-
饮用水消毒是公共卫生安全的保障。然而,在消毒过程中消毒剂会与原水中的天然有机物(NOM)、污染物、溴/碘化合物等生成消毒副产物(DBPs)。DBPs可以通过吸入、皮肤接触以及食用含DBPs的水和食物等进入人体,对人体健康有着严重的危害。自1974年第一次检测出氯仿以来,DBPs已引起国内外学者的广泛关注,当前已有700多种DBPs于饮用水中被检出[1]。
在世界范围内,海水中溴储量占地球总溴储量的99%,平均浓度约为65 mg·L−1。溴通过海水入侵、地质结构变化等自然过程进入地表水和地下水[2]。美国和加拿大23个城市水源Br¯浓度在2.4—1120.0 μg·L−1,在以色列和澳大利亚主要水源中的Br¯浓度高达2000.0 μg·L−1和4000.0 μg·L−1[3]。我国东部地区Br¯浓度在22.0—233.0 μg·L−1[4]。杨永亮等[5]对辽宁省西部和沈阳地区河水及地下水进行溴化物含量检测,地表水和地下水平均Br¯浓度分别为180 μg·L−1和1584 μg·L−1。除自然条件外,溴还会通过燃煤电厂、常规油气开采、工业排放、水力压裂、电子产品回收等人为活动排放至原水中。例如,燃煤电厂在其湿式洗涤器中加入溴,以减少汞排放,该过程导致排放到地表水中的电厂废水中的Br¯浓度高达50 mg·L−1;水力压裂产生的废水中含有的Br¯浓度高达60 mg·L−1[6]。当水厂原水中含有Br—时,经饮用水消毒后溴代消毒副产物(Br-DBPs)的生成会增加[7],研究表明,其细胞和遗传毒性要高于同系氯代消毒副产物(Cl-DBPs)[8-9],并且与出生缺陷率呈正相关关系[10-11]。
目前,我国的饮用水水质标准仅对三溴甲烷(TBM)、一氯二溴甲烷(CDBM)、二氯一溴甲烷(DCBM)等3种THMs以及二溴乙腈(DBAN)和溴酸盐(BrO3¯)做了相关规定。美国环境保护署(USEPA)除此之外还对一溴乙酸(MBAA)和二溴乙酸(DBAA)两种卤乙酸(HAAs)进行监管。Br¯含量、加氯量、pH值、温度、DOC等与Br-DBPs的生成有着紧密的联系。本文总结了已知的Br-DBPs种类、存在情况、影响因素和未知的Br-DBPs的识别进展,以期为水中Br-DBPs的生成控制提供参考。
-
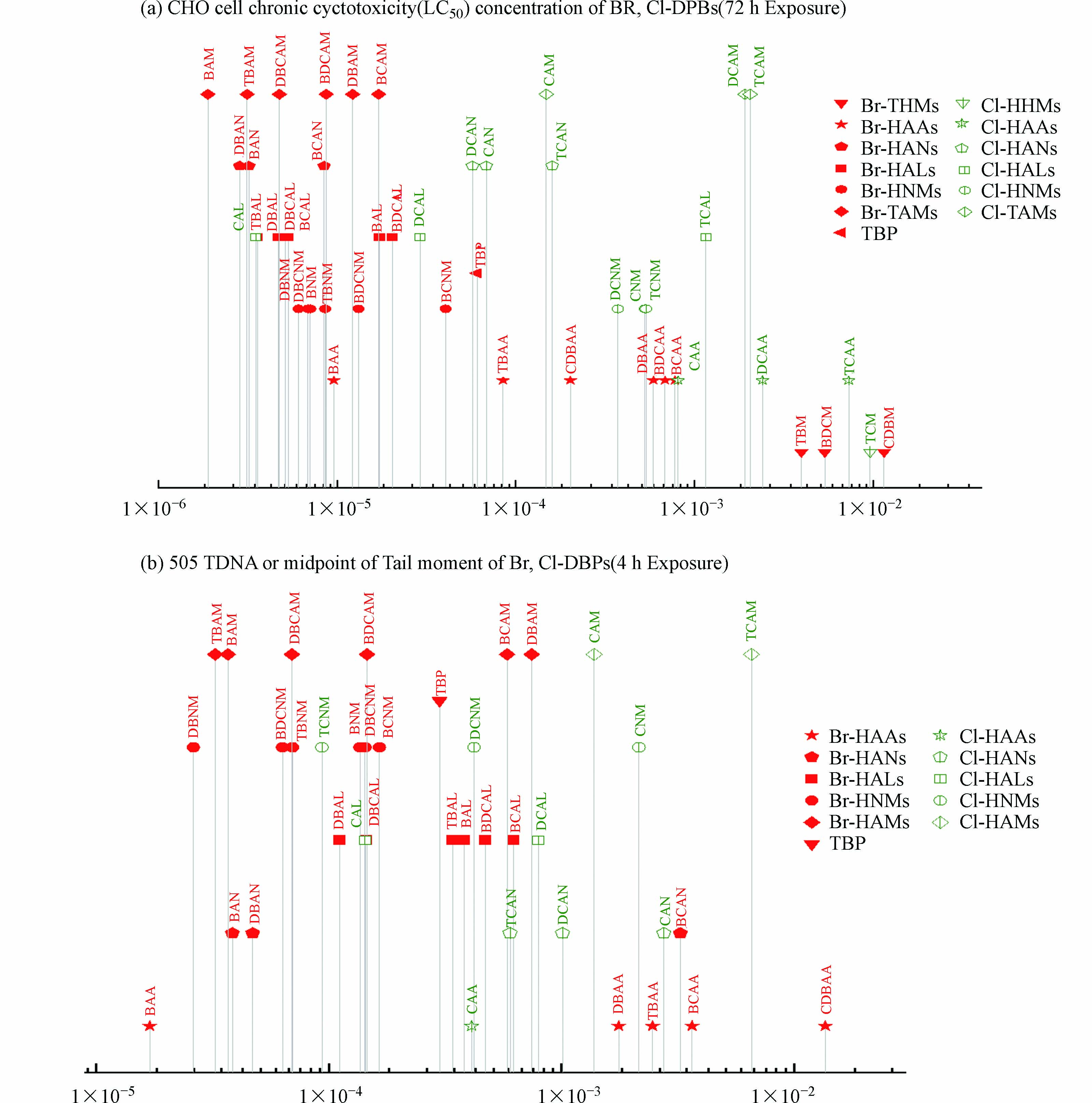
目前已知的Br-DBPs可分为八大类,如表1—5所示。饮用水中的Br-DBPs浓度多在ng·L−1至μg·L−1水平,其中Br-THMs的检出率较高。尽管Br-DBPs浓度通常低于Cl-DBPs浓度,但它们具有更高的细胞毒性和遗传毒性(图1)[12]。
-
Br-THMs是在饮用水检出最多的Br-DBPs,TBM和二溴一氯甲烷(DBCM)约占饮用水总THMs的20%。Dobaradaran等[13]对伊朗布什尔省某些水厂出水的THMs浓度进行调查,TBM的检出浓度为29.5—47.7 μg·L−1 ,DBCM检出浓度为21.1—38.5 μg·L−1,一溴二氯甲烷(BDCM)检出浓度为12—25 μg·L−1。Richardson等[7]对以色列的某些水厂进行检测,经氯消毒、二氧化氯加氯胺消毒以及二氧化氯加氯消毒后TBM检出浓度为19.50—61.00 μg·L−1,DBCM检出浓度在1.90—9.39 μg·L−1之间,BDCM检出浓度在0.20—0.64 μg L−1之间,还发现当氯消毒时这3种Br-THMs年平均浓度最高。我国饮用水中也普遍检测出了Br-THMs,刘晓琳等[14]对江苏某水厂的DBPs进行检测,氯消毒后城市管网中Br-THMs浓度高达50.18 μg·L−1,占总THMs的57.27%。王姗姗[15]对辽宁省6座城市饮用水中的THMs进行检测,DBCM、BDCM和TBM检出率分别为75.6%、67.8%、35.6%。
世界卫生组织(WHO)在2011年修订的第四版《饮用水水质准则》中规定BDCM、DBCM、TBM的浓度不得高于60、100、100 μg·L−1。国际瘤症研究机构(IARC)将BDCM列为2B组(可能对人类致癌),DBCM和TBM为第3组(不能按其对人类的致癌性进行分组)。BDCM具有致瘤性,最新的核苷三磷酸(NTP)生物测试还表明BDCM可能对生育造成影响(增加自发性流产或死胎的风险)。
-
溴代乙酸(Br-HAAs)是Br-DBPs中非常重要的一类。以色列在高溴(Br¯浓度为2 mg·L−1)饮用水中检测出二溴乙酸(DBAA)和溴氯乙酸(BCAA),检出浓度为12.00—38.70、1.00—3.90 μg·L−1[7](表2)。孟丽苹等[16]对全国34个城市117家自来水厂出水中9种卤乙酸(HAAs)含量进行调查,BCAA检出率可达31.6%,一溴二氯乙酸(BDCAA)、DBAA和二溴一氯乙酸(DBCAA)检出率相对较低,分别为13.7%、10.3%和5.1%,未检测出溴乙酸(BAA)和三溴乙酸(TBAA)。
Br-HAAs中BAA最具细胞毒性和遗传毒性[12]。国内外目前并没有规定Br-HAAs的限值,这是由于缺少包括亚慢性和慢性毒性、多代生殖毒性、标准的发育毒性和致癌性的相关数据。
-
溴代乙腈(Br-HANs)、溴代乙酰胺(Br-HAMs)、溴代硝基甲烷(Br-HNMs)和溴代砒咯(TBP)是毒性很强的含氮消毒副产物(N-DBPs)。相关研究表明(表3),饮用水中普遍检测出了Br-HANs[17-18]。Ding等[17]对中国31个城市70家水厂的DBPs进行检测发现,溴乙腈(BAN)、溴氯乙腈(BCAN)、二溴乙腈(DBAN)的检出频率分别为56%、40%、8%。美国水行业协会(AWWA)调查美国500家大型水厂发现,出厂水中BCAN和DBAN浓度范围在0.5—40.0 μg·L−1。目前,《饮用水水质准则》(2011)中只规定了DBAN准则值为70 μg·L−1。
目前国内已有许多学者对饮用水中Br-HAMs进行了检测[19-21]。Br-HANs在水解和降解过程中会生成Br-HAMs,且氯化程度越高,Br-HANs水解程度越大[22]。Br-HAMs有独立的形成机理,腐殖质已被证明是其有效的前体物。尽管Br-HANs和Br-HAMs浓度较某些碳基Br-DBPs(如Br-THMs、Br-HAAs)低,但毒性更强。在所有已知的Br-DBPs中,Br-HAMs细胞毒性最强,遗传毒性仅次于Br-HANs[23]。BCAN在细菌检测中具有致突变性。
1991年,第一种被发现的溴代硝基甲烷(Br-HNMs)是三溴硝基甲烷(TBNM),当时被检测到的浓度约为2 μg·L−1[24]。虽然大多数Br-HNMs在消毒(氯和氯胺消毒)过的饮用水中(不含臭氧)也有发现[25-26],但含量较臭氧消毒后的低得多,这表明臭氧可以促进Br-HNMs生成[24]。Br-HNMs是毒性很高的哺乳动物细胞毒素和基因毒素,Plewa等[12]的研究表明Br-HNMs的半数致死浓度(LC50)在10−5 mg·L−1左右。
溴代砒咯,2,3,5-三溴吡咯(TBP)在2003年首次作为饮用水DBPs被发现[7]。THMs和HAAs的主要前驱物是黄腐酸,而TBP主要由腐殖酸形成。它的细胞毒性是受监管的DBAA的8倍,是中华仓鼠卵巢细胞(CHO)细胞中DNA损伤的有效诱导剂[9]。Yang等[27]在氯化的盐水废水中检测并鉴定出一组卤代吡咯,包括四溴吡咯、三溴氯吡咯、三溴碘吡咯和三溴吡咯,其中以四溴吡咯为主。
-
有研究表明,没食子酸(gallic acid,3,4,5-三羟基苯甲酸)是水源水中普遍存在的多酚类化合物,可作为一些极性苯酚类Br-DBPs的前驱物[23]。Pan等[4]对添加了没食子酸的模拟饮用水进行分析,首次发现了2,6-二氯-4-溴苯酚、2,6-二溴-4-氯苯酚、2,4,6-三溴苯酚、3-溴-5-氯-4-羟基苯甲醛、3,5-二溴-4-羟基苯甲醛、3-溴-5-氯-4-羟基苯甲酸、3,5-二溴-4-羟基苯甲酸、3-溴-5-氯水杨酸、3,5-二溴水杨酸等9种芳香族Br-DBP s的生成(表4),并对我国东部8个重点城市饮用水中上述副产物进行检测,浓度范围如表4所示。近年来,已经有33种新型卤代苯酚类消毒副产物被鉴定[28]。这些芳香族Br-DBPs比脂肪族以及常规链状结构DBPs更具生长抑制性、发育毒性和慢性细胞毒性,并且其还可以分解形成Br-THMs或(和)Br-HAAs。
-
卤代醛(HALs)从质量浓度上看是饮用水中仅次于THMs和HAAs的第三大类DBPs[9]。通过与其他5种DBPs(包括THM和HAA)对比总体细胞毒性和遗传毒性,已有学者将HALs列为第二大类细胞毒性DBPs[29]。
Plewa等[12]用CHO对已知Br-DBPs的细胞毒性进行检测,研究发现三溴乙醛(TBAL)的LC50最低,是BAA的一半,比Br-THMs低3个数量级。Gao等[30]研究了加拿大两个大型饮用水处理厂的HALs浓度,对5种溴代乙醛进行了检测,由于原水溴含量较低,所以未检出二溴乙醛(DBAL)与溴氯乙醛(BCAL),二溴一氯乙醛(DBCAL)与一溴二氯乙醛(BDCAL)含量较高。当由于盐水入侵而导致原水中溴化物含量较高时,水厂出水中会存在更高水平的溴氯乙醛(BCAL)[31]。在美国对部分饮用水系统进行的一项研究发现,原水中高浓度溴化物的含量(540 μg·L−1)会导致Br-HALs的大量生成[29](表5)。
-
溴源是饮用水中形成Br-DBPs的先决条件。自然界中的溴离子(Br-)、溴酸盐(BrO3¯)和溴化有机物是水厂原水的主要溴源。原水中Br¯浓度是影响Br-DBPs生成的关键因素。给水处理通常需要投加混凝剂去除溶解性有机碳(DOC),但这个过程无法降低Br¯浓度,而且会导致混凝出水中Br¯/DOC增大,更易形成Br-DBPs [32]。Zhang等[33]研究发现当原水中溴化物浓度超过100 μg·L−1时,溴代三卤甲烷(Br-THMs)是总三卤甲烷(THMs)的主要成分。富含Br¯的原水在氯或氯胺消毒后,Br-DBPs的生成量明显高于相同处理条件下低Br¯浓度的原水[34]。随着Br¯浓度的增加,Chang等[35]发现THMs和HAAs的形态逐渐由氯化物变为溴氯化合物再到溴化物。当原水中溴化物浓度低于10 μg·L−1时,Kolb等[36]检测出Br-THMs生成;当溴化物浓度超过100 μg·L−1时,Br-THMs是THMs的主体部分[33]。Zhou等[37]研究发现HNMs与Br¯浓度有显著的正相关关系,与THMs、HAAs、HANs不同,即使Br¯在低浓度范围,HNMs的生成也会明显受到Br¯的影响。
-
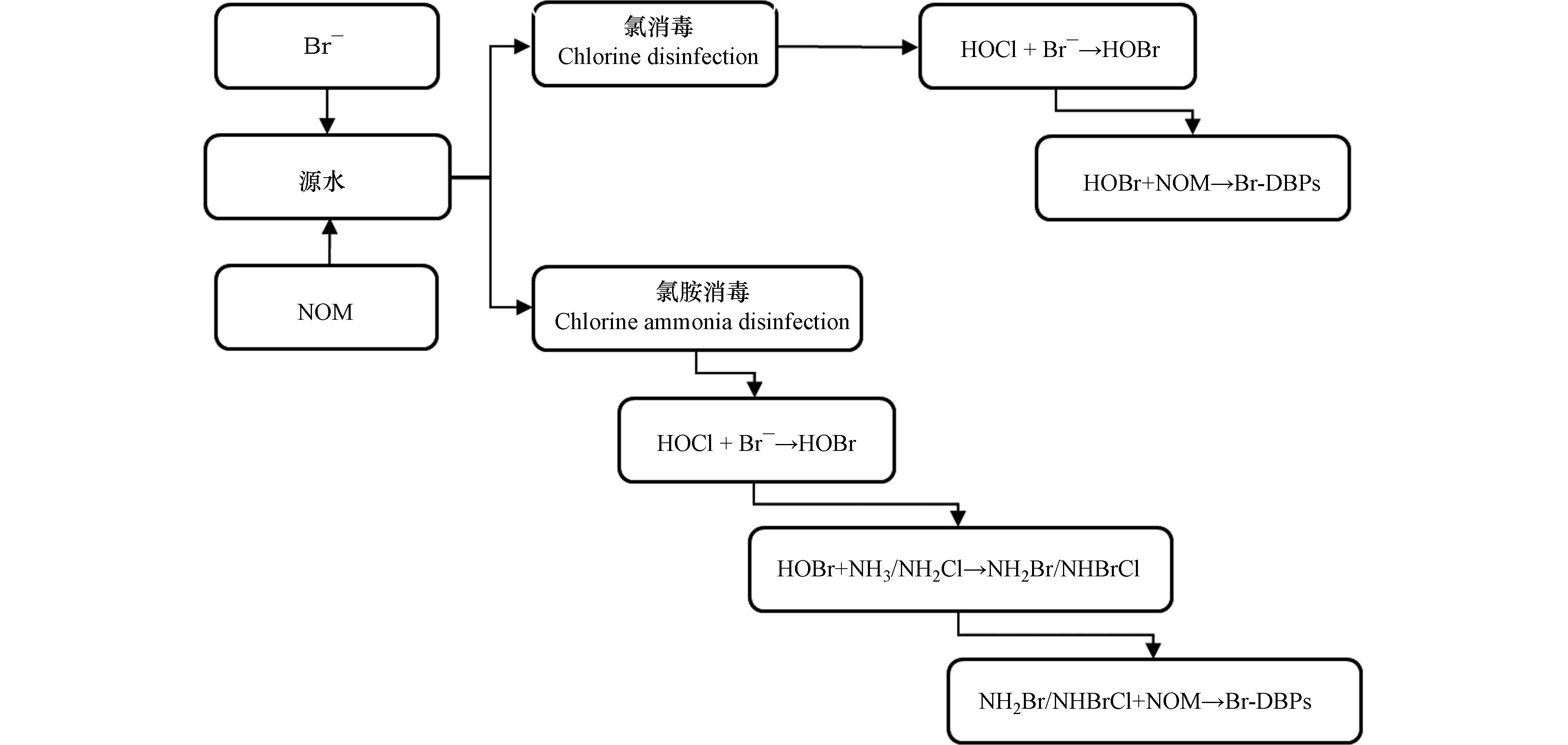
氯与氯胺消毒是自来水厂常用的两种消毒方式,两者消毒过程中Br-DBPs生成的机理略有不同(图2)。采用氯消毒时,极性高分子Br-DBPs会迅速生成,后分解为低分子Br-DBPs。与氯消毒不同的是,氯胺消毒易形成芳香族和含氮的极性Br-DBPs,且会在水中积累。
天然水体中存在的NOM是Br-DBPs生成的主要前驱物,具有胺、氨基酸、芳香族氨基酸、羧酸和苯酚等基团的NOM,均易与Br—生成Br-DBPs。在氯和氯氨消毒过程中,氧化剂次氯酸(HOCl)会结合溴离子生成次溴酸(HOBr),HOBr相较于HOCl反应活性更强[38],在氯胺消毒过程中,HOBr会分别与胺和氯胺结合生成溴胺(NH2Br)和溴氯胺(NHBrCl)。NOM包括能够与HOBr、NH2Br以及NHBrCl发生快速反应的NOMfast和缓慢反应的NOMslow,HOBr、NH2Br以及NHBrCl会结合NOMfast生成Br-DBPs的中间产物(Br-DBPsint),之后Br-DBPsint与HOCl反应生成Cl-DBPs;NOMslow会与HOBr、NH2Br以及NHBrCl反应直接生成Br-DBPs[39-40]。主要化学反应式和速率常数如表6。
Br-DBPs在管道的输配水过程中也有生成。由于pH值的变化、溶解氧的存在,以及微生物的繁殖,管道会受到化学和生物腐蚀。管道的腐蚀会导致管内壁结垢以及金属离子、金属氧化物向水中释放。例如,应用广泛的铸铁管道中存在铁单质、氧化铁、铁盐类氧化物等物质,它们会与水中残留的Br¯、余氯、NOM、DBPs等发生复合反应(氧化、还原、吸附、催化等),这不仅会消耗水中的余氯,还会进一步影响Br-DBPs的生成。有研究表明,铁质管道内水中的THMs含量会增加[41-42],而HAAs的形成会受到抑制[43]。Hozalski等[44]研究发现零价铁在供水系统中对HAAs有一定的降解作用,TBAA甚至可以完全脱卤为乙酸。Yang等[45]发现铁离子的存在会降低HANs的生成。
当水体中存在铜离子(Cu2+)或铅离子(Pb2+)时,Zha等[46]发现基于原电池原理的Cu/Fe、Pb/Fe的双金属系统,在不同离子浓度和pH值下,对TBM和TBAA均有不同程度的降解作用。刘立超[47]的研究表明Pb2+的存在会促进Br-THMs的生成。另外,含溴水中的钙离子、镁离子会成为催化剂共同促进Br¯与腐殖酸上官能团结合,导致Br-THMs的增加[48]。金属氧化物对饮用水中的溴转化起催化作用,而碘离子可以被金属氧化物氧化,所以相比于金属氧化物对碘代消毒副产物的生成影响[43],Br-DBPs受到的影响不大。除此之外,DBPs的质量浓度还会随管线的延长呈上升趋势,由此带来的健康风险也会随之增加[49]。
-
当水体pH值上升时会导致THMs和HNMs的生成量增加,同时HAAs、HANs的生成量会减少[50]。有学者指出,THMs生成量增多是因为pH的升高增强了DBPs的水解(脱卤)[51]。Sohn等[32]研究发现当pH值由6.5增大到8.5,Br-THM与THMs之比上升,BrO3¯浓度也会增大,但pH对Br-HAAs影响较小。实际上,pH值的变化,更多是在影响HOCl与HOBr的形态变化。pH值低时,HOCl的氧化性强,更易生成HOBr,反之,HOBr生成量少便会降低Br-DBPs的生成[52]。
饮用水加热过程可影响Br-DBPs的生成。Pan等[4]发现煮沸后大多数的极性Br-DBPs都是不稳定的。沸腾5 min后,TBAA、DBCAA和BDCAA分别减少了90.5%,86.9%和79.6%,热脱羧是这些非挥发性的DBPs煮沸后分解的原因。但是,煮沸会导致自来水中一些极性Br-DBPs的积累,包括两种二卤乙酸(DBAA和BCAA)以及4种芳香族极性Br-DBPs(2,4,6-三溴酚、3,5-二溴-4羟基苯甲醛、3,5-二溴水杨酸和3,5-二溴-4-羟基苯甲酸)。整体而言,根据毒性检测结果表明,煮沸会降低人类摄入Br-DBPs的风险。
NOM是大多数水体内总有机碳(TOC)的主要成分,也是DBPs的主要前驱物。根据水源地地质和生态环境的不同,NOM有亲水和疏水两种性质,亲水性不同,NOM的分布也就随之不同,进而会影响到饮用水中DBPs的分布。此外,含溴原水中高浓度的有机物会增加Br-DBPs的生成量。混凝、沉淀、过滤是常规给水处理过程的基本工艺,它能去除水中胶体和悬浮杂质,降低水中DOC的含量。但是经过这些处理后水中Br¯浓度变化较小,出水中Br¯/DOC增大,饮用水中会更易形成Br-DBPs而不是Cl-DBPs[32]。不同地域、季节原水中有机物的含量不同,通常在亚热带、热带气候的南方以及降水量丰富的夏季,有机物浓度会随之升高,但是沿海城市由于枯水期的咸潮入侵现象,大量溴化物进入地表水源,这会导致饮用水中的Br-HANs的含量高于丰水期[53]。
加氯量对含溴原水中DBPs的生成也有显著影响,一般用Br¯/HOCl的形式表示,Br¯/HOCl比例变化会调节DBPs的产物分布,随着Br¯/HOCl的升高会更易形成Br-DBPs,同时减少Cl-DBPs的生成量[54]。
此外,接触消毒剂的时间长短不同,水中Br-DBPs的种类也会随之变化。随接触时间的延长,氯消毒过程会使高分子量Br-DBPs在强氧化剂的作用下被破坏,增加水中低分子量Br-DBPs的积累;而氯胺消毒较为缓和,高分子量Br-DBPs可以在长时间接触氧化剂的情况下在水中积累。
-
目前,饮用水中有50%以上的DBPs仍未被定量检测[1]。由于缺少分析标准品,以及合适的检测分析方法,所以对未知DBPs定量检测非常具有挑战性。
气相色谱(GC)是测定水中挥发性DBPs常用的方法,常用的有液液萃取-气相色谱-电子捕获检测(LLE-GC/ECD)[55-56];液液萃取-气相色谱-质谱法(LLE-GC/MS);顶空-气相色谱-质谱法(HS-GC/MS)等[57-59]。Nikolaou等[58]研究发现LLE-GC/ECD方法对THMs、HANs等DBPs的测定非常灵敏,其次是LLE-GC/MS。但是,GC/MS只能捕获非极性、挥发性或半挥发性化合物,除非使用衍生化作用(如卤代酸的甲基化和卤代醛的五氟苄基羟胺的衍生化作用)[1]。因此GC/MS方法会丢失饮用水中的高极性、亲水性或非挥发性的Br-DBPs。
非目标超高分辨率方法可以准确的测量离子的元素和同位素组成,可以用于鉴定或进一步表征未知Br-DBPs。傅立叶变换离子回旋共振质谱(FT-ICR-MS)作为具有超高分辨率和质量准确度的非目标技术已被研究人员广泛用于对未知DBPs的鉴定[60]。Zhang等[61]利用负离子电喷雾电离(ESI)FT-ICR-MS表征饮用水中的Br-DBPs,共检测到441种一溴产物和37种二溴产物,其中大多数都未曾报道过。Luek等[62]通过该技术以及同位素模拟和MS-MS碎片光谱鉴定并确认了多组水力压裂废水中的卤代有机化合物的分子式。
ESI是一种软电离技术,主要形成分子离子或假分子离子,配合串联质谱形成碎片离子,可以测量高极性和高分子量DBPs(<1000 Da),通常与液相色谱(LC)或超高效液相色谱(UPLC)结合使用[1]。三重四极杆(QQQ)质谱仪在前体离子扫描(PIS)模式下可以鉴定DBPs[40],将PIS设置m/z为79/81,便可以显示Br-DBPs的整个光谱[3]。Pan等[63]利用超高效液相色谱/电喷雾电离三重四极杆质谱(UPLC/ESI-tqMS)鉴定出氯化饮用水中11种新型芳香族Br-DBPs。
-
总有机卤素(TOX)代表水样品中所有氯化,溴化和碘化的有机化合物。在饮用水处理中,TOX通常用于衡量消毒过后的饮用水中总卤代DBPs的含量。据报道,在氯消毒的水中,已知的DBPs仅占TOX的16%–70%[64-66],而在氯胺消毒的水中,只有不到20%的TOX是已知的DBPs。因此,TOX的测量既包括已知的DBPs,也包括尚未识别和表征的大部分DBPs。对TOX的进一步分析可以为水行业和监管机构提供一种量化DBPs中不同卤素含量的方法。
有些研究者将TOX与离子色谱(IC)[67]或电感耦合等离子质谱(ICP-MS)[68]、UPLC/ESI-MS[63]等联合起来,对具有异质性的各种卤化物进行定量分析[69]。例如,总有机溴(TOBr)测定提供了未知Br-DBPs的替代量,因此在估算总Br-DBPs的程度及其风险方面起着重要作用[1, 69]。Kristiana等[70]使用TOX-IC系统对原水预处理后含有中等程度的溴化物(346 μg·L−1)和极低的DOC(0.3 mg·L−1)的水厂水进行检测,测得总溴回收率为89%(消毒后的饮用水中的溴含量/经过预处理后的原水的溴含量)。Tan等[65] 使用TOX-IC系统检测了原水经过处理后的水厂出水(Br¯浓度754 μg·L−1,DOC浓度1.2 mg·L−1;Br¯浓度415 μg·L−1,DOC浓度3.7 mg·L−1),观察到总溴回收率为103%和82%。
-
目前在许多国家的经过氯和氯胺消毒过后的饮用水中均检测到了不同浓度的Br-DBPs,最重要的是,饮用水中的Br-DBPs给人类带来的健康风险远远高于Cl-DBPs,因此,Br-DBPs值得更多的关注与研究。Br¯浓度、pH值、温度、NOM含量以及与氧化剂接触时间都会影响Br-DBPs的生成,其中高浓度的Br¯是后续消毒处理过程生成更多Br-DBPs的关键。但是有关不同影响因素之间的相互作用的相关研究较少。
一些研究人员提出通过去除Br-DBPs的前驱物以及优化消毒方式来减少Br-DBPs的生成。但是有机前驱物的去除过程无法有效去除溴化物,反而会导致溴化物与溶解有机碳(DOC)的比率(Br/DOC)上升,在消毒处理后的水中会优先形成Br-DBPs。因此,溴化物的去除显得尤为必要。通过强化混凝、活性炭吸附、阴离子交换树脂等工艺可以有效去除溴化物,为减少Br-DBPs的生成提供了有效途径。在今后对Br-DBPs生成机理探究的同时,研究者需要对未知的Br-DBPs进行进一步鉴别与量化,并对不同条件下饮用水的DBPs进行综合风险评估,切实保障饮用水安全,为大众身体健康筑下坚实的防线。
水中溴代消毒副产物的生成综述
Brominated disinfection by-products formation in water: A review
-
摘要: 溴代消毒副产物(Br-DBPs)由于其高细胞毒性和基因毒性被广泛关注。本文介绍了饮用水中多种已知Br-DBPs(溴代甲烷、溴代乙酸、溴代乙腈、溴代乙酰胺、溴代硝基甲烷、溴代乙醛、溴代芳香族DBPs)的检出浓度、结构与毒性。综述了常用消毒方式(氯和氯胺消毒)过程Br-DBPs的生成机理,输配过程对Br-DBPs的生成影响。此外,还讨论了未知Br-DBPs的识别进展以及Br¯浓度、pH值、温度、天然有机物等对Br-DBPs生成的影响。由于海水入侵、地质结构变化等原因,水源中的Br¯浓度升高会使Br-DBPs增多,溴化物的去除能够有效控制Br-DBPs。本文为今后对Br-DBPs的种类、检测等研究提供了参考,以更好地评估暴露在饮用水中的Br-DBPs的健康风险。Abstract: Brominated disinfection by-products (Br-DBPs) have attracted increased attention because of their high cytotoxicities and genotoxicities. This review introduced the concentration ranges and toxicities of known Br-DBPs in drinking water including bromo-methane, bromo-acetic acid, bromo-acetonitrile, bromo-acetamide, bromo-nitromethane, bromo-acetaldehyde, and bromo-aromatic DBPs. The mechanisms of Br-DBPs formation in the widely used disinfection processes (chlorination and chloramination), and Br-DBPs formation process in the distribution system were reviewed. In addition, the identification of unknown Br-DBPs and the factors (e.g., Br- concentration, pH, temperature, natural organic matter, etc.) affecting the formation of Br-DBPs were also discussed. Due to seawater invasion and geological structural changes, increasing Br¯ concentration in the water source has led to growing Br-DBPs formation. Thus, Br¯ removal provides an effective way to control Br-DBPs formation. It is expected to provide a summary for the types and detection of Br-DBPs to better assess the health risks of Br-DBPs in drinking water.
-

-
表 1 已知溴代甲烷类消毒副产物种类与检出浓度
Table 1. Known Br-THMs species and their detection levels
化合物
Compound英文名称及缩写
Name and abbreviation结构式
Structure检出浓度/(ng·L−1)
Detection concentration参考文献
References一溴二氯甲烷 Bromodichloromethane
BDCM
12.00—25.00(伊朗)
0.29—13.00(中国)[12]
[13]二溴一氯甲烷 Dibromochloromethane
DBCM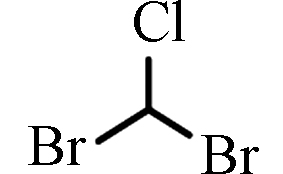
21.10—38.50(伊朗)
1.59—26.80(中国)[12]
[13]三溴甲烷 Bromoform
TBM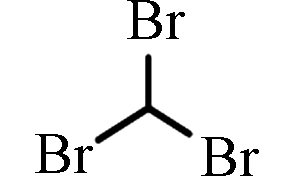
29.50—47.70(伊朗)
ND—7.56(中国)[12]
[13]注:ND 未检出.not detected. 表 2 已知溴代乙酸类消毒副产物种类与检出浓度
Table 2. Known Br-HAAs species and their detection levels
化合物
Compound英文名称及缩写
Name and abbreviation结构式
Structure检出浓度/ (ng·L−1)
Detection concentration参考文献
References溴乙酸 Bromoacetic acid
BAA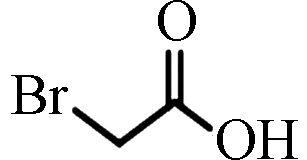
ND(以色列)
0.20—1.63(中国)[7]
[15]二溴乙酸 Dibromoacetic acid
DBAA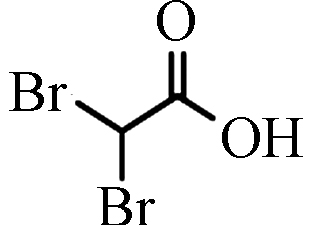
12.00—38.70(以色列)
<0.37—5.0(中国)[7]
[15]溴氯乙酸 Bromochloroacetic acid
BCAA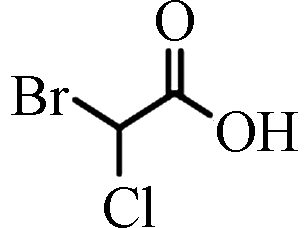
1.00—3.90(以色列)
<0.75— 4.4(中国)[7]
[15]三溴乙酸 Tribromoacetic acid
TBAA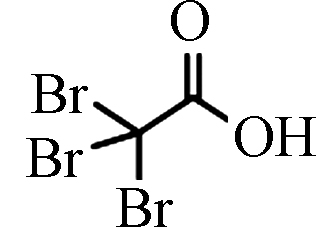
ND(以色列)
0.24—10.70(中国)[7]
[15]二溴一氯乙酸 Dibromochloroacetic acid
DBCAA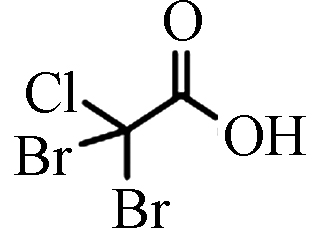
ND(以色列)
<0.95—4.40(中国)[7]
[15]一溴二氯乙酸 Bromodichloroacetic acid
BDCAA
ND(以色列)
0.98—3.80(中国)[7]
[15]注: ND 未检出. not detected. 表 3 已知含氮溴代消毒副产物种类与检出浓度
Table 3. Known nitrogenous brominated DBPs species and their detection levels
化合物
Compound英文名称及缩写
Name and abbreviation结构式
Structure检出浓度/(ng·L−1)
Detection concentration参考文献
References溴乙腈 Bromoacetonitrile
BAN
ND—0.15(中国) [17] 二溴乙腈 Dibromoacetonitrile
DBAN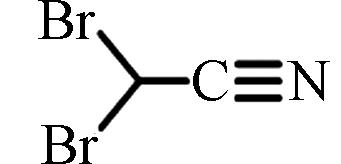
ND—0.87(中国)
ND—1.90(中国)[17]
[18]溴氯乙腈 Bromochloroacetonitrile
BCAN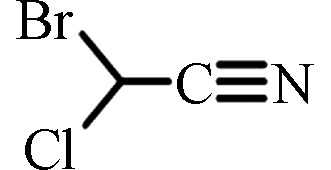
ND(中国)
ND—1.56(中国)[17]
[18]溴乙酰胺 Bromoacetamide
BAM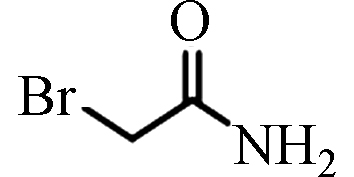
ND—1.92(中国) [20] 二溴乙酰胺 Dibromoacetamide
DBAM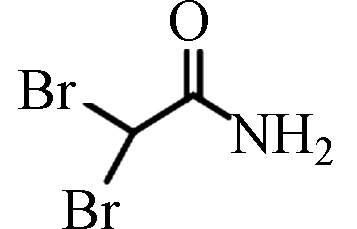
ND—0.76(中国)
0.35—1.25(中国)[20]
[21]溴氯乙酰胺 Bromochloroacetamide
BCAM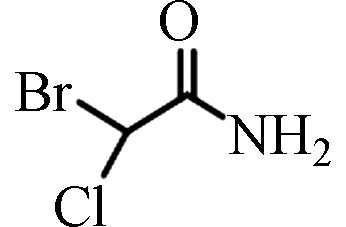
ND—1.34(中国)
0.22—1.32(中国)[20]
[21]三溴乙酰胺 Tribromoacetamide
TBAM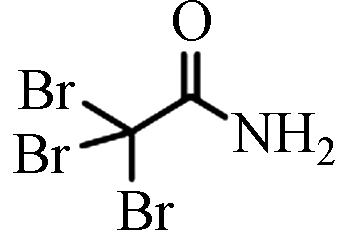
ND—0.15(中国)
ND—0.56(中国)[20]
[21]二溴一氯乙酰胺 Dibromochloroacetamide
DBCAM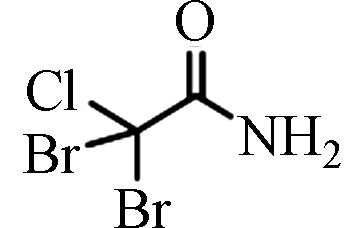
ND—0.22(中国)
0.34—0.50(中国)[20]
[21]一溴二氯乙酰胺 Bromodichloroacetamide
BDCAM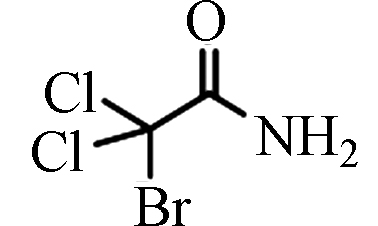
ND—0.80(中国)
0.04—0.25(中国)[20]
[21]溴硝基甲烷 Bromonitromethane
BNM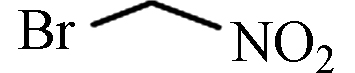
ND—0.30(美国)
ND(中国)[25]
[26]二溴硝基甲烷 Dibromonitromethane
DBNM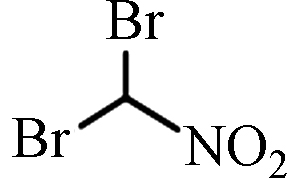
ND—0.50(美国)
ND(中国)[25]
[26]溴氯硝基甲烷 Bromochloronitromethane
BCNM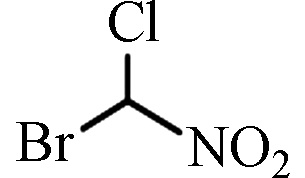
ND—3.00(美国)
ND(中国)[25]
[26]三溴硝基甲烷 Tribromonitromethane
TBNM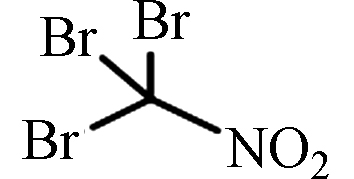
ND—5.00(美国)
ND(中国)[25]
[26]二溴一氯硝基甲烷 Dibromochloronitromethane
DBCNM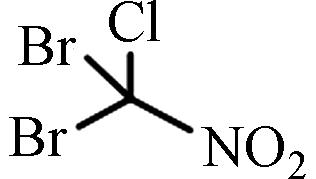
ND—3.00(美国)
ND—0.30(中国)[25]
[26]一氯二溴硝基甲烷 Bromodichloronitromethane
BDCNM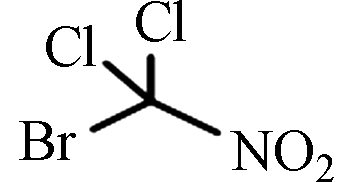
ND—3.00(美国)
0.10—0.90(中国)[25]
[26]2,3,5-三溴吡咯 2,3,5-Tribromopyrrole
2,3,5-TBP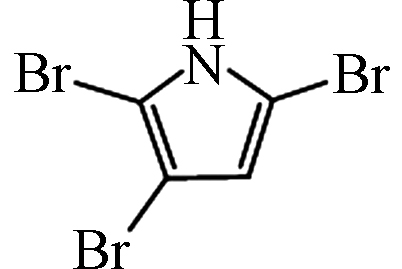
— [27] 四溴吡咯 Tetrabromopyrrole 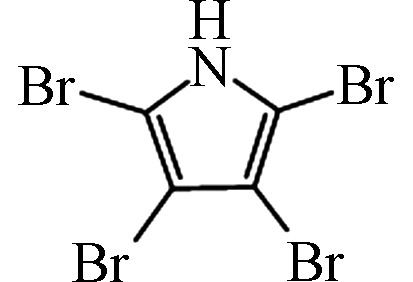
— [27] 三溴氯吡咯 Tribromochloropyrrole 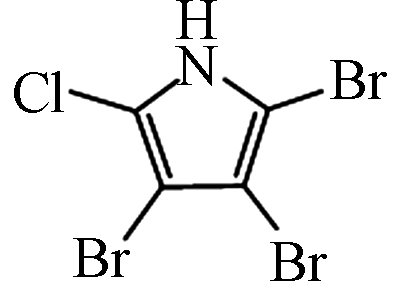
— [27] 三溴碘吡咯 Tribromoiodopyrrole 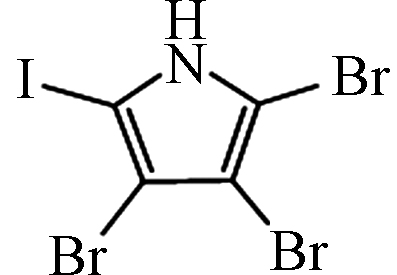
— [27] 注: ND 未检出.not detected; —,暂无相关数据. No data available at this time. 表 4 已知芳香族溴代消毒副产物种类与检出浓度
Table 4. Known aromatic brominated DBPs species and their detection levels
化合物
Compound英文名称及缩写
Name and abbreviation结构式
Structure检出浓度/(ng·L−1)
Detection concentration参考文献
References2,6-二氯-4-溴苯酚 2,6-Dichloro-4-bromophenol 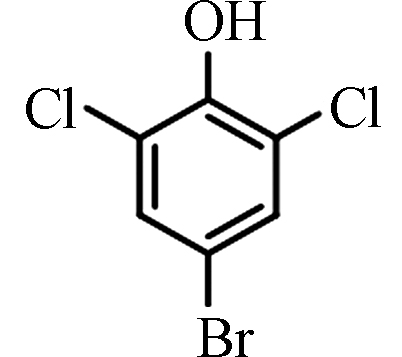
<1.10—72.50 [4] 2,6-二溴-4-氯苯酚 2,6-Dibromo-4-chlorophenol 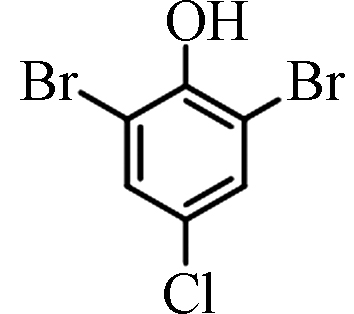
<0.50—12.10 [4] 2,4,6-三溴苯酚 2,4,6-Tribromophenol 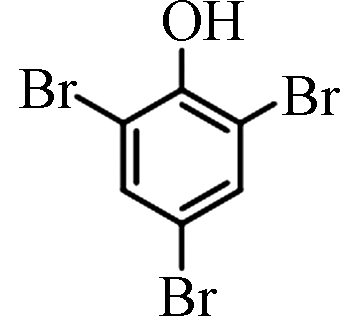
<2.60—56.90 [4] 3-溴-5-氯-4-羟基苯
甲醛3-Bromo-5-chloro-4-hydroxybenzaldehyde 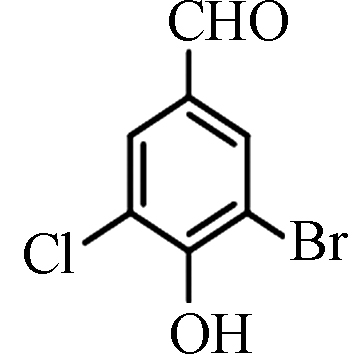
<0.70—61.40 [4] 3,5-二溴-4-羟基苯
甲醛3,5-Dibromo-4-hydroxybenzaldehyde 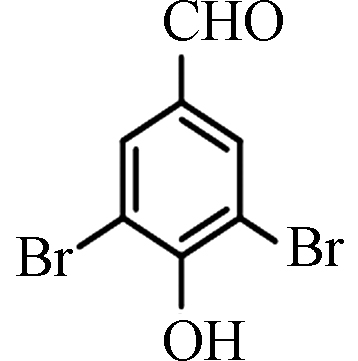
<0.70—43.20 [4] 3-溴-5-氯-4-羟基苯
甲酸3-Bromo-5-chloro-4-hydroxybenzoic acid 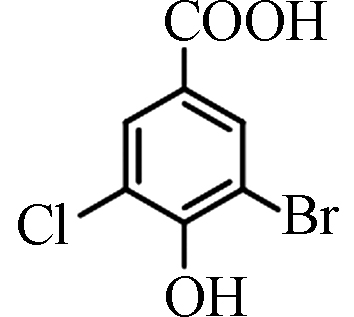
<1.90 [4] 3,5-二溴-4-羟基苯
甲酸3,5-Dibromo-4-hydroxybenzoic acid 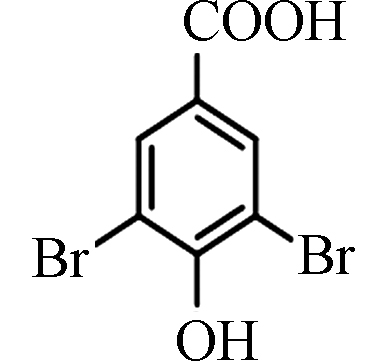
<3.20—12.50 [4] 3-溴-5-氯水杨酸 3-Bromo-5-chlorosalicylic acid 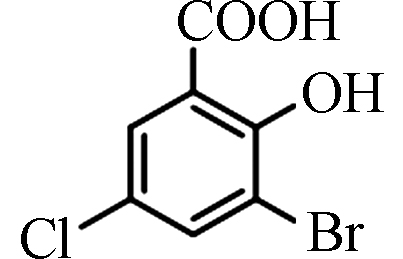
<0.50—35.30 [4] 3,5-二溴水杨酸 3,5-Dibromosalicylic acid 
<0.70—75.90 [4] 表 5 已知溴代乙醛消毒副产物种类与检出浓度
Table 5. Known Br-HALs species and their detection levels
化合物
Compound英文名称及缩写
Name and abbreviation结构式
Structure检出浓度/(ng·L−1)
Detection concentration参考文献
References溴乙醛 Bromoacetaldehyde
BAL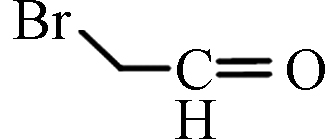
<LOQ—1.50(美国) [29] 二溴乙醛 Dibromoacetaldehyde
DBAL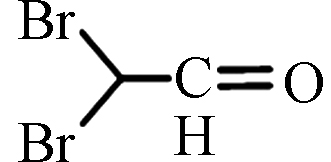
<LOQ—3.20(美国)
ND(加拿大)[29]
[30]溴氯乙醛 Bromochloroacetaldehyde
BCAL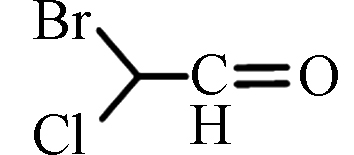
<LOQ—2.30(美国)
0.07—0.23(加拿大)[29]
[30]三溴乙醛 Tribromoacetaldehyde
TBAL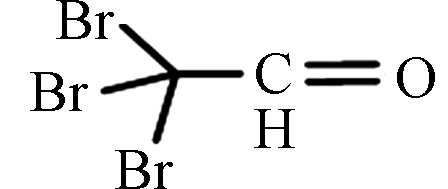
ND—13.12(美国)
ND(加拿大)[29]
[30]二溴一氯乙醛 Dibromochloroacetaldehyde
DBCAL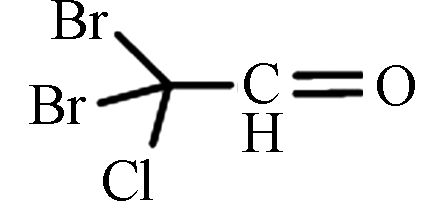
<LOQ—2.90(美国)
0.57—1.75(加拿大)[29]
[30]一溴二氯乙醛 Bromodichloroacetaldehyde
BDCAL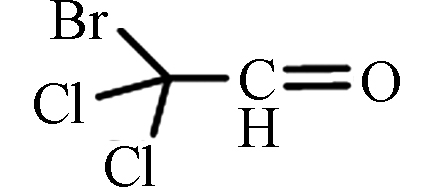
1.00—2.30(美国)
0.75—2.54(加拿大)[29]
[30]注:LOQ 定量极限. limit of quantitation; ND 未检出. not detected 表 6 饮用水氯和氯胺消毒生成溴代消毒副产物的反应方程式
Table 6. Reactions and rate constants in chlorination and chloramination of producing Br-DBPs
消毒类型
Disinfection type反应式
Chemical equation速率常数
Rate constant氯消毒 HOCl + Br¯→HOBr + Cl¯ 7.75 × 102 L·mol−1·s−1 HOBr + NOMfast→Br-DBPs,int 1.36 × 106 L·mol −1·s−1 HOBr + NOMslow→Br-DBPs 6.5 L·mol −1·s−1 HOCl + Br-DBPs,int→Cl-DBPs + Br¯ 6.0 L·mol −1·s−1 氯胺消毒 NH2Cl + H2O→HOCl + NH3 3.0 × 10−5 s−1 HOCl + Br¯→HOBr + Cl¯ 7.75 × 102 L·mol −1·s−1 HOBr + NH3→NH2Br + H2O 5.1 × 106 L·mol −1·s−1 HOBr + NH2Cl→NHBrCl + H2O 2.7 × 105 L·mol −1·s−1 NH2Br + NOMfast→Br-DBPs,int + NH3 1.0 × 102 L·mol −1·s−1 NH2Br + NOMslow→Br-DBPs + NH3 28 L·mol −1·s−1 NHBrCl + NOMfast→Br-DBPs,int + NH2Cl 40 L·mol −1·s−1 NHBrCl + NOMslow→Br-DBPs + NH2Cl 1.6 × 10−2 L·mol −1·s−1 -
[1] DONG H Y, QIANG Z M, RICHARDSON S D. Formation of iodinated disinfection byproducts (I-DBPs) in drinking water: Emerging concerns and current issues [J]. Accounts of Chemical Research, 2019, 52(4): 896-905. doi: 10.1021/acs.accounts.8b00641 [2] DAVIS S N, FABRYKA-MARTIN J T, WOLFSBERG L E. Variations of bromide in potable ground water in the United States [J]. Ground Water, 2010, 42(6): 902-909. [3] PAN Y, ZHANG X R, WAGNER E D, et al. Boiling of simulated tap water: Effect on polar brominated disinfection byproducts, halogen speciation, and cytotoxicity [J]. Environmental Science & Technology, 2014, 48(1): 149-156. [4] PAN Y, WANG Y, LI A M, et al. Detection, formation and occurrence of 13 new polar phenolic chlorinated and brominated disinfection byproducts in drinking water [J]. Water Research, 2017, 112: 129-136. doi: 10.1016/j.watres.2017.01.037 [5] 杨永亮, 刘崴, 刘晓端, 等. 辽宁省西部和沈阳地区河水及地下水中溴的分布与污染特征 [J]. 环境化学, 2009, 28(6): 924-928. doi: 10.3321/j.issn:0254-6108.2009.06.029 YANG Y L, LIU W, LIU X D, et al. Distribution and contamination characteristics of bromine in surface water and ground water from the western Liaoning and Shenyang area [J]. Environmental Chemistry, 2009, 28(6): 924-928(in Chinese). doi: 10.3321/j.issn:0254-6108.2009.06.029
[6] KIDD J, BARRIOS A, APUL O, et al. Removal of bromide from surface water: Comparison between silver-impregnated graphene oxide and silver-impregnated powdered activated carbon [J]. Environmental Engineering Science, 2018, 35(9): 988-995. doi: 10.1089/ees.2017.0485 [7] RICHARDSON S D, THRUSTON A D, RAV-ACHA C, et al. Tribromopyrrole, brominated acids, and other disinfection byproducts produced by disinfection of drinking water rich in bromide [J]. Environmental Science & Technology, 2003, 37(17): 3782-3793. [8] RICHARDSON S D, FASANO F, ELLINGTON J J, et al. Occurrence and mammalian cell toxicity of iodinated disinfection byproducts in drinking water [J]. Environmental Science & Technology, 2008, 42(22): 8330-8338. [9] RICHARDSON S D, PLEWA M J, WAGNER E D, et al. Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research [J]. Mutation Research/Reviews in Mutation Research, 2007, 636(1-3): 178-242. doi: 10.1016/j.mrrev.2007.09.001 [10] CHISHOLM K, COOK A, BOWER C, et al. Risk of birth defects in Australian communities with high levels of brominated disinfection by-products [J]. Environmental Health Perspectives, 2008, 116(9): 1267-1273. doi: 10.1289/ehp.10980 [11] NIEUWENHUIJSEN M J, TOLEDANO M B, EATON N E, et al. Chlorination disinfection byproducts in water and their association with adverse reproductive outcomes: A review [J]. Occupational and Environmental Medicine, 2000, 57(2): 73-85. doi: 10.1136/oem.57.2.73 [12] PLEWA M J, WAGNER E D. Charting a new path to resolve the adverse health effects of DBPs [M]. Washington: American Chemical Society, 2015: 3-23 [13] DOBARADARAN S, SHABANKAREH FARD E, TEKLE-RÖTTERING A, et al. Age-sex specific and cause-specific health risk and burden of disease induced by exposure to trihalomethanes (THMs) and haloacetic acids (HAAs) from drinking water: An assessment in four urban communities of Bushehr Province, Iran, 2017 [J]. Environmental Research, 2020, 182: 109062. doi: 10.1016/j.envres.2019.109062 [14] 刘晓琳, 郑唯韡, 韦霄, 等. 江苏省某水厂含碳、含氮和碘系消毒副产物现况调查 [J]. 中华预防医学杂志, 2012, 46(2): 133-138. doi: 10.3760/cma.j.issn.0253-9624.2012.02.009 LIU X L, ZHENG W W, WEI X, et al. Investigation on the levels of carbon-, nitrogen-, iodine-containing disinfection by-products in a water plant in Jiangsu Province, China [J]. Chinese Journal of Preventive Medicine, 2012, 46(2): 133-138(in Chinese). doi: 10.3760/cma.j.issn.0253-9624.2012.02.009
[15] 王姗姗. 辽宁省六城市出厂水中三卤甲烷类消毒副产物的调查 [C]//第十四届沈阳科学学术年会论文集(理工农医). 中共沈阳市委、沈阳市人民政府: 沈阳市科学技术协会, 2017: 677-681. WANG S S. Investigation of trihalomethanes disinfection by-products in six cities of Liaoning Province [C]// Proceedings of the 14th Shenyang Science Annual Conference (Science, engineering, agriculture and medicine). Shenyang municipal Party committee and Shenyang Municipal People's Government: Shenyang Science and Technology Association, 2017: 677-681 (in Chinese).
[16] 孟丽苹, 董兆敏, 胡建英. 全国自来水厂卤乙酸浓度调查、风险评估与标准建议 [J]. 中国环境科学, 2012, 32(4): 721-726. doi: 10.3969/j.issn.1000-6923.2012.04.023 MENG L P, DONG Z M, HU J Y. National survey and risk assessment of haloacetic acids in drinking water in China for reevaluation of the drinking water standards [J]. China Environmental Science, 2012, 32(4): 721-726(in Chinese). doi: 10.3969/j.issn.1000-6923.2012.04.023
[17] DING H H, MENG L P, ZHANG H F, et al. Occurrence, profiling and prioritization of halogenated disinfection by-products in drinking water of China [J]. Environmental Science. Processes & Impacts, 2013, 15(7): 1424-1429. [18] 董蕾, 王海燕, 蔡宏铨, 等. 我国六城市饮用水中含氮消毒副产物的现状调查 [J]. 环境与健康杂志, 2016, 33(3): 232-235. DONG L, WANG H Y, CAI H Q, et al. Investigation on nitrogenous disinfection by-products in drinking water in six cities, China [J]. Journal of Environment and Health, 2016, 33(3): 232-235(in Chinese).
[19] YU Y, RECKHOW D A. Formation and occurrence of N-chloro-2, 2-dichloroacetamide, a previously overlooked nitrogenous disinfection byproduct in chlorinated drinking waters [J]. Environmental Science & Technology, 2017, 51(3): 1488-1497. [20] CHU W, GAO N, YIN D, et al. Trace determination of 13 haloacetamides in drinking water using liquid chromatography triple quadrupole mass spectrometry with atmospheric pressure chemical ionization [J]. Journal of chromatography A, 2012, 1235: 178-181. doi: 10.1016/j.chroma.2012.02.074 [21] ZHOU R, XU Z, ZHU J, et al. Determination of 10 Haloacetamides in drinking water by gas chromatography with automated solid phase extraction [J]. Journal of Chromatography B, 2020, 1150: 122191. doi: 10.1016/j.jchromb.2020.122191 [22] GLEZER V, HARRIS B, TAL N. Hydrolysis of haloacetonitriles: Linear free energy relationship, kinetics and products [J]. Water Research, 1999, 33(8): 1938-1948. doi: 10.1016/S0043-1354(98)00361-3 [23] 王莹, 陈泽智, 李爱民, 等. 13种新型极性苯酚类氯/溴代消毒副产物的生成机理 [J]. 环境化学, 2017, 36(10): 2089-2099. doi: 10.7524/j.issn.0254-6108.2017021501 WANG Y, CHEN Z Z, LI A M, et al. Formation mechanism of 13 new polar phenolic chlorinated and brominated disinfection byproducts in drinking water [J]. Environmental Chemistry, 2017, 36(10): 2089-2099(in Chinese). doi: 10.7524/j.issn.0254-6108.2017021501
[24] PLEWA M J, WAGNER E D, JAZWIERSKA P, et al. Halonitromethane drinking water disinfection byproducts: chemical characterization and mammalian cell cytotoxicity and genotoxicity [J]. Environmental Science & Technology, 2004, 38(1): 62-68. [25] KRASNER S W, WEINBERG H S, RICHARDSON S D, et al. Occurrence of a new generation of disinfection byproducts [J]. Environmental Science & Technology, 2006, 40(23): 7175-7185. [26] HUANG F, RUAN M, YAN J, et al. An improved method for determining HNMs in drinking water [J]. Water Science and Technology:Water Supply, 2013, 13(5): 1257-1264. doi: 10.2166/ws.2013.135 [27] YANG M T, ZHANG X R. Halopyrroles: A new group of highly toxic disinfection byproducts formed in chlorinated saline wastewater [J]. Environmental Science & Technology, 2014, 48(20): 11846-11852. [28] HUANG Y, LI H, ZHOU Q, et al. New phenolic halogenated disinfection byproducts in simulated chlorinated drinking water: Identification, decomposition, and control by ozone-activated carbon treatment [J]. Water Research, 2018, 146: 298-306. doi: 10.1016/j.watres.2018.09.031 [29] JEONG C H, POSTIGO C, RICHARDSON S D, et al. Occurrence and comparative toxicity of haloacetaldehyde disinfection byproducts in drinking water [J]. Environmental Science & Technology, 2015, 49(23): 13749-13759. [30] GAO J N, PROULX F, RODRIGUEZ M J. Occurrence and spatio-temporal variability of halogenated acetaldehydes in full-scale drinking water systems [J]. The Science of the Total Environment, 2019, 693: 133517. doi: 10.1016/j.scitotenv.2019.07.323 [31] WEINBERG H, KRASNER S, RICHARDSON S, et al. The occurrence of disinfection by-products (DBPs) of health concern in drinking water: Results of a nationwide DBP occurrence study [R]. U. S. EPA, Washington, D. C. , 2002: EPA/600/R02/068. [32] SOHN J, AMY G, YOON Y. Bromide ion incorporation into brominated disinfection by-products [J]. Water, Air, and Soil Pollution, 2006, 174(1/2/3/4): 265-277. [33] ZHANG J Z, YU J W, AN W, et al. Characterization of disinfection byproduct formation potential in 13 source waters in China [J]. Journal of Environmental Sciences (China), 2011, 23(2): 183-188. doi: 10.1016/S1001-0742(10)60440-8 [34] WINID, BOGUMILA. Bromine and water quality - Selected aspects and future perspectives [J]. Applied Geochemistry, 2015, 63: 413-435. doi: 10.1016/j.apgeochem.2015.10.004 [35] CHANG E E, LIN Y P, CHIANG P C. Effects of bromide on the formation of THMs and HAAs [J]. Chemosphere, 2001, 43(8): 1029-1034. doi: 10.1016/S0045-6535(00)00210-1 [36] KOLB C, FRANCIS R A, VANBRIESEN J M. Disinfection byproduct regulatory compliance surrogates and bromide-associated risk [J]. Journal of Environmental Sciences (China), 2017, 58: 191-207. doi: 10.1016/j.jes.2017.05.043 [37] ZHOU X L, ZHENG L L, CHEN S Y, et al. Factors influencing DBPs occurrence in tap water of Jinhua Region in Zhejiang Province, China [J]. Ecotoxicology and Environmental Safety, 2019, 171: 813-822. doi: 10.1016/j.ecoenv.2018.12.106 [38] CRIQUET J, RODRIGUEZ E M, ALLARD S, et al. Reaction of bromine and chlorine with phenolic compounds and natural organic matter extracts: Electrophilic aromatic substitution and oxidation [J]. Water Research, 2015, 85: 476-486. doi: 10.1016/j.watres.2015.08.051 [39] ZHAI H Y, ZHANG X R, ZHU X H, et al. Formation of brominated disinfection byproducts during chloramination of drinking water: New polar species and overall kinetics [J]. Environmental Science & Technology, 2014, 48(5): 2579-2588. [40] ZHAI H Y, ZHANG X R. Formation and decomposition of new and unknown polar brominated disinfection byproducts during chlorination [J]. Environmental Science & Technology, 2011, 45(6): 2194-2201. [41] HASSAN K Z A, BOWER K C, MILLER C M. Iron oxide enhanced chlorine decay and disinfection by-product formation [J]. Journal of Environmental Engineering, 2006, 132(12): 1609-1616. doi: 10.1061/(ASCE)0733-9372(2006)132:12(1609) [42] ROSSMAN L A, BROWN R A, SINGER P C. DBP formation kinetics in a simulated distribution system [J]. Water Research, 2001, 35(14): 3483-3489. doi: 10.1016/S0043-1354(01)00059-8 [43] 樊陈锋, 朱志良, 刘绍刚. 金属离子对在饮用水氯化过程中形成消毒副产物的影响的研究进展 [J]. 化学通报, 2011, 74(7): 612-616. FAN C F, ZHU Z L, LIU S G. Progress of the effect of metal ions on the formation of disinfection by-products during chlorination [J]. Chemistry, 2011, 74(7): 612-616(in Chinese).
[44] HOZALSKI R M, ZHANG L, ARNOLD W A. Reduction of haloacetic acids by Fe0: implications for treatment and fate [J]. Environmental Science & Technology, 2001, 35(11): 2258-2263. [45] YANG X, GUO W, ZHANG X, et al. Formation of disinfection by-products after pre-oxidation with chlorine dioxide or ferrate [J]. Water Research, 2013, 47(15): 5856-5864. doi: 10.1016/j.watres.2013.07.010 [46] ZHA X, MA L, LIU Y. Reductive dehalogenation of brominated disinfection byproducts by iron based bimetallic systems [J]. RSC Advances, 2016, 6(20): 16323-16330. doi: 10.1039/C5RA26882F [47] 刘立超. 金属离子对饮用水氯化消毒副产物影响的研究[D]. 天津: 河北工业大学, 2017. LIU L C. Effect of metal ions on chlorination disinfection by-products of drinking water [D]. Tianjin: Hebei University of Technology, 2017(in Chinese).
[48] 王怡, 塔娜, 安乌云. 饮用水中三卤甲烷的生成机理与影响因素研究进展 [J]. 环境污染与防治, 2020, 42(4): 500-506. WANG Y, TA N, AN W Y. Research progress on the formation mechanism and influencing factors of trihalomethanes in drinking water [J]. Environmental Pollution and Control, 2020, 42(4): 500-506(in Chinese).
[49] 陈梦杰, 张凤娥, 董良飞, 等. 供水管网中氯化消毒副产物健康风险评价 [J]. 常州大学学报(自然科学版), 2016, 28(2): 46-49,87. CHEN M J, ZHANG F E, DONG L F, et al. Health risk assessment of chlorinated disinfection by-products in water distribution system [J]. Journal of ChangZhou University (Natural Science Edition), 2016, 28(2): 46-49,87(in Chinese).
[50] HUNG Y C, WATERS B W, YEMMIREDDY V K, et al. pH effect on the formation of THM and HAA disinfection byproducts and potential control strategies for food processing [J]. Journal of Integrative Agriculture, 2017, 16(12): 2914-2923. doi: 10.1016/S2095-3119(17)61798-2 [51] LIU J Q, LI Y, JIANG J Y, et al. Effects of ascorbate and carbonate on the conversion and developmental toxicity of halogenated disinfection byproducts during boiling of tap water [J]. Chemosphere, 2020, 254: 126890. doi: 10.1016/j.chemosphere.2020.126890 [52] 魏源源, 刘燕, 代瑞华. 饮用水消毒溴代副产物及其健康风险 [J]. 化学通报, 2009, 72(12): 1051-1056. WEI Y Y, LIU Y, DAI R H. Brominated by-products of drinking water disinfection and their health risks [J]. Chemistry Bulletin, 2009, 72(12): 1051-1056(in Chinese).
[53] 朱有长, 刘敬雅, 赵尔格, 等. 饮用水消毒副产物比较分析与健康风险评估 [J]. 净水技术, 2019, 38(5): 45-50. ZHU Y C, LIU J Y, ZHAO E G, et al. Comparative analysis and health risk assessment of disinfection by-products (DBPs) in drinking water [J]. Water Purification Technology, 2019, 38(5): 45-50(in Chinese).
[54] ICHIHASHI K, TERANISHI K, ICHIMURA A. Brominated trihalomethane formation in halogenation of humic acid in the coexistence of hypochlorite and hypobromite ions [J]. Water Research, 1999, 33(2): 477-483. doi: 10.1016/S0043-1354(98)00227-9 [55] SIMPSON K L, HAYES K P. Drinking water disinfection by-products: An Australian perspective [J]. Water Research, 1998, 32(5): 1522-1528. doi: 10.1016/S0043-1354(97)00341-2 [56] NIKOLAOU A D, LEKKAS T D, KOSTOPOULOU M N, et al. Investigation of the behaviour of haloketones in water samples [J]. Chemosphere, 2001, 44(5): 907-912. doi: 10.1016/S0045-6535(00)00536-1 [57] 秦无双. 顶空气相色谱法测定自来水中6种卤代烃类消毒副产物残留 [J]. 分析仪器, 2020(2): 35-39. doi: 10.3969/j.issn.1001-232x.2020.02.008 QIN W S. Determination of six halogenated hydrocarbon disinfection by-products residues in tap water by headspace gas chromatography [J]. Analytical Instrumentation, 2020(2): 35-39(in Chinese). doi: 10.3969/j.issn.1001-232x.2020.02.008
[58] NIKOLAOU A D, LEKKAS T D, GOLFINOPOULOS S K, et al. Application of different analytical methods for determination of volatile chlorination by-products in drinking water [J]. Talanta, 2002, 56(4): 717-726. doi: 10.1016/S0039-9140(01)00613-0 [59] KUIVINEN J, JOHNSSON H. Determination of trihalomethanes and some chlorinated solvents in drinking water by headspace technique with capillary column gas-chromatography [J]. Water Research, 1999, 33(5): 1201-1208. doi: 10.1016/S0043-1354(98)00311-X [60] GONSIOR M, MITCHELMORE C, HEYES A, et al. Bromination of marine dissolved organic matter following full scale electrochemical ballast water disinfection [J]. Environmental Science & Technology, 2015, 49(15): 9048-9055. [61] ZHANG H F, ZHANG Y H, SHI Q, et al. Characterization of unknown brominated disinfection byproducts during chlorination using ultrahigh resolution mass spectrometry [J]. Environmental Science & Technology, 2014, 48(6): 3112-3119. [62] LUEK J L, SCHMITT-KOPPLIN P, MOUSER P J, et al. Halogenated organic compounds identified in hydraulic fracturing wastewaters using ultrahigh resolution mass spectrometry [J]. Environmental Science & Technology, 2017, 51(10): 5377-5385. [63] PAN Y, ZHANG X R. Four groups of new aromatic halogenated disinfection byproducts: Effect of bromide concentration on their formation and speciation in chlorinated drinking water [J]. Environmental Science & Technology, 2013, 47(3): 1265-1273. [64] RICHARDSON S D, KIMURA S Y. Water analysis: Emerging contaminants and current issues [J]. Analytical Chemistry, 2016, 88(1): 546-582. doi: 10.1021/acs.analchem.5b04493 [65] TAN J, ALLARD S, GRUCHLIK Y, et al. Impact of bromide on halogen incorporation into organic moieties in chlorinated drinking water treatment and distribution systems [J]. The Science of the Total Environment, 2016, 541: 1572-1580. doi: 10.1016/j.scitotenv.2015.10.043 [66] PRESSMAN J G, RICHARDSON S D, SPETH T F, et al. Concentration, chlorination, and chemical analysis of drinking water for disinfection byproduct mixtures health effects research: US EPA's four lab study [J]. Environmental Science & Technology, 2010, 44(19): 7184-7192. [67] HUA G H, RECKHOW D A. Determination of TOCl, TOBr and TOI in drinking water by pyrolysis and off-line ion chromatography [J]. Analytical and Bioanalytical Chemistry, 2006, 384(2): 495-504. [68] YANG Y, KOMAKI Y, KIMURA S Y, et al. Toxic impact of bromide and iodide on drinking water disinfected with chlorine or chloramines [J]. Environmental Science & Technology, 2014, 48(20): 12362-12369. [69] KIMURA S Y, CUTHBERTSON A A, BYER J D, et al. The DBP exposome: Development of a new method to simultaneously quantify priority disinfection by-products and comprehensively identify unknowns [J]. Water Research, 2019, 148: 324-333. doi: 10.1016/j.watres.2018.10.057 [70] KRISTIANA I, MCDONALD S, TAN J, et al. Analysis of halogen-specific TOX revisited: Method improvement and application [J]. Talanta, 2015, 139: 104-110. doi: 10.1016/j.talanta.2015.02.029 -



 下载:
下载: