-
抗生素是一类常用药品,其中氟喹诺酮(FQs)类药物是抗生素的主要代表品种之一,其广泛应用于医疗、畜牧和水产养殖业等[1-4]. 但由于抗生素的滥用和不当处置导致FQs等药物污染水体[5-6];据国内外报道,诸多国家的江、河、湖泊等地表水中均检测发现FQs[7-9]. 在众多水处理工艺中,相比于混凝、光催化及高级氧化等[10-12],吸附法具有操作简单且处理效果佳等优点[13-14]. 近年来,天然高分子类吸附剂由于其价格低廉且环境友好等重要特点而被广泛关注[15]. 单宁酸(TA)是自然界中仅次于纤维素和木质素的第三大天然高分子材料[16-17],存在于柿子皮、葡萄皮及杨梅等植物中,来源广泛,成本低廉,易提取且生物相容性好。此外,TA含有丰富的酚羟基及羧基等含氧官能团,可与多种重金属及有机污染物等通过鳌合、静电吸引、氢键等相互作用[18-19]. 然而,TA在去除FQs污染物中的研究还鲜有报道,但FQs与TA在分子结构上均含有芳香环及羟基等官能团,它们应能以多种相互作用方式有效结合[16-20]. 因此TA被认为是一种潜在可高效去除FQs的绿色吸附剂材料. 然而TA易溶于水,一般不能直接作为吸附剂使用[20]. 由于TA可与多种金属离子发生鳌合作用,如:铁(Fe)、锆(Zr)、钛(Ti)、铜(Cu)、钼(Mo)、钌(Ru)、钆(Gd)等,可有效提高其成形性[21-23],且这种鳌合作用反应条件温和,反应效率高且操作简单[24]. 其中钛离子为+4价,具有较高电荷,除可有效鳌合固定TA还可与水体中多种有机污染物作用,具有潜在的协同吸附作用,且金属钛生物毒性低环境友好[25]. 开发基于天然高分子单宁酸复合钛盐的绿色吸附剂材料具有重要的现实意义,然而至今上述相关材料的开发及应用机制研究还很不充分.
本文采用四氯化钛为改性剂,通过调节钛盐用量,一步法制备系列单宁酸-钛(TA-Ti)复合吸附剂材料. 应用傅里叶红外光谱(FTIR)、X射线光电子能谱(XPS)、环境扫描电子显微镜(ESEM)、能量色散X射线能谱(EDS)、X射线衍射(XRD)及Zeta电位等手段考察复合吸附剂的分子结构及表面微观形貌. 以一种常见的FQs氧氟沙星(OFL)为代表,系统考察TA-Ti对OFL的吸附性能,详细研究溶液pH、污染物浓度、吸附时间及共存无机盐与天然有机物等对其吸附性能影响。对具有与OFL相似结构的FQs环丙沙星(CIP)和恩诺沙星(ENR)及其两种亚结构类似物氟甲喹(FLU)和1-(2-氟苯基)哌嗪(FPP)的吸附效果进行对比,结合吸附前后吸附剂FTIR和XPS的变化,以及吸附动力学与等温吸附模型分析,深入探讨TA-Ti对OFL的吸附机制.
-
TA (A.R.)、四氯化钛(A.R.)、3-吗啉丙磺酸(MOPS, A.R.)、OFL(>98%)、CIP (>98%)、ENR(>98%)、FLU(>98%)及腐殖酸(HA, > 90%)均购自美国阿拉丁有限公司;FPP(>98%)购自梯希爱(上海)化成工业发展有限公司;乙腈(HPLC级)与乙酸(HPLC级)购自上海麦克林生化科技有限公司;氢氧化钠(A.R.)、盐酸(A.R.)、氯化钠(A.R.)等其它药剂均购于国药集团化学试剂有限公司.
-
TA-Ti复合吸附剂采用一步法合成.在室温条件下,将100 mL浓度为 0.1 mol·L−1 的TA加入50 mL MOPS缓冲液中,通过磁力搅拌混合均匀,再分别加入0.01 mol、0.02 mol及0.03 mol 四氯化钛,反应1.0 min后过滤洗涤,冷冻干燥48 h得到相应的粉末状吸附剂,按照其钛盐投加量分别命名为TA-Ti1,TA-Ti2及TA-Ti3.
-
分别采用FTIR(Thermo Fisher Scientific,美国NICOLET公司)、XPS(PHI 5000 Versaprobe III,日本UlVAC-PHI公司)及XRD(D8 ADVANCE型X射线衍射仪,德国Bruker公司)表征样品分子结构,FTIR波数扫描范围为400—4000 cm−1;XPS扫描范围为0—1100 eV;XRD2θ扫描范围为10°—60°,Cu靶(λ =0.15428 nm). 应用配有EDS的ESEM(SSX-550,日本Shimadzu公司)直接观察样品的表面微观形貌及元素分布,加速电压为25.0 kV;分别将5.0 mg的TA和TA-Ti样品溶解或分散在pH 3—10的水溶液中,采用Zeta电位仪(Nano-Z,英国马尔文公司)检测TA及吸附剂表面电荷性质.
-
本文分析了溶液pH、污染物浓度(等温吸附)、吸附时间(吸附动力学)、共存无机盐(NaCl)和天然有机物(HA)对TA-Ti吸附性能的影响,还考察了TA-Ti1对其他两种FQs(CIP与ENR)及其亚结构类似物(FLU和FPP)的吸附效果,具体的实验条件列于表1中,其中未明确备注的FQs为OFL,直接使用粉末状TA-Ti进行吸附实验.
-
采用紫外吸收光谱仪(UV-Vis 8453,美国安捷伦公司)检测FQs的特征吸收峰. 吸附前后的FQs溶液浓度采用安捷伦HPLC 1200液相色谱进行检测,色谱柱型号为Agilent ® ZorbaTM C18(4.6 mm × 250 mm, 5 μm),检测器为二极管阵列检测器(DAD). 色谱条件如下:等度洗脱流速为1.0 mL·min−1,进样前使用0.45 μm滤膜过滤,进样体积为10 μL,柱温为298 K. 各种FQs的分子结构及具体HPLC分析条件列于表2中.
通过计算吸附前后污染物浓度的变化,计算平衡吸附容量(qe, mmol·g−1),如式(1)所示:
其中,C0和Ce分别为吸附前和吸附平衡时溶液的污染物浓度(mmol·L−1);V为溶液的体积(L);m为吸附剂的质量(g).
-
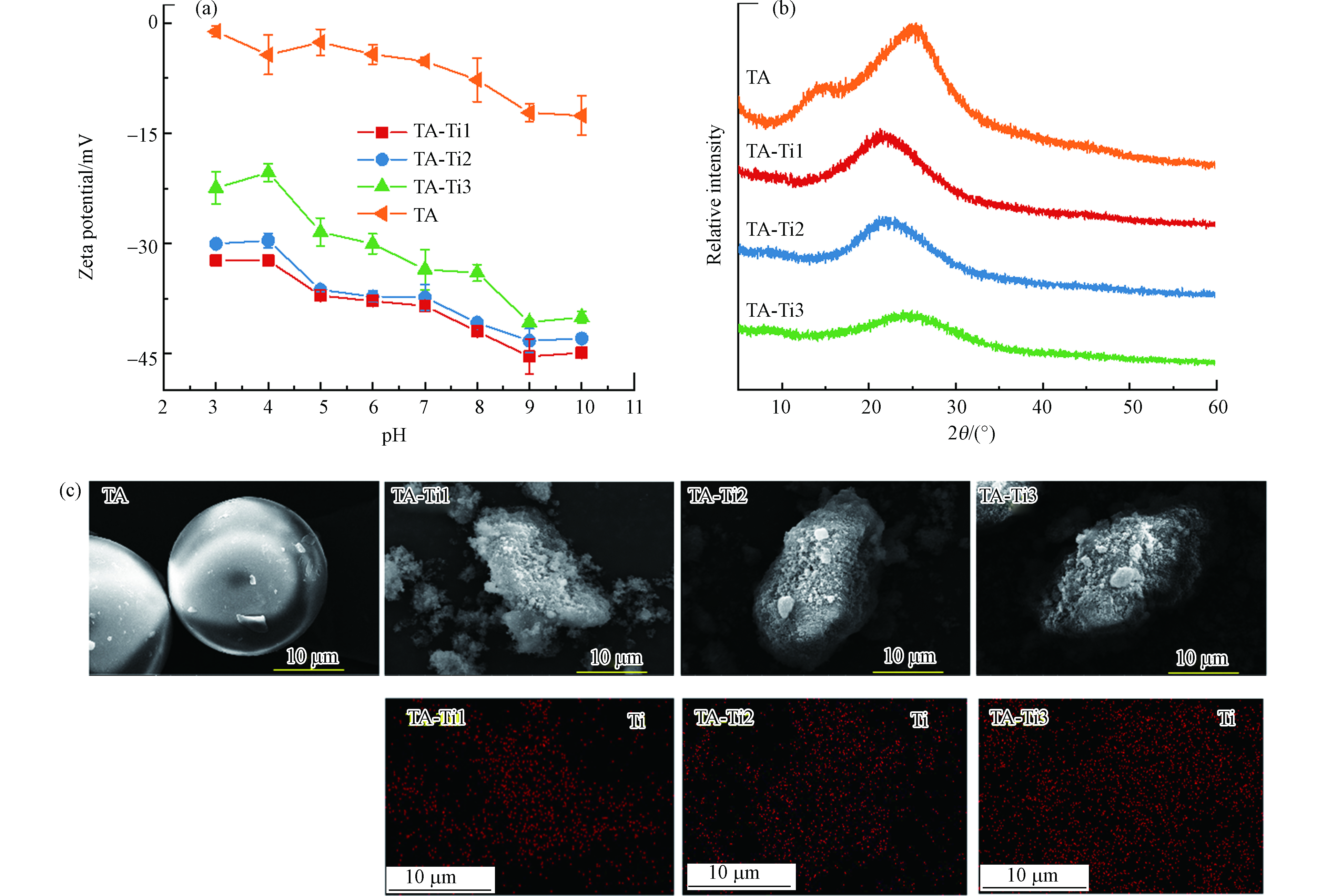
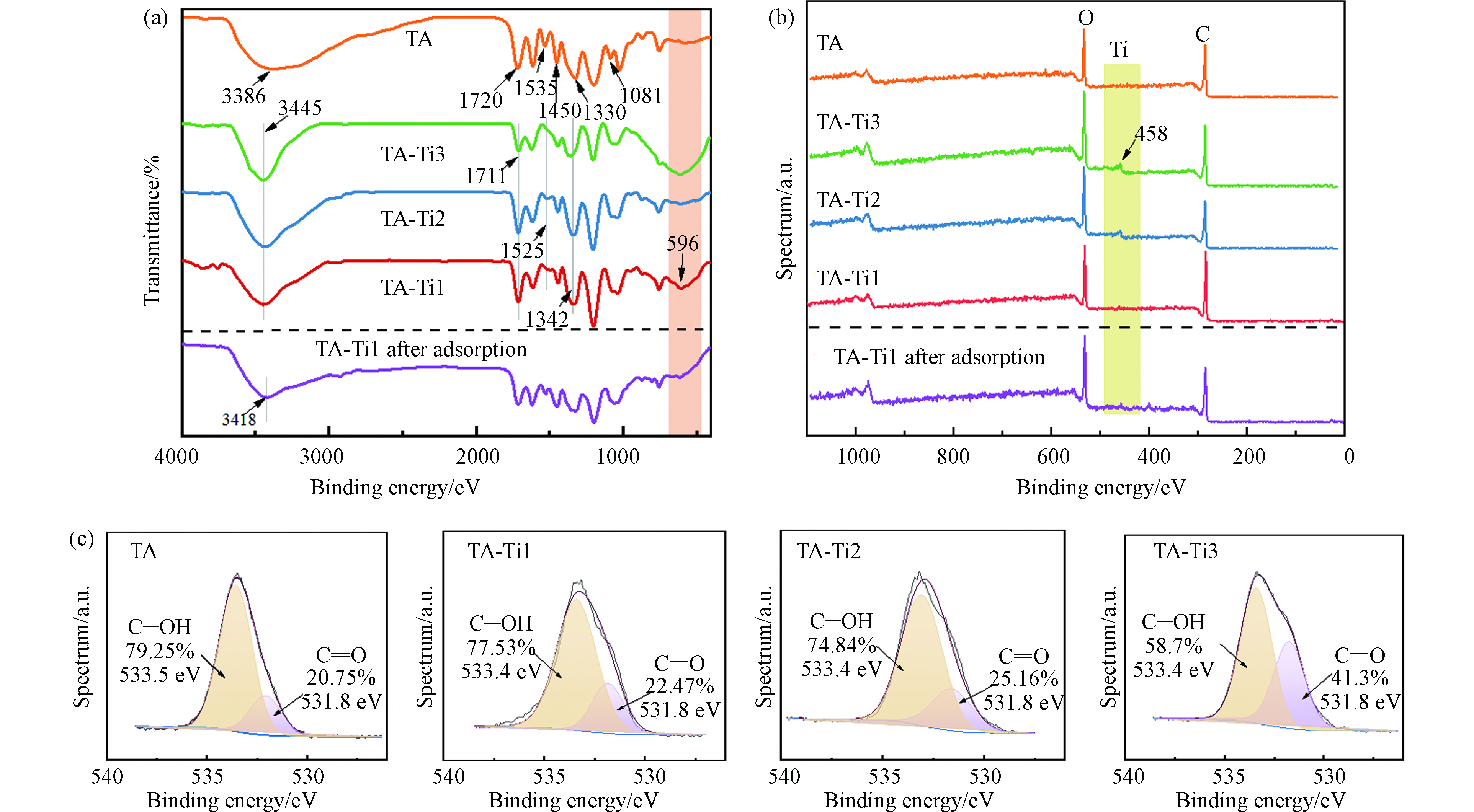
分别采用FTIR、XPS及XRD表征TA-Ti复合吸附剂分子结构,ESEM、EDS及Zeta电位考察其表面微观形貌和电荷性质,结果见图1. 根据图1a,TA在3386 cm−1处有较宽的羟基伸缩振动峰,1720 cm−1处特征峰对应TA分子中羰基的伸缩振动峰[28],1535 cm−1、1450 cm−1、1330 cm−1及1081 cm−1处特征峰为TA酚羟基的弯曲振动峰[29]. TA与Ti4+复合后,在3386 cm−1处的羟基特征峰峰宽明显变窄且向3445 cm−1处发生蓝移,说明羟基参与了复合反应;而 C=O伸缩振动峰从1720 cm−1向1711 cm−1处红移,这可能是由于反应过程中形成了C=O∙∙∙Ti配位结构[28];1535 cm−1处特征峰红移至1525 cm−1,单宁酸1330 cm−1处的酚羟基特征峰发生蓝移至1342 cm−1,这可能是反应中形成了C—O∙∙∙Ti配位结构[28,30],在596 cm−1处出现O—Ti的特征峰进一步表明 Ti4+通过与TA上多种含氧功能基团发生鳌合作用成功制得复合吸附剂材料[31].
TA中含有丰富的酚羟基,具有较强的氢键作用,可采用式(2)[32]对复合反应前后的氢键键能进行估算,TA的氢键键能为19.50 kJ·mol−1,与Ti4+反应后氢键键能降低为14.69 kJ·mol−1,进一步说明在与钛盐反应过程中,消耗了TA的-OH,削弱了TA分子间氢键作用[33].
其中,V0为自由−OH的标准振动频率(3650 cm−1)[32],V为实际红外谱图中对应−OH的伸缩振动频率,K=1.6×10−2 kcal−1.
图1b是不同TA材料的XPS谱图,TA与Ti4+反应后,XPS谱图在458 eV处出现了Ti特征峰并且其峰强随Ti投加量增加而增强,表明Ti4+成功与单宁酸复合. 分析不同TA-Ti样品的O1s XPS特征谱图(图1c),在533.5 eV与531.8 eV处分别为C—OH和C=O的特征峰[34-35];根据图1c,TA和Ti4+复合后,C—OH的结合能略有下降;此外,随着Ti4+的投加量增加,C—OH特征峰面积逐渐减小,且C—OH/C=O特征峰面积比也逐渐下降,说明Ti4+的投加会增加对TA上含氧官能团的消耗,特别是酚羟基,Ti4+与酚羟基等发生鳌合实现对TA的交联作用.
测定TA及吸附剂水溶液在不同pH条件下的Zeta电位,如图2a所示,TA和TA-Ti在所检测的pH范围内表面均带有负电荷,且随着pH的增加表面负电性增强,这是由于TA和吸附剂表面酸性含氧功能基团与溶液中OH−作用发生去质子化作用而造成的;TA与Ti4+反应后,TA-Ti的Zeta电位相比TA大幅降低,这是由于TA上酚羟基等含氧功能基团与Ti4+鳌合后进一步促进质子离解所引起的[28,36];不同TA-Ti吸附剂之间,随着钛盐投加量的增加,在相同pH条件下,TA-Ti 的Zeta电位大致平行上升,这是由于在TA-Ti4+的鳌合网络中,引入较多带正电荷的金属离子,导致了表面电荷升高. 根据图2b,不同TA-Ti吸附剂XRD谱图中均在2θ为22°—25°处出现宽峰,说明复合吸附剂主要为无定型非晶结构[29-30]. 然而随着Ti量的增多,TA-Ti在22°—25°间的宽峰逐渐向高角度移动,根据布拉格公式,衍射角θ增大,晶格间距d变小,这可能是由于随着Ti负载量的增多,其作为交联剂使得TA-Ti交联密度增大,导致复合吸附剂微观局部区域趋于有序,且该有序结构间距随交联密度增大而变小。从图2c的ESEM图可以看出,TA微球直径约为20 μm,复合后TA-Ti转变为直径在10 μm左右的不规则微球,且材料表面变得更为粗糙,有利于与溶液中吸附质结合进一步提高其吸附量. 同时通过EDS对TA-Ti表面Ti元素进行分析,根据图2c,随着Ti4+投加量的增加,TA-Ti表面Ti元素分布逐渐趋于均匀且Ti元素含量增加,这与XPS结果一致(图1c)。
-
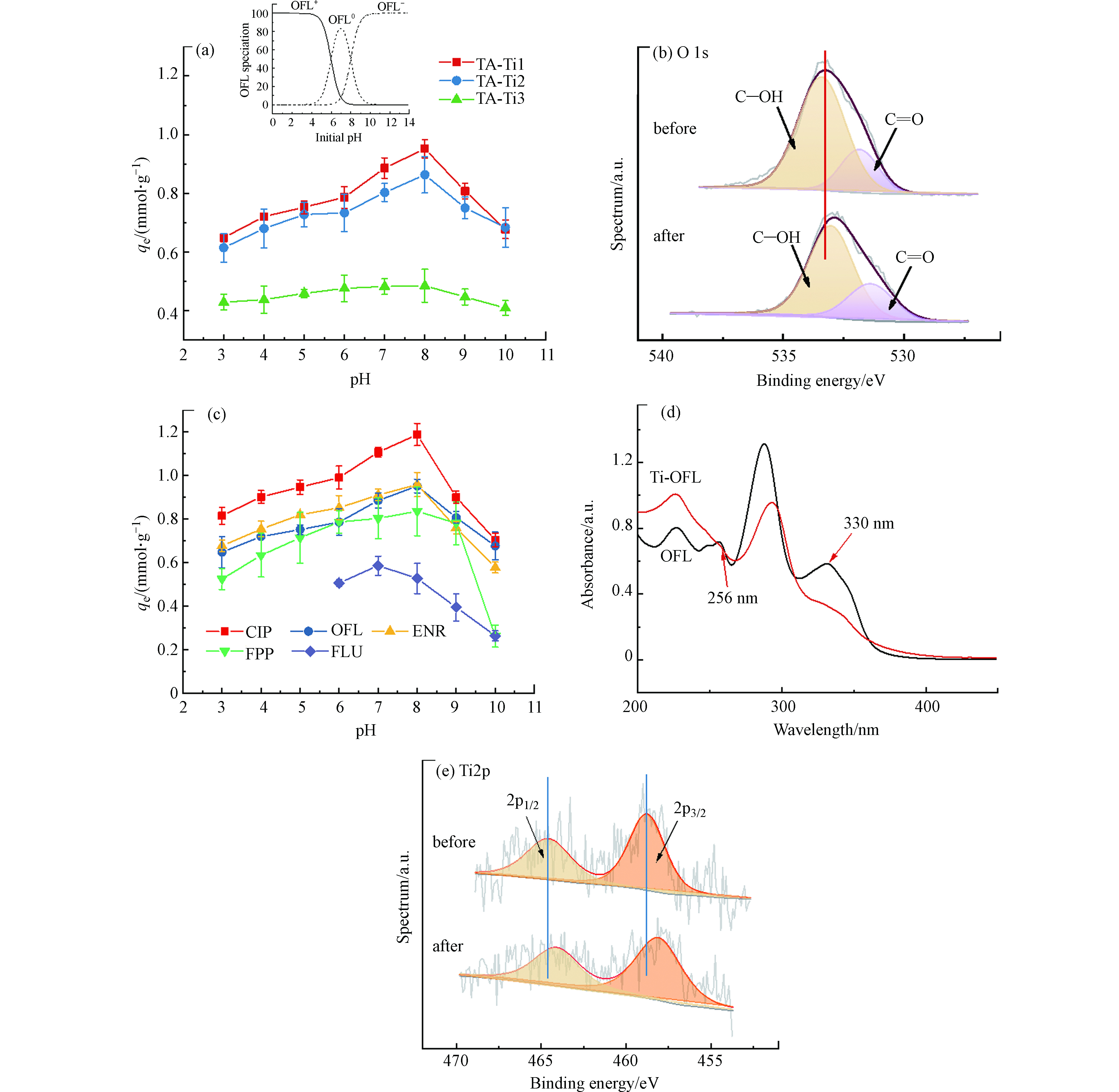
溶液 pH 值可改变溶液中吸附质和吸附剂表面电荷性质,特别是其含有的可离子化官能团,从而显著影响吸附过程[13-14]. 从图3a可以看出,不同TA-Ti吸附剂对OFL的吸附均表现出相似的pH依赖性,说明其应依从着相似的吸附机制. 此外,根据图3a,不同TA-Ti吸附剂对OFL的吸附效果为TA-Ti1 > TA-Ti2 > TA-Ti3,这可能是由于在TA-Ti鳌合网络结构中,Ti4+用量增加导致消耗TA中酚羟基等吸附活性位点增多,使得TA-Ti的表面负电荷减少,Zeta电位升高(图2a),可能存在的与OFL静电吸引及氢键等相互作用降低,导致吸附效果下降.然而,在TA-Ti制备过程中进一步降低TiCl4用量,TA成形性下降,因此TA-Ti1在该研究范围内具有最佳吸附效果.
图3a显示TA-Ti对OFL吸附量随着pH的升高先增大后减小,在pH为8.0时吸附量最大,这是由于不同pH条件下,TA-Ti和OFL具有不同的溶液结构形态导致两者间在不同pH时存在不同的作用方式而影响其吸附行为. 根据OFL分子结构特征(表2),其在溶液中可能存在有3种形态(OFL+、OFL0及OFL−),不同pH下OFL不同形态分布如图3a插图所示.在pH<8.0时,OFL以OFL+或OFL0的结构形态存在,带负电荷的TA-Ti吸附剂可与OFL上质子化的胺基(—NH+)发生静电吸引作用(式3),由于TA-Ti随着pH的升高酚羟基去质子化作用增强其表面负电性进一步提高,其吸附量增加;此外,较强的静电吸附还有利于缩短TA与OFL分子间距离,促进两者间π—π相互作用(式4)及氢键作用,从而进一步提高其吸附量. 图1a是TA-Ti1吸附前后的红外光谱图,吸附后3445 cm−1处—OH特征峰红移至3418 cm−1,根据式2,吸附后氢键键能得以增强[33],说明该吸附过程中TA与OFL间氢键作用显著提高;此外,O1sXPS中C—OH和C=O特征峰结合能及峰强在吸附前后均有明显变化(图3b),表明TA含氧基团可通过氢键作用吸附OFL(式5—6).
然而在pH较低(pH < 4.0)时,大量质子的存在与OFL发生竞争吸附导致其吸附量下降,因此TA-Ti在pH为8.0时吸附量达到最大.在pH>8.0时,OFL羧酸基团去质子化(—COO−),OFL主要以阴离子形态(OFL−)存在,与同样带负电荷的TA-Ti产生了较强的静电排斥作用,吸附效果下降. 然而即使在pH较高(pH > 9.0)时,尽管OFL主要以负离子OFL−形式存在,且由于较强的静电排斥作用使得TA与OFL分子间很难接近,此时两者间π—π相互作用(式4)及普通氢键作用(式5—6)被显著削弱,但pH为10.0时,TA-Ti对OFL仍然有较高的吸附量(0.4—0.6 mmol·L−1)(图3a),这可能是由于在碱性条件下,除了普通氢键作用外,负电荷辅助氢键 (NCAHB)促进了吸附作用. NCAHB通常发生在氢键供体和受体具有相似电离常数时(ΔpKa = pKa,氢键供体 - pKa,氢键受体≤ ~4.00) [14,37]. 在本体系中,ΔpKa, TA-Ti1-COO−…HOOC-FQs小于4.00,表明NCAHB可发生并促进TA-Ti对OFL的吸附(式7—8).
进一步探究TA-Ti对FQs的吸附机理,选取与OFL结构相似的CIP及ENR以及其两种FQs亚结构类似物FPP和FLU进行吸附对比研究(图3c). 从图3c可以看出,TA-Ti对3种FQs均具有良好的吸附效果,证明该复合吸附剂对FQs具有一定的普适性. 另一方面,由于其三者分子结构相似(表2),其吸附量依从着相似的pH依赖性,均在pH为8.0时具有最大吸附量. 此外,三者吸附量大小顺序为CIP > ENR ≈ OFL,这一差别主要是由于CIP分子结构中哌嗪环上为仲胺基,而ENR和OFL哌嗪环上连接的胺基为叔胺基,仲胺基碱性及获得质子的能力均高于叔胺基[38],所以CIP与ENR和OFL相比,与TA-Ti具有更强的静电吸引作用,促进了TA-Ti对CIP的吸附作用,提高了其吸附量. 从两种与FQs亚结构类似物FPP和FLU的吸附效果看(图3c),FPP分子中存在胺基(类似FQs左半边结构,表2)能与TA-Ti产生静电吸引作用,然而由于不存在羧基,FPP与TA-Ti间不会发生NCAHB作用,因此在强碱条件下吸附量迅速降低;而FLU分子中含有羧酸基团(类似FQs右半边结构,表2),pH > 5.0时(FLU在pH < 5.0时难溶于水),FLU呈中性或负电性,与同样带负电的TA-Ti易产生静电排斥作用,因此在近中性及碱性条件下可分别通过普通氢键及NCAHB作用促进吸附,但由于氢键作用弱于静电吸引作用,因此TA-Ti对FLU吸附效果要低于其对FPP的吸附效果.
此外,TA-Ti中Ti4+也可能参与对FQs的吸附,其外层空轨道3d 4s杂化后配位数为6[39],Ti4+可与OFL中含有孤对电子的酚羟基、胺基、羰基等基团发生配位作用[40-41],从而促进其对FQs的吸附. 图3d是OFL与Ti4+反应前后的紫外光谱图对比,反应前OFL在285 nm处出现最大吸收峰,对应于α,β-不饱和羰基的紫外吸收[42],在330 nm左右的弱吸收峰是OFL分子结构中苯环与α,β-不饱和羰基中的双键共轭所引起的弱紫外吸收[43],而波长在256 nm左右的吸收峰为羧酸酯羰基所引起的紫外吸收[42];加入Ti4+后,OFL的紫外吸收峰强度均有一定程度的下降,在285 nm处的最大吸收峰偏移至293 nm,在330 nm处和256 nm处的吸收峰基本消失,说明OFL上羧基和羰基可与Ti4+发生配位作用[41,43-44]. 另外,图3e是TA-Ti1吸附OFL前后的XPS Ti2p谱图,在吸附OFL后,Ti2p特征峰向低结合能位置移动,进一步说明Ti4+作为电子受体可与OFL发生配位作用[45].因此,在该体系中Ti4+不仅作为交联剂固定成形TA,还可以通过配位作用促进该复合吸附剂对FQs的吸附.
-
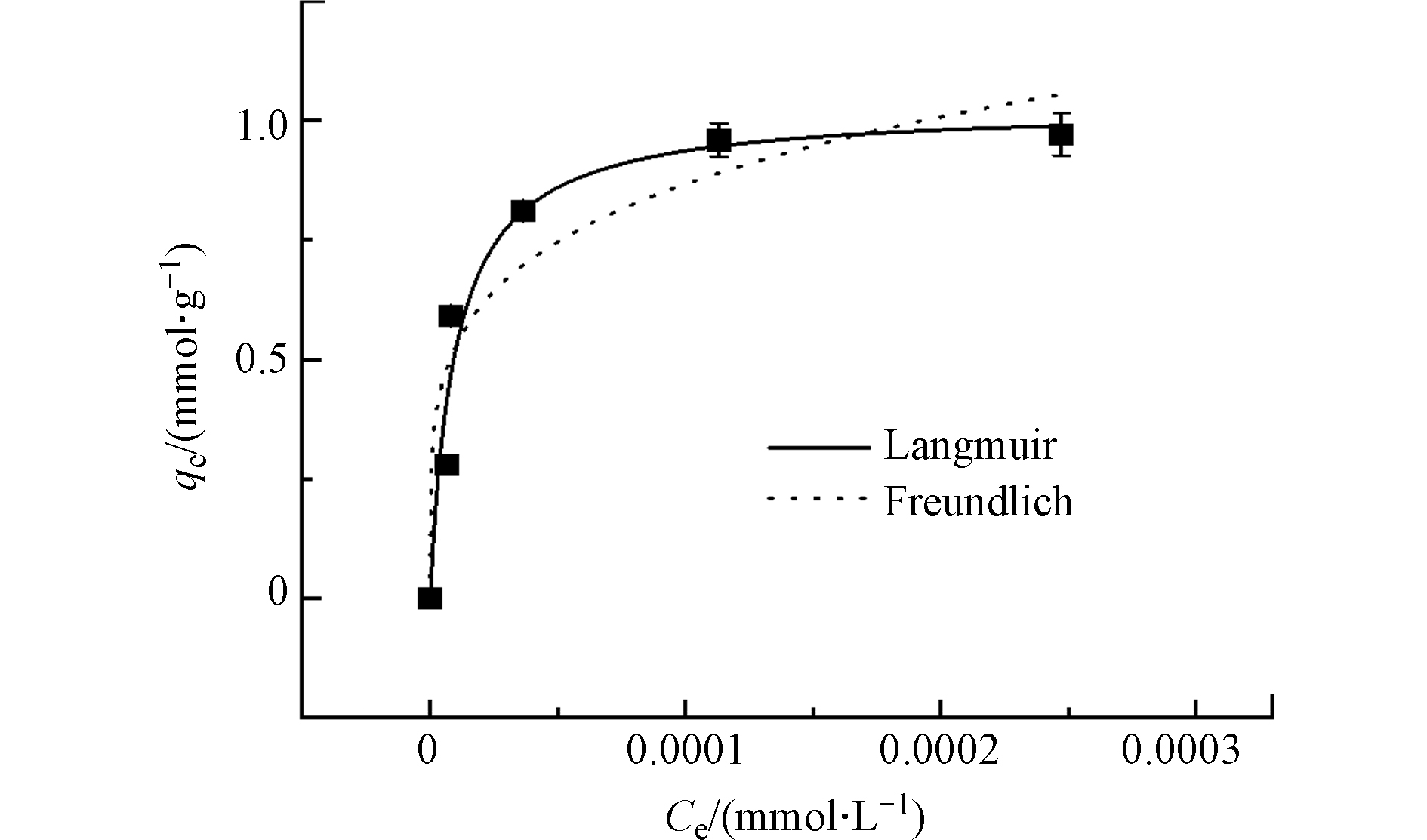
除了溶液pH影响,以吸附性能最佳的TA-Ti1为例,在温度298 K及最佳溶液pH条件下(pH = 8.0),进一步考察其等温吸附行为. 图4是TA-Ti1对不同浓度OFL的吸附效果,随着OFL初始浓度的增加,吸附剂对OFL的吸附量不断增加并逐渐趋于平衡,进一步基于Langmuir[46]和Freundlich[47]模型对吸附等温线进行拟合. Langmuir模型描述的是表面吸附位点分布均匀的单分子层吸附过程[46],而Freundlich模型主要应用于描述非均匀表面的非理想吸附以及多层吸附,该模型认为理论上吸附量可达到无穷大[47],Langmuir和Freundlich模型分别如式(9)及式(10)所示:
其中,qe为平衡时OFL的吸附容量(mmol·g−1),Ce为平衡时溶液中OFL的浓度(mmol·L−1),qm为Langmuir模型下的理论饱和吸附量(mmol·g−1),b为Langmuir等温吸附常数(L·mol−1);Kf是Freundlich等温吸附常数,1/n是异性因子.
拟合结果列于表3中,Langmuir模型拟合结果的相关系数R2为0.958,高于Freundlich模型的相关系数,且获得的对OFL理论最大吸附量为1.028 mmol·g−1 (371.81 mg·g−1),与实验结果0.971 mmol·g−1(351.19 mg·g−1)非常接近;此外,根据前文讨论,TA-Ti主要通过静电吸附、氢键及鳌合作用等与OFL发生吸附,以单分子层吸附为主,进一步证实TA-Ti1对OFL吸附等温线依从Langmuir模型.
-
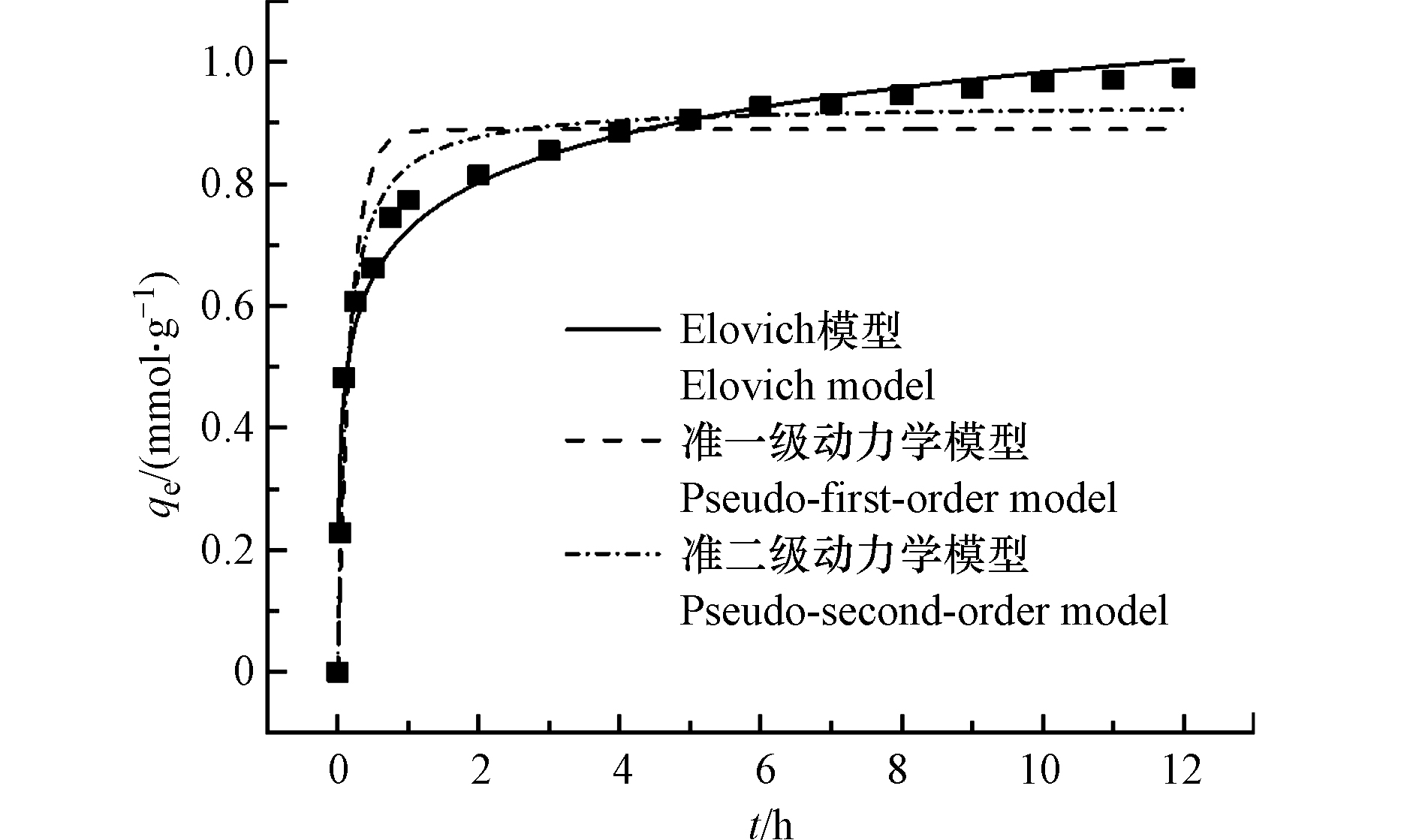
图5描述了TA-Ti1对OFL的吸附动力过程,图5表明在吸附起始1.0 h内TA-Ti1对OFL的吸附量迅速增加,达到吸附总量的75%左右,此后随吸附时间增加,吸附量缓慢增加并逐渐趋于平衡.实验数据分别采用准一级动力学[48],准二级动力学[49],Elovich模型[50]和颗粒内扩散模型[51]进行拟合.准一级动力学模型假设吸附受扩散步骤控制,吸附速率随吸附量的增加线性减小,其表达式为式(11);准二级动力学模型假设吸附速率由吸附剂表面未被占有的吸附空位数目的平方决定,吸附过程受化学吸附机制控制, 如式(12)所示;Elovich 模型适用于描述包括一系列反应机制的动力学过程,如式(13)所示;颗粒内扩散模型主要用于描述物质在颗粒内部扩散的动力学过程[51],其表达式为式(14):
其中,qe和qt (mmol·g−1)分别是在达到吸附平衡后和时间t时的吸附量,k1(h−1)是准一级吸附速率常数;k2 (g·(mmol∙h)−1)是准二级吸附速率常数;AE和BE为Elovich常数;kp(mmol·(g∙h0.5)−1)是内扩散速率常数,C (mmol·g−1)是常数.
吸附动力学拟合结果列于表3,结果表明TA-Ti1对OFL的实际吸附量更接近于根据Elovich模型得到的理论吸附容量1.003 mmol·g−1(362.45 mg·g−1),且其R2更为接近1.0,说明该吸附过程依从多种吸附机制[52]. 颗粒内扩散模型的线性拟合结果也具有较高的R2,说明吸附过程可主要分为三部分:外部扩散,内部扩散及OFL迁移至吸附剂内部活性位点过程,但通过颗粒内扩散模型拟合得到的Cs值并不等于零,进一步说明TA-Ti吸附OFL受到多种机制的影响[53].
-
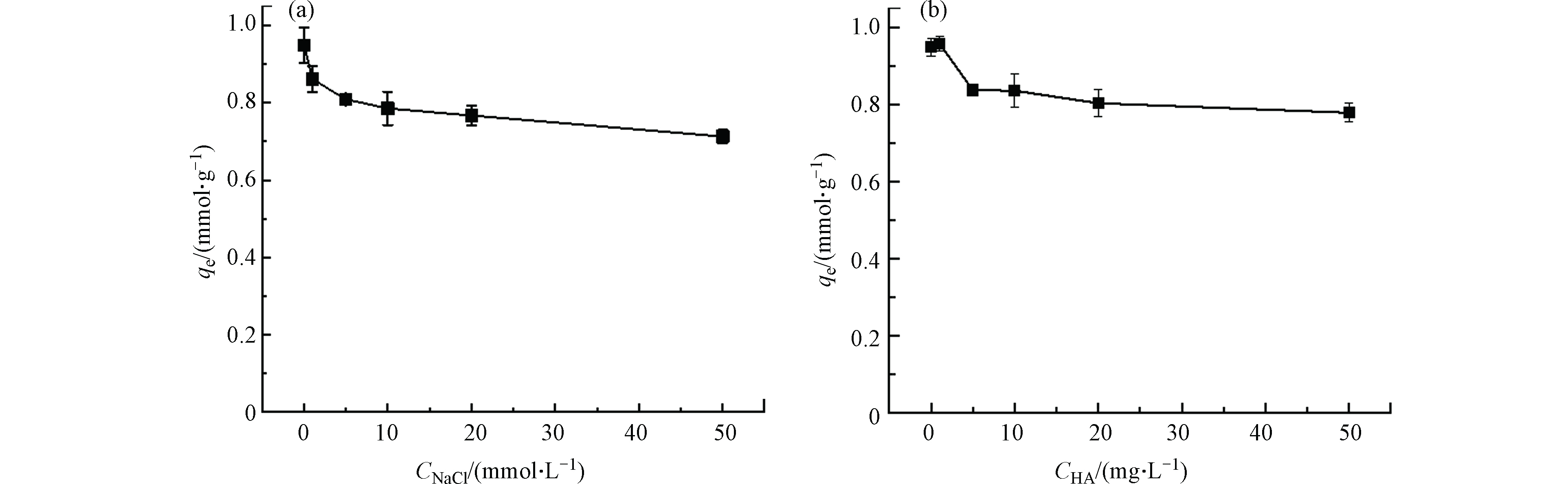
实际水体中常共存有无机盐及水溶性天然有机物,其也会对FQs的吸附过程产生影响. 无机盐(NaCl)对TA-Ti1吸附效果影响如图6a所示,TA-Ti1对OFL吸附量随着无机盐浓度增加而下降并逐渐趋于平衡,这是由于共存无机盐会对吸附剂表面产生电荷屏蔽效应[54-55],从而屏蔽了吸附剂与吸附质间的静电吸引作用. 根据图6a,该静电屏蔽作用在NaCl高浓度条件下(50.0 mmol·L−1)时逐渐平衡,完全抑制了静电吸引;据此可进一步估算静电吸引作用对TA-Ti就OFL吸附的贡献[54-55],50.0 mmol·L−1时NaCl对TA-Ti1吸附OFL的抑制量约为0.249 mmol·g−1,即静电吸引在原吸附过程中贡献约占25%. 进一步证实TA-Ti对OFL的吸附是静电吸引作用协同氢键、π—π相互作用与Ti4+鳌合作用共同作用的结果.
图6b是共存水溶性天然有机物HA对吸附性能的影响,低浓度的HA对OFL的吸附显示出微弱的促进作用,这可能是因为HA和TA的分子结构中均含有大量含氧功能基团,均能与OFL相互作用而促进吸附[56]. 然而随着HA浓度的增加,HA分子上丰富的官能团如羧基、羟基及芳香基团等,会通过氢键等作用与吸附剂进行作用,与OFL发生竞争吸附[57],从而削弱了TA-Ti对OFL的吸附效果,但总体影响较小,在共存HA浓度为50.0 mg·L−1时,吸附量下降约20%. 因此,TA-Ti对共存的无机盐和天然有机物均具有良好的抗干扰能力.
-
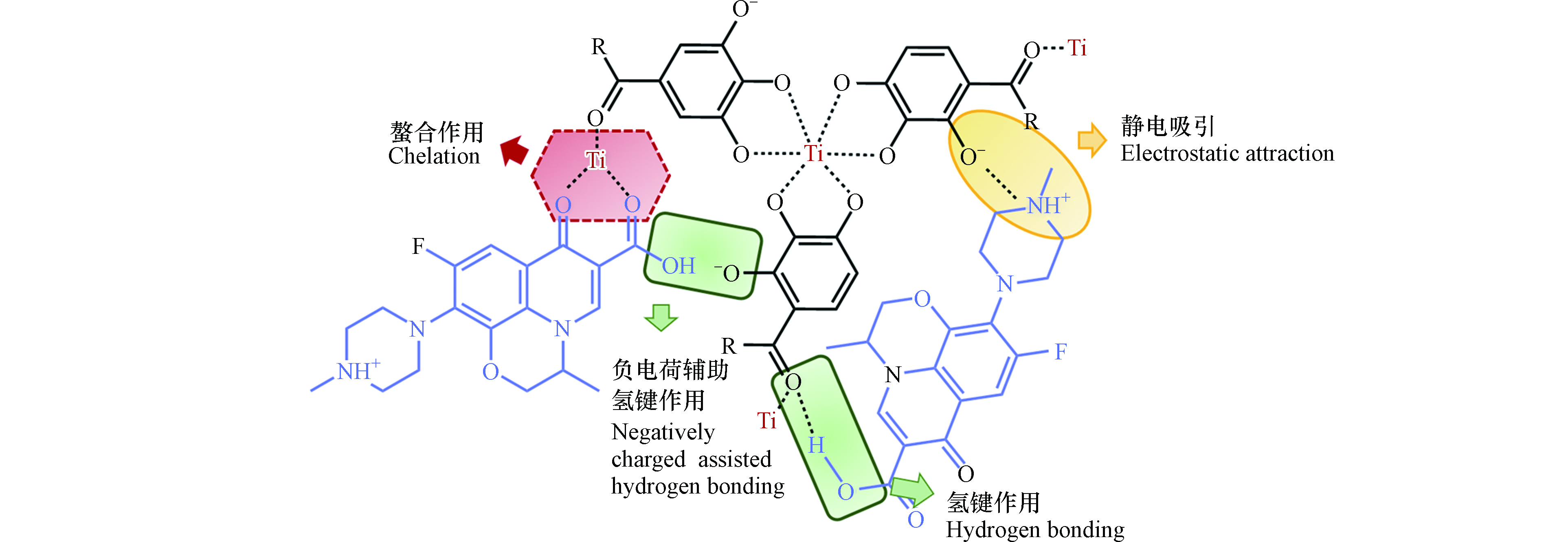
综上所述,TA-Ti吸附FQs存在多种机制共同作用,如图7所示.
TA上丰富的酚羟基、羧基、芳香环等功能基团可分别与FQs上氨基、羧基等发生静电吸引(式3),π—π相互作用及(式4)普通氢键作用(式5—6). 然而在碱性条件下,由于TA-Ti与FQs均带有负电性,静电排斥作用大大削弱了静电吸引、普通氢键及π—π相互作用,但负电荷辅助氢键作用(式7—8)仍使得TA-Ti对FQs具有一定吸附作用. 此外,Ti4+不仅作为交联剂固定成形TA,还可通过螯合作用,进一步有效促进复合吸附剂对FQs的吸附效果.
-
本文采用简便的复合方法制备了一系列单宁酸-钛复合吸附剂材料,TA-Ti对水体中不同FQs均具有良好的吸附性能. TA-Ti在溶液初始pH为8.0时具有最大理论吸附量1.028 mmol·g−1,这是TA和Ti4+协同作用的结果. TA上丰富的功能基团可与FQs发生静电吸引,氢键,负电荷辅助氢键及π—π相互作用,其中静电吸引对总吸附量的贡献约为25%;此外,Ti4+不仅作为交联剂固定成形TA,还可通过螯合作用,有效提高复合吸附剂对FQs的吸附量. TA-Ti对OFL的吸附具有显著的溶液pH依赖性,随着溶液pH的增加吸附量先增加后减少,在pH为8.0时达到最佳吸附效果, 这是由于不同pH条件下,TA-Ti和OFL具有不同的溶液结构形态导致两者间在不同pH时存在不同的作用方式而影响其吸附行为. 在较低pH(pH < 4.0)时,过量的H+会与FQs发生竞争吸附,导致吸附量较低;在较高pH(pH > 9.0)时,TA-Ti与FQs之间的静电排斥作用逐渐增加,严重削弱静电吸引、普通氢键和π—π相互作用,吸附量降低;但负电荷辅助氢键作用使得TA-Ti对FQs在碱性条件下仍具有一定的吸附效果. 吸附动力学和吸附等温线分别符合Elovich模型和Langmuir模型,TA-Ti对FQs的吸附主要为多种吸附作用下的单分子层吸附过程. TA-Ti对共存无机盐NaCl和天然有机物HA表现出较好的抗干扰能力. 总之,TA-Ti不仅制备简单,环境友好,且对FQs具有良好的吸附效果,因而其作为一种绿色高效吸附剂在未来水处理行业中应具有良好的应用前景.
单宁酸-钛复合吸附剂的制备及其对氟喹诺酮类抗生素的吸附去除
Preparation of tannic acid - titanium composite adsorbent and its adsorption performance and mechanisms in removal of fluoroquinolone antibiotics
-
摘要: 本文以一种绿色环保且来源广泛的天然高分子单宁酸(TA)为原材料,四氯化钛为改性剂,一步法制得一系列单宁酸-钛(TA-Ti)复合吸附剂.首先通过多种表征技术考察了吸附剂的分子结构及其表面微观形貌等;针对目前在地表水中已发现的氟喹诺酮类抗生素(FQs),以氧氟沙星(OFL)为代表,系统研究了TA-Ti对OFL的吸附性能,详细考察了溶液pH、污染物浓度、吸附时间及不同共存物等对TA-Ti吸附性能的影响;并对具有与OFL相似结构的FQs及其两种亚结构类似物的吸附效果进行对比,结合吸附前后吸附剂傅里叶红外光谱和X射线光电子能谱谱图的变化,以及吸附动力学与等温吸附模型分析等,深入探讨了TA-Ti对OFL的吸附机制.TA可通过静电吸引、氢键、负电荷辅助氢键及π—π相互作用等与FQs作用;此外,钛离子不仅作为TA的交联剂,同时还可通过螯合作用协同吸附FQs,有效提高TA对FQs的吸附性能,对OFL理论最优吸附量为1.028 mmol·g−1. 本文提供了一种绿色环保的高效吸附剂材料,可有效地净化FQs有机微污染水体,提高用水安全.Abstract: In this work, a series of novel composite adsorbents (TA-Ti) was fabricated using tannic acid (TA) as the raw material and titanium tetrachloride as the modifier. TA is a commonly natural polymer with the advantages of wide source and environmental-friendliness. The molecular structure and surface morphology of this composite adsorbent were investigated by various characterizations. Fluoroquinolones antibiotics (FQs) were currently found in surface water. Among various FQs, ofloxacin (OFL) was selected as a representative in this work. The adsorption performance of TA-Ti in removal of OFL has been investigated systematically. The effects of solution pH, OFL concentration, contact time, coexisting inorganic salt and natural organic substance were studied in detail. The behaviors of TA-Ti in adsorption of some FQs with similar structures to OFL and their two substructural analogies were compared. The adsorption mechanisms have been discussed in detail on the basis of the changes in the Fourier transform infrared and X-ray photoelectron spectra of TA-Ti before and after adsorption as well as the analysis of the adsorption kinetics and isotherms. The efficient adsorption of FQs by TA-Ti was ascribed to the synergetic effects of electrostatic attraction, hydrogen bonding, negatively charged assisted hydrogen bonding, and π—π interactions between TA and FQs; besides, Ti4+ not only acted as a cross-linking agent for TA but also synergistically adsorbed FQs through chelation effect, thereby causing a notably improved adsorption performance. The theoretical maximal OFL uptake of TA-Ti was 1.028 mmol·g−1. In short, this work provided an environmentally-friendly and high-efficient adsorbent that can effectively purify organic micro-polluted water and further improve water safety.
-
Key words:
- fluoroquinolones /
- ofloxacin /
- tannic acid /
- titanium ion /
- adsorption performance /
- adsorption mechanisms
-

-
图 3 (a)不同初始pH对TA-Ti1吸附FQs的影响,(b)TA-Ti1吸附前后的O1s XPS谱图,(c)TA-Ti1对不同FQs在不同初始pH条件下的吸附容量,(d) OFL与Ti4+反应前后的紫外吸收谱图和(e) TA-Ti1吸附前后的Ti2p XPS谱图
Figure 3. (a) Effect of different initial pH on adsorption of OFL, (b) O1s XPS spectra of TA-Ti1 before and after adsorption, (c) the adsorption capacities of TA-Ti1 for various FQs under various pH levels, (d) UV-vis spectra of OFL with and without Ti4+, (e) Ti2p XPS spectra of TA-Ti1 before and after adsorption
表 1 吸附实验条件.
Table 1. Conditions of adsorption experiments.
实验名称
Adsorption experiment吸附剂
Adsorbent投加量/mg Dosage pH 温度/K Temperature 吸附时间/h Contact time FQs浓度/(mol·L−.1)Concentration FQs体积/mL Volume 备注
RemarkspH影响 TA-Ti1
TA-Ti2
TA-Ti35 3.0—10.0 298 12 0.2 30 OFL, CIP, ENR, FLU, FPP 等温吸附 TA-Ti1 5 8.0 298 12 0.05—0.25 30 — 吸附动力学 TA-Ti1 50 8.0 298 0—12 0.2 300 — 无机盐影响 TA-Ti1 5 8.0 298 12 0.2 30 cNaCl = 0—
50 mmol·L−.1有机物影响 TA-Ti1 5 8.0 298 12 0.2 30 cHA = 0—
50 mg·L−.1表 2 氟喹诺酮类抗生素的结构参数及其色谱检测条件.
Table 2. Physicochemical parameters of various FQs and their chromatographic conditions.
FQs 分子结构
Molecular structure分子量
Molecular weightlg Kowa 流动相A和B比例
Ratio of mobile phase
A to B (V:V)紫外检测波长/nm
UV detection
wavelengthOFL[26-27]
pKa1 = 5.98
pKa2 = 8.00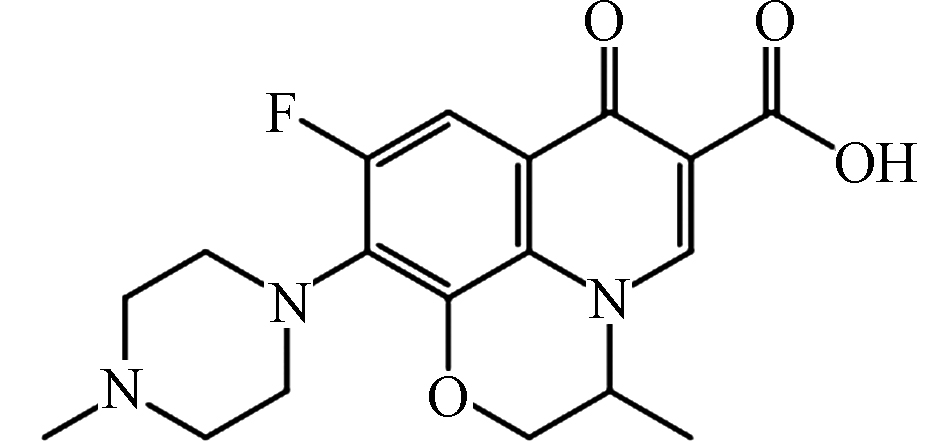
361 −0.39 82:18* 285 CIP[26-27]
pKa1 = 6.14
pKa2 = 8.85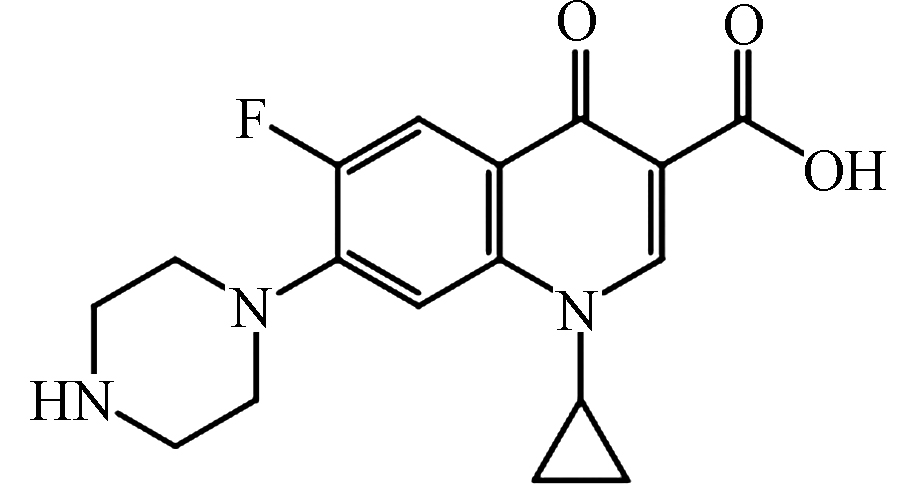
331 0.28 82:18* 285 ENR[26-27]
pKa1 = 6.20
pKa2 = 8.13
359 0.7 82:18* 285 FLU[27]
pKa = 6.29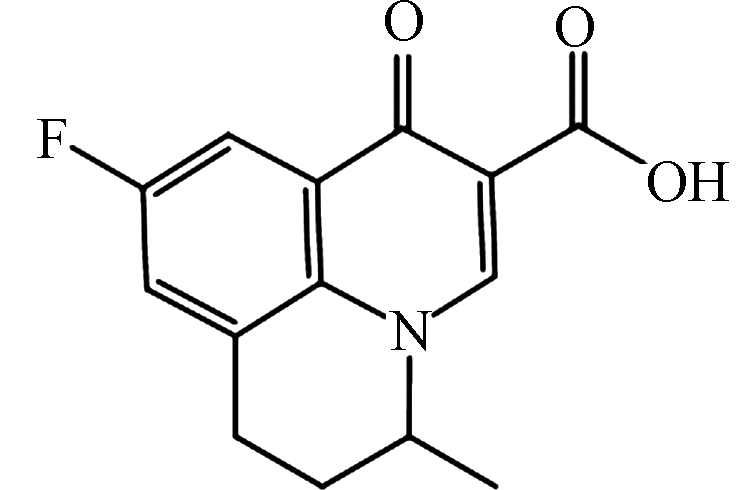
261 50:50* 232 FPP[27]
pKa1 = 4.49
pKa2 = 8.63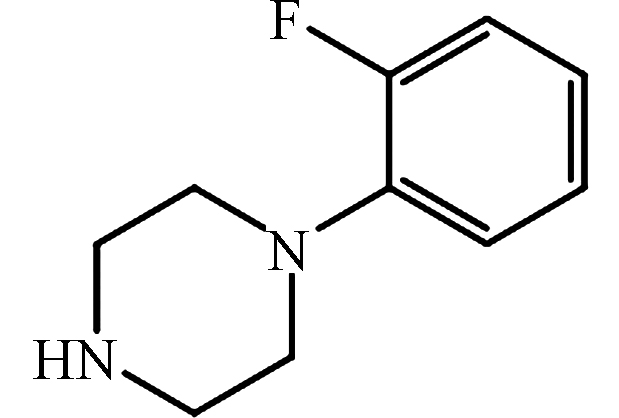
180 85:15* 232 *:流动相A是HPLC级0.8%(V:V)的乙酸溶液,流动相B为HPLC级乙腈.
*:Mobile phase A was HPLC grade 0.8% (V:V) acetic acid solution, and mobile phase B was HPLC grade acetonitrile.表 3 TA-Ti1对OFL吸附的吸附等温线和吸附动力学拟合参数表(pH8.0,温度298 K)
Table 3. Isothermal adsorption and adsorption kinetics fitting parameters of TA-Ti1 in adsorption of OFL at pH of 8.0 and 298 K
qmax,exp/
(mmol·g−1)朗缪尔模型
Langmuir model弗兰德里希模型
Freundlich modelqm /(mmol·g−1) b/(L·mmol−1) R2 Kf n R2 0.971 1.028 101.896 0.958 1.055 4.614 0.906 qmax,exp/
(mmol·g−1)准一级动力学模型
Pseudo-first-order model准二级动力模型
Pseudo-second-order modelElovich 模型
Elovich modelqe1, cal/
(mmol·g−1)k1/h−1 R2 qe2 cal/
(mmol·g−1)k2/
(g·(mmol∙h)−1)R2 qe3 cal/
(mmol·g−1)AE BE R2 0.971 0.889 5.293 0.898 0.932 8.630 0.967 1.003 0.724 0.112 0.982 颗粒内扩散模型
Intraparticle diffusionkp1/
(mmol·(g∙h0.5)−1)C1/
(mmol·g−1)R2 kp2/
(mmol·(g∙h0.5)−1)C2/
(mmol·g−1)R2 kp3/
(mmol·(g∙h0.5)−1)C3/
(mmol·g−1)R2 0.822 0.105 0.889 0.101 0.676 0.946 0.045 0.822 0.946 -
[1] KÜMMERER K. Antibiotics in the aquatic environment - A review - Part I [J]. Chemosphere, 2009, 75(4): 417-434. doi: 10.1016/j.chemosphere.2008.11.086 [2] POLIANCIUC S I, GURZĂU A E, KISS B, et al. Antibiotics in the environment: Causes and consequences [J]. Medicine and Pharmacy Reports, 2020, 93(3): 231-240. [3] LIU X, STEELE J C, MENG X Z. Usage, residue, and human health risk of antibiotics in Chinese aquaculture: A review [J]. Environmental Pollution, 2017, 223: 161-169. doi: 10.1016/j.envpol.2017.01.003 [4] SAPKOTA A, SAPKOTA A R, KUCHARSKI M, et al. Aquaculture practices and potential human health risks: Current knowledge and future priorities [J]. Environment International, 2008, 34(8): 1215-1226. doi: 10.1016/j.envint.2008.04.009 [5] 侯力睿, 傅榆涵, 赵冲, 等. 兽药抗生素对生态环境的混合毒性研究进展 [J]. 环境化学, 2021, 40(1): 55-64. doi: 10.7524/j.issn.0254-6108.2020052502 HOU L R, FU Y H, ZHAO C, et al. Advance on combined toxicity of veterinary antibiotics on ecological environments [J]. Environmental Chemistry, 2021, 40(1): 55-64(in Chinese). doi: 10.7524/j.issn.0254-6108.2020052502
[6] GAO L H, SHI Y L, LI W H, et al. Occurrence of antibiotics in eight sewage treatment plants in Beijing, China [J]. Chemosphere, 2012, 86(6): 665-671. doi: 10.1016/j.chemosphere.2011.11.019 [7] 张国栋, 董文平, 刘晓晖, 等. 我国水环境中抗生素赋存、归趋及风险评估研究进展 [J]. 环境化学, 2018, 37(7): 1491-1500. doi: 10.7524/j.issn.0254-6108.2017112003 ZHANG G D, DONG W P, LIU X H, et al. Occurrence, fate and risk assessment of antibiotics in water environment of China [J]. Environmental Chemistry, 2018, 37(7): 1491-1500(in Chinese). doi: 10.7524/j.issn.0254-6108.2017112003
[8] YANG X, FLOWERS R C, WEINBERG H S, et al. Occurrence and removal of pharmaceuticals and personal care products (PPCPs) in an advanced wastewater reclamation plant [J]. Water Research, 2011, 45(16): 5218-5228. doi: 10.1016/j.watres.2011.07.026 [9] YANG Y, OK Y S, KIM K H, et al. Occurrences and removal of pharmaceuticals and personal care products (PPCPs) in drinking water and water/sewage treatment plants: A review [J]. Science of the Total Environment, 2017, 596/597: 303-320. doi: 10.1016/j.scitotenv.2017.04.102 [10] DONG H, GUO X T, YANG C, et al. Synthesis of g-C3N4 by different precursors under burning explosion effect and its photocatalytic degradation for tylosin [J]. Applied Catalysis B:Environmental, 2018, 230: 65-76. doi: 10.1016/j.apcatb.2018.02.044 [11] DU H W, YANG Z, TIAN Z Q, et al. Enhanced removal of trace antibiotics from turbid water in the coexistence of natural organic matters using phenylalanine-modified-chitosan flocculants: Effect of flocculants’ molecular architectures [J]. Chemical Engineering Journal, 2018, 333: 310-319. doi: 10.1016/j.cej.2017.09.171 [12] MIRZAEI A, CHEN Z, HAGHIGHAT F, et al. Removal of pharmaceuticals from water by Homo/heterogonous Fenton-type processes - A review [J]. Chemosphere, 2017, 174: 665-688. doi: 10.1016/j.chemosphere.2017.02.019 [13] HUANG H L, WANG X H, GE H, et al. Multifunctional magnetic cellulose surface-imprinted microspheres for highly selective adsorption of artesunate [J]. ACS Sustainable Chemistry & Engineering, 2016, 4(6): 3334-3343. [14] KAH M, SIGMUND G, XIAO F, et al. Sorption of ionizable and ionic organic compounds to biochar, activated carbon and other carbonaceous materials [J]. Water Research, 2017, 124: 673-692. doi: 10.1016/j.watres.2017.07.070 [15] GE H, HUANG H L, XU M, et al. Cellulose/poly(ethylene imine) composites as efficient and reusable adsorbents for heavy metal ions [J]. Cellulose, 2016, 23(4): 2527-2537. doi: 10.1007/s10570-016-0973-3 [16] SANTOS S C R, BACELO H A M, BOAVENTURA R A R, et al. Tannin-adsorbents for water decontamination and for the recovery of critical metals: Current state and future perspectives [J]. Biotechnology Journal, 2019, 14(12): 1900060. doi: 10.1002/biot.201900060 [17] CAO Y Y, QI X R, YAN H S. Selective adsorption of tannins over small polyphenols on cross-linked polyacrylamide hydrogel beads and their regeneration with hot water [J]. Reactive and Functional Polymers, 2020, 146: 104398. doi: 10.1016/j.reactfunctpolym.2019.104398 [18] XU Q H, WANG Y L, JIN L Q, et al. Adsorption of Cu (Ⅱ), Pb (Ⅱ) and Cr (Ⅵ) from aqueous solutions using black wattle tannin-immobilized nanocellulose [J]. Journal of Hazardous Materials, 2017, 339: 91-99. doi: 10.1016/j.jhazmat.2017.06.005 [19] DU Y, QIU W Z, WU Z L, et al. Water-triggered self-healing coatings of hydrogen-bonded complexes for high binding affinity and antioxidative property [J]. Advanced Materials Interfaces, 2016, 3(15): 1600167. doi: 10.1002/admi.201600167 [20] WANG G Z, CHEN Y, XU G Q, et al. Effective removing of methylene blue from aqueous solution by tannins immobilized on cellulose microfibers [J]. International Journal of Biological Macromolecules, 2019, 129: 198-206. doi: 10.1016/j.ijbiomac.2019.02.039 [21] 何南, 王振全, 董蕙, 等. 单宁酸-铁离子纳米薄膜的合成及应用现状 [J]. 环境化学, 2021, 40(11): 3553-3560. doi: 10.7524/j.issn.0254-6108.2020071901 HN N, WANG Z Q, DONG H, et al. Synthesis and application of tannic acid - iron ion nano films [J]. Environmental Chemistry, 2021, 40(11): 3553-3560(in Chinese). doi: 10.7524/j.issn.0254-6108.2020071901
[22] GUO J L, PING Y, EJIMA H, et al. Engineering multifunctional capsules through the assembly of metal-phenolic networks [J]. Angewandte Chemie International Edition, 2014, 53(22): 5546-5551. doi: 10.1002/anie.201311136 [23] ZHONG Q Z, PAN S J, RAHIM M A, et al. Spray assembly of metal-phenolic networks: Formation, growth, and applications [J]. ACS Applied Materials & Interfaces, 2018, 10(39): 33721-33729. [24] YAN W T, SHI M Q, DONG C X, et al. Applications of tannic acid in membrane technologies: A review [J]. Advances in Colloid and Interface Science, 2020, 284: 102267. doi: 10.1016/j.cis.2020.102267 [25] LIU X, ZHANG L F. Insight into the adsorption mechanisms of vanadium(Ⅴ) on a high-efficiency biosorbent (Ti-doped chitosan bead) [J]. International Journal of Biological Macromolecules, 2015, 79: 110-117. doi: 10.1016/j.ijbiomac.2015.04.065 [26] JIANG C L, JI Y F, SHI Y Y, et al. Sulfate radical-based oxidation of fluoroquinolone antibiotics: Kinetics, mechanisms and effects of natural water matrices [J]. Water Research, 2016, 106: 507-517. doi: 10.1016/j.watres.2016.10.025 [27] van DOORSLAER X, DEWULF J, van LANGENHOVE H, et al. Fluoroquinolone antibiotics: An emerging class of environmental micropollutants [J]. Science of the Total Environment, 2014, 500/501: 250-269. doi: 10.1016/j.scitotenv.2014.08.075 [28] ZHAO X T, JIA N, CHENG L J, et al. Metal-polyphenol coordination networks: Towards engineering of antifouling hybrid membranes via in situ assembly [J]. Journal of Membrane Science, 2018, 563: 435-446. doi: 10.1016/j.memsci.2018.06.014 [29] WU H Q, XIE J D, MAO L. One-pot assembly tannic acid-titanium dual network coating for low-pressure nanofiltration membranes [J]. Separation and Purification Technology, 2020, 233: 116051. doi: 10.1016/j.seppur.2019.116051 [30] GAO X H, WANG Q, REN L L, et al. Metal-phenolic networks as a novel filler to advance multi-functional immunomodulatory biocomposites [J]. Chemical Engineering Journal, 2021, 426: 131825. doi: 10.1016/j.cej.2021.131825 [31] WANG R X, ZHAO X T, LAN Y Y, et al. In situ metal-polyphenol interfacial assembly tailored superwetting PES/SPES/MPN membranes for oil-in-water emulsion separation [J]. Journal of Membrane Science, 2020, 615: 118566. doi: 10.1016/j.memsci.2020.118566 [32] STRUSZCZYK H. Microcrystalline chitosan. I. preparation and properties of microcrystalline chitosan [J]. Journal of Applied Polymer Science, 1987, 33(1): 177-189. doi: 10.1002/app.1987.070330115 [33] CHEN J D, WANG H Y, GONG Y T, et al. Directly immobilizing a Ru–tannic acid linkage coordination complex on carbon cloth: An efficient and ultrastable catalyst for the hydrogen evolution reaction [J]. Journal of Materials Chemistry A, 2019, 7(18): 11038-11043. doi: 10.1039/C9TA02680K [34] CHEN J Q, PAN S J, ZHOU J J, et al. Programmable permeability of metal-phenolic network microcapsules [J]. Chemistry of Materials, 2020, 32(16): 6975-6982. doi: 10.1021/acs.chemmater.0c02279 [35] HAO B C, WANG F, HUANG H, et al. Tannin foam immobilized with ferric ions for efficient removal of ciprofloxacin at low concentrations [J]. Journal of Hazardous Materials, 2021, 414: 125567. doi: 10.1016/j.jhazmat.2021.125567 [36] YANG C, WU H, YANG X, et al. Coordination-enabled one-step assembly of ultrathin, hybrid microcapsules with weak pH-response [J]. ACS Applied Materials & Interfaces, 2015, 7(17): 9178-9184. [37] GILLI P, PRETTO L, BERTOLASI V, et al. Predicting hydrogen-bond strengths from acid-base molecular properties. The pK(a) slide rule: Toward the solution of a long-lasting problem [J]. Accounts of Chemical Research, 2009, 42(1): 33-44. doi: 10.1021/ar800001k [38] PENG C L, ZHONG Y H, MIN F F. Adsorption of alkylamine cations on montmorillonite (001) surface: A density functional theory study [J]. Applied Clay Science, 2018, 152: 249-258. doi: 10.1016/j.clay.2017.11.021 [39] 李竟先, 庄志强, 吴基球, 等. 水解-水热体系中同质异构TiO2纳米晶生成机理的分子模拟研究 [J]. 中国陶瓷, 2005, 41(5): 11-13,10. doi: 10.3969/j.issn.1001-9642.2005.05.004 LI J X, ZHUANG Z Q, WU J Q, et al. Computation researches of molecular simulation on the principle of producing tautomerism TiO2 nanometer crystal in hydrolytic-hydrothermal system [J]. China Ceramics, 2005, 41(5): 11-13,10(in Chinese). doi: 10.3969/j.issn.1001-9642.2005.05.004
[40] NEDELJKOVIĆ N V, NIKOLIĆ M V, MIJAJLOVIĆ M Ž, et al. Interaction of bioessential metal ions with quinolone antibiotics: Structural features and biological evaluation [J]. Inorganica Chimica Acta, 2021, 527: 120585. doi: 10.1016/j.ica.2021.120585 [41] UIVAROSI V. Metal complexes of quinolone antibiotics and their applications: An update [J]. Molecules (Basel, Switzerland), 2013, 18(9): 11153-11197. doi: 10.3390/molecules180911153 [42] 黄群莲, 邓以平, 徐绍友. 紫外分光光度法测定氧氟沙星的解离常数 [J]. 中国药房, 2010, 21(41): 3907-3909. HUANG Q L, DENG Y P, XU S Y. Determination of dissociation constants of ofloxacin by UV spectrophotometry [J]. China Pharmacy, 2010, 21(41): 3907-3909(in Chinese).
[43] GU X Y, TAN Y Y, TONG F, et al. Surface complexation modeling of coadsorption of antibiotic ciprofloxacin and Cu(Ⅱ) and onto goethite surfaces [J]. Chemical Engineering Journal, 2015, 269: 113-120. doi: 10.1016/j.cej.2014.12.114 [44] 王剑, 申伟, 胡小莉, 等. Pd(Ⅱ)对氟喹诺酮类抗生素的荧光猝灭作用及其分析应用[J]. 科学通报, 2014, 59(S1)∶328-336. WANG J, SHEN W, HU X L, et al. The fluorescence quenching of fluoroquinolones by palladium(Ⅱ) and its analytical application[J]. Chinese Science Bulletin, 2014, 59(Sup 1): 328-336(in Chinese).
[45] MA J, XIONG Y C, DAI X H, et al. Coadsorption behavior and mechanism of ciprofloxacin and Cu(Ⅱ) on graphene hydrogel wetted surface [J]. Chemical Engineering Journal, 2020, 380: 122387. doi: 10.1016/j.cej.2019.122387 [46] LANGMUIR I. The adsorption of gases on plane surfaces of glass, Mica and platinum [J]. Journal of the American Chemical Society, 1918, 40(9): 1361-1403. doi: 10.1021/ja02242a004 [47] FREUNDLICH H. Over the adsorption in solution [J]. Journal of Physical Chemistry, 1906, 57: 385-470. [48] ANIRUDHAN T S, RADHAKRISHNAN P G. Chromium(Ⅲ) removal from water and wastewater using a carboxylate-functionalized cation exchanger prepared from a lignocellulosic residue [J]. Journal of Colloid and Interface Science, 2007, 316(2): 268-276. doi: 10.1016/j.jcis.2007.08.051 [49] HO Y S, MCKAY G, WASE D A J, et al. Study of the sorption of divalent metal ions on to peat [J]. Adsorption Science & Technology, 2000, 18(7): 639-650. [50] LOW M J D. Kinetics of chemisorption of gases on solids [J]. Chemical Reviews, 1960, 60(3): 267-312. doi: 10.1021/cr60205a003 [51] YURDAKOÇ M, SEKI Y, KARAHAN S, et al. Kinetic and thermodynamic studies of boron removal by Siral 5, Siral 40, and Siral 80 [J]. Journal of Colloid and Interface Science, 2005, 286(2): 440-446. doi: 10.1016/j.jcis.2004.12.047 [52] WU F C, TSENG R L, JUANG R S. Characteristics of Elovich equation used for the analysis of adsorption kinetics in dye-chitosan systems [J]. Chemical Engineering Journal, 2009, 150(2/3): 366-373. [53] FAN R Y, MIN H Y, HONG X X, et al. Plant tannin immobilized Fe3O4@SiO2 microspheres: A novel and green magnetic bio-sorbent with superior adsorption capacities for gold and palladium [J]. Journal of Hazardous Materials, 2019, 364: 780-790. doi: 10.1016/j.jhazmat.2018.05.061 [54] LIAN F, SUN B B, SONG Z G, et al. Physicochemical properties of herb-residue biochar and its sorption to ionizable antibiotic sulfamethoxazole [J]. Chemical Engineering Journal, 2014, 248: 128-134. doi: 10.1016/j.cej.2014.03.021 [55] TEIXIDÓ M, PIGNATELLO J J, BELTRÁN J L, et al. Speciation of the ionizable antibiotic sulfamethazine on black carbon (biochar) [J]. Environmental Science & Technology, 2011, 45(23): 10020-10027. [56] JIN J, FENG T Y, GAO R, et al. Ultrahigh selective adsorption of zwitterionic PPCPs both in the absence and presence of humic acid: Performance and mechanism [J]. Journal of Hazardous Materials, 2018, 348: 117-124. doi: 10.1016/j.jhazmat.2018.01.036 [57] GAO B Q, CHANG Q Q, CAI J, et al. Removal of fluoroquinolone antibiotics using Actinia-shaped lignin-based adsorbents: Role of the length and distribution of branched-chains [J]. Journal of Hazardous Materials, 2021, 403: 123603. doi: 10.1016/j.jhazmat.2020.123603 -



 下载:
下载: