的研究[D]. 大连: 大连理工大学, 2021. ZHANG D M. Synthesis and photocatalytic degradation performance of recyclable TiO2 and ZnO nanocomposite photocatalysts[D]. Dalian: Dalian University of Technology, 2021 (in Chinese).
[28] RAJABI H R, ARJMAND H, KAZEMDEHDASHTI H, et al. A comparison investigation on photocatalytic activity performance and adsorption efficiency for the removal of cationic dye: Quantum dots vs. magnetic nanoparticles[J]. Journal of Environmental Chemical Engineering, 2016, 4(3): 2830-2840. doi: 10.1016/j.jece.2016.05.029 [29] 毕祥. 二氧化钛基和氧化锌基半导体光催化剂的制备与性能研究[D]. 西安: 陕西科技大学, 2021. BI X. Study on preparation and performance of titanium dioxide-based and zinc oxide-based semiconductor photocatalysts[D]. Xi'an: Shaanxi University of Science & Technology, 2021 (in Chinese).
[30] 刘庆祎, 肖桐, 孙文杰, 等. 纳米二氧化钛强化的相变储能研究进展[J]. 化工学报, 2022, 73(5): 1863-1882. LIU Q W, XIAO T, SUN W J, et al. Progress in the research of phase change energy storage enhanced by titanium dioxide nanoparticles[J]. CIESC Journal, 2022, 73(5): 1863-1882 (in Chinese).
[31] 彭伟校, 王开军, 胡劲, 等. 纯/掺杂纳米氧化锆粉体水热法制备研究进展[J]. 材料导报, 2013, 27(19): 146-149. PENG W X, WANG K J, HU J, et al. Research progress on preparation of pure/doped nano-zirconia powders by hydrothermal[J]. Materials Review, 2013, 27(19): 146-149 (in Chinese).
[32] 杜炜, 艾超前, 马雪东, 等. 无机晶须的制备及应用进展[J]. 应用化工, 2019, 48(5): 1163-1166,1171. doi: 10.3969/j.issn.1671-3206.2019.05.040 DU W, AI C Q, MA X D, et al. Progress in the application and preparation of inorganic whiskers[J]. Applied Chemical Industry, 2019, 48(5): 1163-1166,1171 (in Chinese). doi: 10.3969/j.issn.1671-3206.2019.05.040
[33] BAO Y, ZHANG Y H, MA J Z, et al. Progress in preparation and application of one-dimensional nano zinc oxide[J]. Cailiao Gongcheng/Journal of Materials Engineering, 2015, 43(2): 103-112. [34] 邸琬茗. ZnO的均匀沉淀法制备及其在费托反应中的应用[D]. 杭州: 浙江工业大学, 2015. DI W M. The preparation of ZnO by homogeneous precipitation method and its applications in Fischer-Tropsch synthesis[D]. Hangzhou: Zhejiang University of Technology, 2015 (in Chinese).
[35] 廖培义, 陈延明, 王立岩, 等. 醇-水体系无表面活性剂纳米Zn O的制备及表征[J]. 材料导报, 2021, 35(S1): 108-111. LIAO P Y, CHEN Y M, WANG L Y. Synthesis and characterization of ZnO nanocrystals in alcohol-water without surfactants[J]. Materials Reports, 2021, 35(S1): 108-111 (in Chinese).
[36] QUINTERO Y, MOSQUERA E, DIOSA J, et al. Ultrasonic-assisted Sol–gel synthesis of TiO2 nanostructures: Influence of synthesis parameters on morphology, crystallinity, and photocatalytic performance[J]. Journal of Sol-Gel Science and Technology, 2020, 94(2): 477-485. doi: 10.1007/s10971-020-05263-6 [37] SIWIŃSKA-STEFAŃSKA K, KUBIAKA A, PIASECKI A, et al. TiO2-ZnO binary oxide systems: Comprehensive characterization and tests of photocatalytic activity[J]. Materials (Basel, Switzerland), 2018, 11(5): 841. doi: 10.3390/ma11050841 [38] UPADHYAY G K, RAJPUT J K, PATHAK T K, et al. Synthesis of ZnO: TiO2 nanocomposites for photocatalyst application in visible light[J]. Vacuum, 2019, 160: 154-163. doi: 10.1016/j.vacuum.2018.11.026 [39] IDRIS N J, ABU BAKAR S, MOHAMED A, et al. Photocatalytic performance improvement by utilizing GO_MWCNTs hybrid solution on sand/ZnO/TiO2-based photocatalysts to degrade methylene blue dye[J]. Environmental Science and Pollution Research, 2021, 28(6): 6966-6979. doi: 10.1007/s11356-020-10904-y [40] DUBEY R S. Temperature-dependent phase transformation of TiO2 nanoparticles synthesized by Sol-gel method[J]. Materials Letters, 2018, 215: 312-317. doi: 10.1016/j.matlet.2017.12.120 [41] TEKIN D, KIZILTAS H, UNGAN H. Kinetic evaluation of ZnO/ TiO2 thin film photocatalyst in photocatalytic degradation of Orange G[J]. Journal of Molecular Liquids, 2020, 306: 112905. doi: 10.1016/j.molliq.2020.112905 [42] RAHMANI F, ARDYANIAN M. Fabrication and characterization of ZnO/TiO2 multilayers, deposited via spin coating method[J]. Journal of Materials Science:Materials in Electronics, 2018, 29(5): 4285-4293. doi: 10.1007/s10854-017-8375-3 [43] AL-HAZMI F E, YAKUPHANOGLU F. Photoconducting and photovoltaic properties of ZnO: TiO2 composite/p-silicon heterojunction photodiode[J]. Silicon, 2018, 10(3): 781-787. doi: 10.1007/s12633-016-9530-9 [44] KUBIAK A, SIWIŃSKA-CIESIELCZYK K, JESIONOWSKI T. Titania-based hybrid materials with ZnO, ZrO2 and MoS2: A review[J]. Materials, 2018, 11(11): 2295. doi: 10.3390/ma11112295 [45] CHENG P F, WANG Y L, XU L P, et al. High specific surface area urchin-like hierarchical ZnO-TiO2 architectures: Hydrothermal synthesis and photocatalytic properties[J]. Materials Letters, 2016, 175: 52-55. doi: 10.1016/j.matlet.2016.03.120 [46] ZHOU Y, TAN Y W, XIANG Y, et al. Construction of urchin-like ZnO/TiO2 direct Z-scheme system to improve charge separation[J]. ChemistrySelect, 2019, 4(44): 12963-12970. doi: 10.1002/slct.201903905 [47] XUAN M C T, TRAN T N, BOTTO C, et al. Zinc-containing precursor dependence of hydrothermal method for the synthesis of N-doped ZnO photocatalysts[J]. Chemical Engineering Communications, 2021, 208(2): 149-158. doi: 10.1080/00986445.2019.1694917 [48] ZHU L, LI Y Q, ZENG W. Hydrothermal synthesis of hierarchical flower-like ZnO nanostructure and its enhanced ethanol gas-sensing properties[J]. Applied Surface Science, 2018, 427: 281-287. doi: 10.1016/j.apsusc.2017.08.229 [49] RAJENDRAN K, GAJENDIRAN M, KIM S, et al. Synthesis and characterization of biocompatible zinc oxide nanorod doped-titanium dioxide nanosheet[J]. Journal of Industrial and Engineering Chemistry, 2018, 57: 387-395. doi: 10.1016/j.jiec.2017.08.047 [50] SUTAR R S, BARKUL R P, PATIL M K. Sunlight assisted photocatalytic degradation of different organic pollutants and simultaneous degradation of cationic and anionic dyes using titanium and zinc based nanocomposites[J]. Journal of Molecular Liquids, 2021, 340: 117191. doi: 10.1016/j.molliq.2021.117191 [51] ZHA R H, NADIMICHERLA R, GUO X. Ultraviolet photocatalytic degradation of methyl orange by nanostructured TiO2/ZnO heterojunctions[J]. Journal of Materials Chemistry A, 2015, 3(12): 6565-6574. doi: 10.1039/C5TA00764J [52] HUO S P, DING S W, ZHAO C Y, et al. Growth and photocatalytic activities of porous ZnO/TiO2 composite microspheres with crystalline–amorphous phase boundary[J]. Catalysis Letters, 2021, 151(7): 1937-1947. doi: 10.1007/s10562-020-03463-x [53] ZHANG X, CHEN G H, LI W H, et al. Preparation and photocathodic protection properties of ZnO/TiO2 heterojunction film under simulated solar light[J]. Materials (Basel, Switzerland), 2019, 12(23): 3856. doi: 10.3390/ma12233856 [54] 李化全, 邱贵宝, 吕学伟. 脱硝催化剂载体二氧化钛制备研究进展[J]. 钢铁钒钛, 2022, 43(2): 7-14,20. doi: 10.7513/j.issn.1004-7638.2022.02.002 LI H Q, QIU G B, LV X W. Research progress in preparation of titanium dioxide catalyst carrier for denitrification[J]. Iron Steel Vanadium Titanium, 2022, 43(2): 7-14,20 (in Chinese). doi: 10.7513/j.issn.1004-7638.2022.02.002
[55] 李红霖. 氧化锌及其复合半导体材料的制备、表征与性能研究[D]. 西安: 陕西科技大学, 2014. LI H L. Synthesis, characterization and properties research of zinc oxide and its composite[D]. Xi'an: Shaanxi University of Science & Technology, 2014 (in Chinese).
[56] 卫世腾. 络合沉淀制备CeO2及其光屏蔽性能研究[D]. 沈阳: 东北大学, 2020. WEI S T. Preparation of CeO2 by complex precipitation and its light-shieding performance[D]. Shenyang: Northeastern University, 2020 (in Chinese).
[57] NIBRET A, YADAV O P, DÍAZ I, et al. Cr-N co-doped ZnO nanoparticles: Synthesis, characterization and photocatalytic activity for degradation of thymol blue[J]. Bulletin of the Chemical Society of Ethiopia, 2015, 29: 247-258. doi: 10.4314/bcse.v29i2.8 [58] MORLANDO A, CARDILLO D, DEVERS T, et al. Titanium doped tin dioxide as potential UV filter with low photocatalytic activity for sunscreen products[J]. Materials Letters, 2016, 171: 289-292. doi: 10.1016/j.matlet.2016.02.094 [59] LACHOM V, POOLCHARUANSIN P, LAOKUL P. Preparation, characterizations and photocatalytic activity of a ZnO/TiO2nanocomposite[J]. Materials Research Express, 2017, 4(3): 035006. doi: 10.1088/2053-1591/aa60d1 [60] HAGHIGHATZADEH A, HOSSEINI M, MAZINANI B, et al. Improved photocatalytic activity of ZnO-TiO2 nanocomposite catalysts by modulating TiO2 thickness[J]. Materials Research Express, 2019, 6(11): 115060. doi: 10.1088/2053-1591/ab49c4 [61] BROOMS T J, OTIENO B, ONYANGO M S, et al. Photocatalytic degradation of P-Cresol using TiO2/ZnO hybrid surface capped with polyaniline[J]. Journal of Environmental Science and Health, Part A, Toxic/Hazardous Substances & Environmental Engineering, 2018, 53(2): 99-107. [62] ARABNEZHAD M, AFARANI M S, JAFARI A. Co-precipitation synthesis of ZnO–TiO2 nanostructure composites for arsenic photodegradation from industrial wastewater[J]. International Journal of Environmental Science and Technology, 2019, 16(1): 463-468. doi: 10.1007/s13762-017-1585-7 [63] 朱雪松. 尖晶石型锌铁氧体的水热法制备及其磁性能研究[D]. 马鞍山: 安徽工业大学, 2021. ZHU X S. Preparation and magnetic properties of spinel zinc ferrite obtained by A hydrothermal method[D]. Maanshan: Anhui University of Technology, 2021 (in Chinese).
[64] RILDA Y, DAMARA D, Syukri, et al. Synthesis of ZnO-TiO2/chitosan nanorods by using precipitation methods and studying their structures and optics properties at different precursor molar compositions[J]. IOP Conference Series:Earth and Environmental Science, 2019, 217(1): 12015. [65] KUMARI P, BAHADUR N, CONLAN X A, et al. Stimuli-responsive heterojunctions based photo-electrocatalytic membrane reactors for reactive filtration of persistent organic pollutants[J]. Chemical Engineering Journal, 2023, 452: 139374. doi: 10.1016/j.cej.2022.139374 [66] ZHOU Y, YANG L Y, LU J, et al. Photoelectric properties of three-dimensional urchin-like zinc oxide/titanium dioxide composite micronanostructures[J]. Micro & Nano Letters, 2016, 11(5): 277-280. [67] KHAN M I, IMRAN S, SHAHNAWAZ, et al. Annealing effect on the structural, morphological and electrical properties of TiO2/ZnO bilayer thin films[J]. Results in Physics, 2018, 8: 249-252. doi: 10.1016/j.rinp.2017.12.030 [68] BUTALID ROGEL JAN B, CRISTOBAL ANNA PATRICIA S, MONTALLANA ARANTXA DANIELLE S, et al. Stability of TiO2-coated ZnO photocatalytic thin films for photodegradation of methylene blue[J]. Journal of Vacuum Science & Technology B, 2020, 38(6): 062205. [69] 张瑞英. 基于席夫碱和氯己定的共价有机聚合物的合成及性质研究[D]. 沈阳: 辽宁大学, 2021. ZHANG R Y. Synthesis and properties of covalent organic polymer based on schiff base and chlorhexidine[D]. Shenyang: Liaoning University, 2021 (in Chinese).
[70] ULLAH N, ERTEN-ELA Ş, MUJTABA SHAH S, et al. Selected organic dyes (carminic acid, pyrocatechol violet and dithizone) sensitized metal (silver, neodymium) doped TiO2/ZnO nanostructured materials: A photoanode for hybrid bulk heterojunction solar cells[J]. Spectrochimica Acta Part A:Molecular and Biomolecular Spectroscopy, 2022, 278: 121387. doi: 10.1016/j.saa.2022.121387 [71] MOFOKENG S J, KUMAR V, KROON R E, et al. Structure and optical properties of Dy3+ activated Sol-gel ZnO-TiO2 nanocomposites[J]. Journal of Alloys and Compounds, 2017, 711: 121-131. doi: 10.1016/j.jallcom.2017.03.345 [72] ZHAN J H, CHEN H Y, ZHOU H J, et al. Mt-supported ZnO/TiO2 nanocomposite for agricultural antibacterial agent involving enhanced antibacterial activity and increased wettability[J]. Applied Clay Science, 2021, 214: 106296. doi: 10.1016/j.clay.2021.106296 [73] SHATHY R A, FAHIM S A, SARKER M, et al. Natural sunlight driven photocatalytic removal of toxic textile dyes in water using B-doped ZnO/TiO2 nanocomposites[J]. Catalysts, 2022, 12(3): 308. [74] 胡敏, 郭嘉, 吴华东, 等. N-Zn/ TiO2光催化氧化脱硫废液中亚硫酸钠的研究[J]. 无机盐工业, 2020, 52(10): 151-156. HU M, GUO J, WU H D, et al. Photocatalytic oxidation of sodium sulfite in desulfurization wastewater by N-Zn/TiO2[J]. Inorganic Chemicals Industry, 2020, 52(10): 151-156 (in Chinese).
[75] WANG Y M, LIU X X, GUO L, et al. Metal organic framework-derived C-doped ZnO/TiO2 nanocomposite catalysts for enhanced photodegradation of Rhodamine B[J]. Journal of Colloid and Interface Science, 2021, 599: 566-576. doi: 10.1016/j.jcis.2021.03.167 [76] ZHANG L Y, YANG J J, YOU Y H. Construction and photocatalytic performance of fluorinated ZnO-TiO2 heterostructure composites[J]. RSC Advances, 2021, 11(61): 38654-38666. doi: 10.1039/D1RA07757K [77] KHAKI M R D, SHAFEEYAN M S, RAMAN A A A, et al. Enhanced UV–visible photocatalytic activity of Cu-doped ZnO/TiO2 nanoparticles[J]. Journal of Materials Science:Materials in Electronics, 2018, 29(7): 5480-5495. doi: 10.1007/s10854-017-8515-9 [78] CHEN W J, HSU K C, FANG T H, et al. Characteristics and heterostructure of metal-doped TiO2/ZnO nanocatalysts[J]. Current Applied Physics, 2022, 38: 1-6. doi: 10.1016/j.cap.2022.03.001 [79] 陈昕海. Nd-Er/ZnO-TiO2光催化材料制备及水体有机酚类化合物光催化降解研究[D]. 重庆: 重庆三峡学院, 2018. CHEN X H. Preparation and characterization of Nd-Er/ZnO-TiO2 photocatalytic materials study on photocatalytic degradation of organic phenols compounds in surface water[D]. Chongqing: Chongqing Three Gorges University, 2018 (in Chinese).
[80] PANWAR S, UPADHYAY G K, PUROHIT L P. Gd-doped ZnO: TiO2 heterogenous nanocomposites for advance oxidation process[J]. Materials Research Bulletin, 2021, 145: 111534. [81] LIANG Y, LI W, WANG X, et al. TiO2-ZnO/Au ternary heterojunction nanocomposite: Excellent antibacterial property and visible-light photocatalytic hydrogen production efficiency[J]. Ceramics International, 2022, 48(2): 2826-2832. doi: 10.1016/j.ceramint.2021.10.072 [82] XIANG X B, YU Y, WEN W, et al. Construction of hierarchical Ag@TiO2@ZnO nanowires with high photocatalytic activity[J]. New Journal of Chemistry, 2018, 42(1): 265-271. doi: 10.1039/C7NJ02302B [83] 李汉明. CsPbI3量子点表面调控构筑高效稳定发光二极管研究[D]. 长春: 吉林大学, 2022. LI H M. Research on efficient and stable light-emitting diodes constructed by surface control of CsPbI3 quantum dot[D]. Changchun: Jilin University, 2022 (in Chinese).
[84] 都晗. 基于碳量子点和DNA四面体信号放大的两种食源性致病菌夹心生物传感器机理研究[D]. 泰安: 山东农业大学, 2022. DU H. Mechanism research of sandwich biosensor for two foodborne pathogens based on carbon quantum dots and DNA tetrahedron signal amplification[D]. Taian: Shandong Agricultural University, 2022 (in Chinese).
[85] BAJOROWICZ B, KOBYLAŃSKI M P, GOŁĄBIEWSKA A, et al. Quantum dot-decorated semiconductor micro- and nanoparticles: A review of their synthesis, characterization and application in photocatalysis[J]. Advances in Colloid and Interface Science, 2018, 256: 352-372. doi: 10.1016/j.cis.2018.02.003 [86] LIU J C, ZHU W Y, YU S Y, et al. Three dimensional carbogenic dots/TiO2 nanoheterojunctions with enhanced visible light-driven photocatalytic activity[J]. Carbon, 2014, 79: 369-379. doi: 10.1016/j.carbon.2014.07.079 [87] ZHU M, DENG X C, LIN X, et al. The carbon quantum dots modified ZnO/TiO2 nanotube heterojunction and its visible light photocatalysis enhancement[J]. Journal of Materials Science:Materials in Electronics, 2018, 29(13): 11449-11456. doi: 10.1007/s10854-018-9237-3 [88] LEE J H, AHN H J, YOUN J, et al. Synthesis and characterization of ZnO/TiO2 photocatalyst decorated with PbS QDs for the degradation of aniline blue solution[J]. Korean Journal of Metals and Materials, 2018, 56(12): 900-909. doi: 10.3365/KJMM.2018.56.12.900 [89] 朱世从. 两种氧化物/石墨烯量子点/介孔二氧化钛光催化剂的制备及其在光催化苯乙烯环氧化反应中的应用[D]. 淮北: 淮北师范大学, 2021. ZHU S C. Preparation of two kinds of oxide/graphene quantum dots/mesoporous titanium dioxide photocatalysts and their application in photocatalytic epoxidation of styrene[D]. Huaibei: Huaibei Normal University, 2021 (in Chinese).
[90] AN M Z, LI L, WU Q Q, et al. CdS QDs modified three-dimensional ordered hollow spherical ZnTiO3-ZnO-TiO2 composite with improved photocatalytic performance[J]. Journal of Alloys and Compounds, 2022, 895: 162638. doi: 10.1016/j.jallcom.2021.162638 [91] BAI S L, CHU H M, XIANG X, et al. Fabricating of Fe2O3/BiVO4 heterojunction based photoanode modified with NiFe-LDH nanosheets for efficient solar water splitting[J]. Chemical Engineering Journal, 2018, 350: 148-156. doi: 10.1016/j.cej.2018.05.109 [92] FATIMAH I, FADILLAH G, YANTI I, et al. Clay-supported metal oxide nanoparticles in catalytic advanced oxidation processes: A review[J]. Nanomaterials (Basel, Switzerland), 2022, 12(5): 825. doi: 10.3390/nano12050825 [93] LI J T, WU N Q. Semiconductor-based photocatalysts and photoelectrochemical cells for solar fuel generation: A review[J]. Catalysis Science & Technology, 2015, 5(3): 1360-1384. [94] ZHANG H Y, YU H, DAI J D, et al. Microstructure, photoluminescence and photocatalytic activity of ZnO-MoS2-TiO2 composite[J]. Chinese Journal of Physics, 2018, 56(6): 3053-3061. doi: 10.1016/j.cjph.2018.10.015 [95] LIU D D, LIANG H O, XU T, et al. Construction of ternary hollow TiO2-ZnS@ZnO heterostructure with enhanced visible-light photoactivity[J]. Journal of Molecular Structure, 2022, 1248: 131493. doi: 10.1016/j.molstruc.2021.131493 [96] ISAC L, ANDRONIC L, VISA M, et al. Selective photocatalytic degradation of organic pollutants by Cu xS/ZnO/TiO2 heterostructures[J]. Ceramics International, 2020, 46(4): 4265-4273. doi: 10.1016/j.ceramint.2019.10.147 [97] PRASAD C, YANG X F, LIU Q Q, et al. Recent advances in MXenes supported semiconductors based photocatalysts: Properties, synthesis and photocatalytic applications[J]. Journal of Industrial and Engineering Chemistry, 2020, 85: 1-33. doi: 10.1016/j.jiec.2019.12.003 [98] KUANG P Y, LOW J X, CHENG B, et al. MXene-based photocatalysts[J]. Journal of Materials Science & Technology, 2020, 56: 18-44. [99] ZHAN X X, SI C, ZHOU J, et al. MXene and MXene-based composites: Synthesis, properties and environment-related applications[J]. Nanoscale Horizons, 2020, 5(2): 235-258. doi: 10.1039/C9NH00571D [100] ABBAS K K, ABDULKADHIMAL-GHABAN A M H, RDEWI E H. Synthesis of a novel ZnO/TiO2-nanorod MXene heterostructured nanophotocatalyst for the removal pharmaceutical ceftriaxone sodium from aqueous solution under simulated sunlight[J]. Journal of Environmental Chemical Engineering, 2022, 10(4): 108111. doi: 10.1016/j.jece.2022.108111 [101] RDEWI E H, ABBAS K K, ABDULKADHIMAL-GHABAN A M H. Removal pharmaceutical carbamazepine from wastewater using ZnO-TiO2-MXene heterostructural nanophotocatalyst under solar light irradiation[J]. Materials Today:Proceedings, 2022, 60(P3): 1702-1711. [102] JOSEPH A, VIJAYANANDAN A. Review on support materials used for immobilization of nano-photocatalysts for water treatment applications[J]. Inorganica Chimica Acta, 2023, 545: 121284. doi: 10.1016/j.ica.2022.121284 [103] HAGHIGHAT N, VATANPOUR V, SHEYDAEI M, et al. Preparation of a novel polyvinyl chloride (PVC) ultrafiltration membrane modified with Ag/TiO2 nanoparticle with enhanced hydrophilicity and antibacterial activities[J]. Separation and Purification Technology, 2019, 237(24): 116374. [104] ULLAH S, FERREIRA-NETO E P, KHAN A A, et al. Supported nanostructured photocatalysts: The role of support-photocatalyst interactions[J]. Photochemical & Photobiological Sciences, 2023, 22(1): 219-240. [105] ENESCA A, CAZAN C. Polymer composite-based materials with photocatalytic applications in wastewater organic pollutant removal: A mini review[J]. Polymers, 2022, 14(16): 3291. doi: 10.3390/polym14163291 [106] YAR A, HASPULAT B, ÜSTÜN T, et al. Electrospun TiO2/ZnO/PAN hybrid nanofiber membranes with efficient photocatalytic activity[J]. RSC Advances, 2017, 7(47): 29806-29814. doi: 10.1039/C7RA03699J [107] GARBA A A, MATMIN J, TOEMEN S. Highly Stable Photocatalytic Removal of Paraquat Dichloride using ZnO/TiO2 supported on PVC[J]. Malaysian Journal of Fundamental and Applied Sciences, 2021, 17(5): 647-658. doi: 10.11113/mjfas.v17n5.2424 [108] LI N, TIAN Y, ZHANG J, et al. Precisely-controlled modification of PVDF membranes with 3D TiO2/ZnO nanolayer: Enhanced anti-fouling performance by changing hydrophilicity and photocatalysis under visible light irradiation[J]. Journal of Membrane Science, 2017, 528: 359-368. doi: 10.1016/j.memsci.2017.01.048 [109] 赵瑨云, 徐婕, 穆寄林, 等. 磁性Fe3O4@TiO2/ZnO的制备及光催化性能研究[J]. 人工晶体学报, 2020, 49(7): 1223-1228. doi: 10.3969/j.issn.1000-985X.2020.07.012 ZHAO J Y, XU J, MU J L, et al. Preparation and photocatalytic property of magnetic Fe3O4@TiO2/ZnO[J]. Journal of Synthetic Crystals, 2020, 49(7): 1223-1228 (in Chinese). doi: 10.3969/j.issn.1000-985X.2020.07.012
[110] 白宁娜. ZnO光催化材料的改性及TiO2/ZnO@PC光催化剂的制备及性能[D]. 西安: 西安科技大学, 2021. BAI N N. Modification of ZnO photocatalytic materials and preparation and properties of TiO2/ZnO@PC photocatalyst[D]. Xi'an: Xi'an University of Science and Technology, 2021 (in Chinese).
[111] PANT B, OJHA G P, KUK Y S, et al. Synthesis and characterization of ZnO-TiO2/carbon fiber composite with enhanced photocatalytic properties[J]. Nanomaterials (Basel, Switzerland), 2020, 10(10): 1960. doi: 10.3390/nano10101960 [112] HUANG Y Z, LI R K, CHEN D P, et al. Synthesis and characterization of CNT/TiO2/ZnO composites with high photocatalytic performance[J]. Catalysts, 2018, 8(4): 151. doi: 10.3390/catal8040151 [113] RANGKOOY H A, JAHANI F, SIAHI AHANGAR A. Photocatalytic removal of xylene as a pollutant in the air using ZnO-activated carbon, TiO2-activated carbon, and TiO2/ZnOactivated carbon nanocomposites[J]. Environmental Health Engineering and Management, 2020, 7(1): 41-47. doi: 10.34172/EHEM.2020.06 [114] SAGHARLOO N G, RABANI M, SALIMI L, et al. Immobilized ZnO/TiO2 activated carbon (I ZnO/TiO2 AC) to removal of arsenic from aqueous environments: Optimization using response surface methodology and kinetic studies[J]. Biomass Conversion and Biorefinery, 2021,13(12):10483–10494. [115] OTIENO B O, APOLLO S O, NAIDOO B E, et al. Photodecolorisation of melanoidins in vinasse with illuminated TiO2-ZnO/activated carbon composite[J]. Journal of Environmental Science and Health, Part A, Toxic/Hazardous Substances & Environmental Engineering, 2017, 52(7): 616-623. [116] RAGHAVAN N, THANGAVEL S, VENUGOPAL G. Enhanced photocatalytic degradation of methylene blue by reduced graphene-oxide/titanium dioxide/zinc oxide ternary nanocomposites[J]. Materials Science in Semiconductor Processing, 2015, 30: 321-329. doi: 10.1016/j.mssp.2014.09.019 [117] PAN X Y, YANG P, NAN H, et al. Preparation and enhanced visible-light photoelectrocatalytic activity of ternary TiO2ZnO/RGO nanocomposites[J]. Electrochimica Acta, 2018, 261: 284-288. doi: 10.1016/j.electacta.2017.12.143 [118] VAIZOĞULLAR A İ. TiO2/ZnO supported on sepiolite: Preparation, structural characterization, and photocatalytic degradation of flumequine antibiotic in aqueous solution[J]. Chemical Engineering Communications, 2017, 204(6): 689-697. doi: 10.1080/00986445.2017.1306518 [119] YEASMIN Z, ALIM A, AHMED S, et al. Synthesis, characterization and efficiency of HAp-TiO2-ZnO composite as a promising photocatalytic material[J]. Transactions of the Indian Ceramic Society, 2018, 77(3): 161-168. doi: 10.1080/0371750X.2018.1512380 [120] ASADZADEH PATEHKHOR H, FATTAHI M, KHOSRAVI-NIKOU M. Synthesis and characterization of ternary chitosan-TiO2-ZnO over graphene for photocatalytic degradation of tetracycline from pharmaceutical wastewater[J]. Scientific Reports, 2021, 11(1): 24177. doi: 10.1038/s41598-021-03492-5 [121] YANG B B, MA Z X, WANG Q, et al. Synthesis and photoelectrocatalytic applications of TiO2/ZnO/diatomite composites[J]. Catalysts, 2022, 12(3): 268. doi: 10.3390/catal12030268 -



 下载:
下载: