-
近年来,随着纺织、造纸、矿山开采等行业的发展,水体中染料和重金属污染日趋严重[1-4],且染料废水属于长期困扰人们生活的难处理的工业废水之一[5]. 研究表明,燃料电池技术可以在降解污染物的同时将化学能转换为电能[6-9],阳极发生电子-空穴对分离,分解水中有机污染物,产生的电子通过外电路传导到阴极,发生氧化还原反应[10]。燃料电池技术既能较好的解决环境污染问题,又可以产生新的能源,实现节能环保[11].
g-C3N4是一种具有适宜禁带宽度的半导体材料,其化学性质稳定[12],且不溶于乙醇、水、甲苯等溶剂. 此外,g-C3N4还具有廉价、无毒、原材料易得等优点[13],被人们广泛应用。g-C3N4具有独特的电子结构以及较大的比表面积,为光催化过程提供更多的活性位点,有利于提高光的捕获率,再者,高的比表面积材料可减少光生电子与空穴对的扩散路径,增加反应动力学,被广泛应用于光催化去除无机、有机污染物[14]、氧化还原反应[15]、锂离子电池[16]、生物燃料电池[17]等领域.
过渡金属二硫族化合物(TMDs)因其带隙可控、电子迁移率高和尺寸可达原子尺度等性能特点,被广泛应用于电池、超级电容器、光伏材料、催化剂等领域[18-20]。TMDs主要包括二硫化钼(MoS2)、硒化钨(WSe2)、和硒化钼(MoSe2)等[21]. 二硫化钼(MoS2)是一个典型的层状化合物,层间是以微弱的范德华力结合在一起[22]. 二硫化钼具有低成本、边沿暴露活性位点可调控、耐酸性条件等优点[23],属于n型半导体;在光催化剂、电化学等领域广泛应用[24-25]。但MoS2电导率较差,限制了其广泛应用[25, 27]. 为克服这一缺点,可将其粒径减小到纳米尺寸,与导电剂混合,通过改变形貌,将二维纳米片转为稳定的三维矩阵,或通过添加模板减小MoS2纳米片的堆叠,以提高其电子传递速率[24,27].
WSe2具有高的光吸收、阳离子间的层状排列、抗光腐蚀能力强等优点,是光电转换和光伏太阳能转换的重要材料。目前,国内外对其形貌的研究相对较少,且较少合成出比纳米WSe2纯度更高的其他形貌的WSe2[28]. 由于WSe2和MoS2已经表现出p型和n型半导体的性质[29],MoS2和WSe2之间可以形成内电场,促进电子空穴的分离. WSe2可作为辅助剂,与MoS2形成p-n异质结,提高材料的电子和空穴分离效率,进一步提高催化效率.
本文通过水热法将硒化钨和二硫化钼进行复合,形成MoS2基范德华异质结复合材料MoS2/WSe2(MW),并对其进行XRD、SEM以及TEM表征. 采用g-C3N4作为阴极,MW作为阳极,构建自偏压式燃料电池,研究其对不同类型污染物的去除性能. 值得注意的是,为探求能源的最小消耗以及电池的实际应用性能,所有试验均无需光照,本实验利用电池自身产生的内偏压进行污染物降解,能耗仅为曝气。研究表明,MW表现出比MoS2、WSe2更为优越的催化活性,并通过改变污染物浓度以及污染物种类探究该电池对污染物去除效果的影响.
-
实验所用试剂均为分析纯,试剂与WSe2材料均从上海阿拉丁生化科技股份有限公司处采购.
-
(1) MoS2制备
MoS2的合成采用水热合成法[25],将0.72 g Na2MoO4·2H2O和0.69 g CH4N2S溶于1 mL浓度为1 mol·L−1的1-丁基-3-甲基咪唑氯盐(BMMI[Cl])中,随后将60 mL浓度为0.2 mol·L-1的稀盐酸溶液逐滴加入,持续搅拌12 h. 搅拌结束后,将溶液转移至100 mL带聚四氟乙烯内胆的水热反应釜中,密闭,于220 ℃条件下反应24 h. 反应结束后,将固体样品离心分离,用去离子水和乙醇清洗3次以上,于60 ℃烘箱中烘干,制得黑色MoS2粉末.
(2) MW复合材料制备
将0.72 g Na2MoO4·2H2O和0.69 g CH4N2S溶于1 mL浓度为1 mol·L−1的BMMI[Cl]中,随后逐滴加入60 mL浓度为0.2 mol·L−1的稀盐酸溶液,将0.4 g WSe2加入上述溶液中,持续搅拌12 h. 搅拌完成后,转移至100 mL聚四氟乙烯水热反应釜内胆中,密闭,于220 ℃条件下反应24 h,通过离心分离,去离子水和乙醇清洗,最终得到MW复合物样品.
(3) g-C3N4制备
取3 g三聚氰胺于坩埚中,用马弗炉以5 ℃·min-1的升温速率升至550 ℃,保持4 h,反应完成后,对样品进行研磨,得到g-C3N4淡黄色粉末.
(4) 电池电极制作
选用碳纤维刷为电极基底材料,将催化剂样品与中性硅溶胶溶液通过超声、搅拌混合均匀后,将催化剂均匀的涂覆于碳纤维刷表面,自然风干,制得相应的催化电极. 其中,阴极催化剂为g-C3N4,阳极催化剂分别为MoS2、WSe2和MW.
-
如图1所示,电池反应器为20 cm×10 cm×10 cm的空心立方体结构,主体材料为聚甲基丙烯酸甲酯,侧壁为石英玻璃. 反应器为双室结构,两室相互独立,将上述电极分别置于阳极室与阴极室中,污染物溶液用质子交换膜分隔,可以同时去除相同或不同污染物. 外部电路串联500 Ω外接电阻,反应期间的输出电压用数据收集装置记录.
通过对比不同阳极材料的燃料电池对污染物的去除效率,探究3种材料在无光条件下的催化性能强弱. 改变不同实验条件:溶液pH,电解质溶液浓度,污染物浓度,探究其对无光条件下同时去除有机污染物与重金属性能的影响.
-
图2为MoS2、WSe2、MW和g-C3N4的SEM图. 图2(a)中可以看出,MoS2为花球状结构,且分散性较差,多团簇在一起. 如图2(b)所示,WSe2的形貌呈层片状结构,堆叠在一起,且大小不一. 图2(c)为MW复合材料的SEM图,其形貌与MoS2类似,可以观察到部分MoS2花球包裹WSe2的现象,团簇现象减少。由图2(d)所示,g-C3N4为纳米层片结构.
-
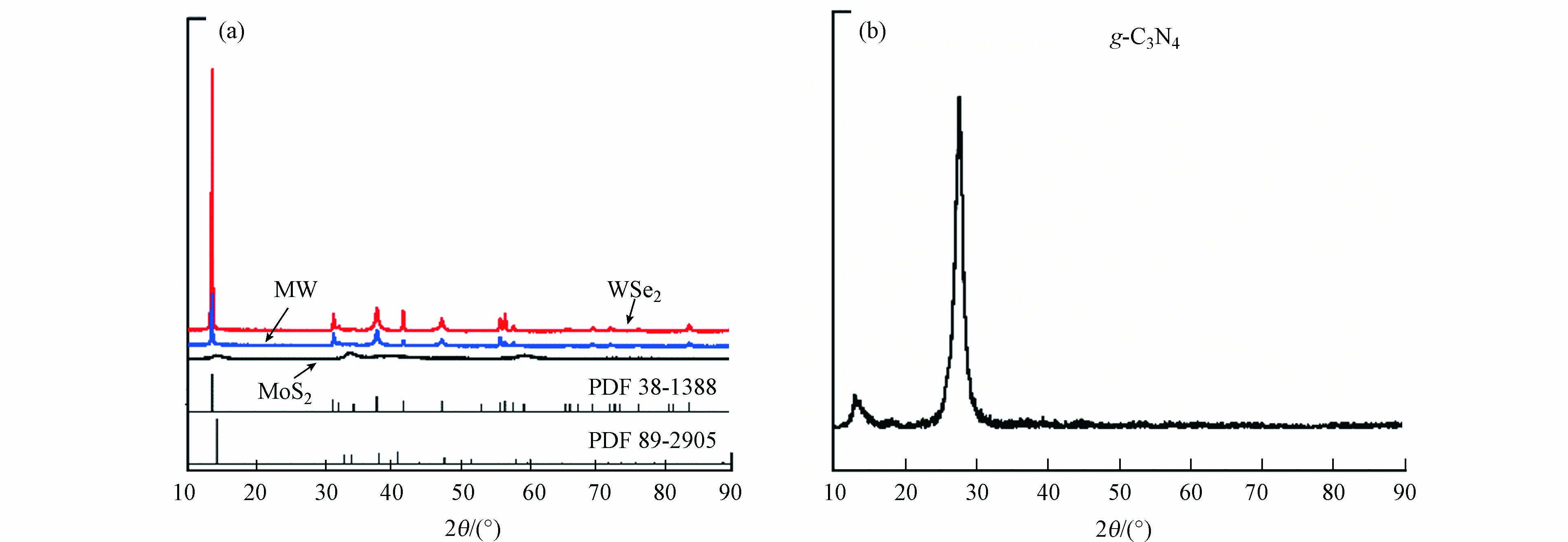
对样品进行XRD分析可以进一步分析样品的晶体结构和组成,有利于确定所得样品的成分。图3为MoS2、WSe2、MW与g-C3N4等材料的XRD表征图。X射线衍射测试的2θ的范围选定为10°—90°. 如图3(a)所示,MoS2所测得的谱图上可找到对应于PDF#89-2905和PDF#87-2416卡片的衍射峰,且匹配度较高,说明本实验合成的MoS2中存在3R-MoS2. 此外,WSe2与PDF#38-1388相匹配,且响应值较高,说明WSe2的结晶度较好. 如图3(a)所示,在MW材料的XRD谱图上发现了对应于原材料MoS2(PDF#89-2905)和WSe2(PDF#13-1388)的特征峰,说明MoS2与WSe2这两种成分都存在于复合材料中。如图3(b)所示,本文所合成的g-C3N4材料于13.1°和27.7°处的衍射峰可与g-C3N4的(100)和(002)晶面相对应,说明成功合成g-C3N4[8,30].
-
如图4(a)所示,距离为0.64 nm的晶格条纹,对应于MoS2的特征条纹. 图4(b)中,间距为0.33 nm和0.38 nm的晶格条纹,分别对应于WSe2的(1,1,0)晶面和(1,0,0)晶面[31],如图4(c)所示,中间黑色部分为WSe2,边缘处为MoS2,说明MoS2和WSe2成功复合在一起,且MoS2包覆于WSe2表面. 观察图4(d)可以清楚地发现对应于Mo、S、W和Se的EDS特征峰。综上,说明MoS2已成功包裹于WSe2表面.
-
(1)阳极材料优化
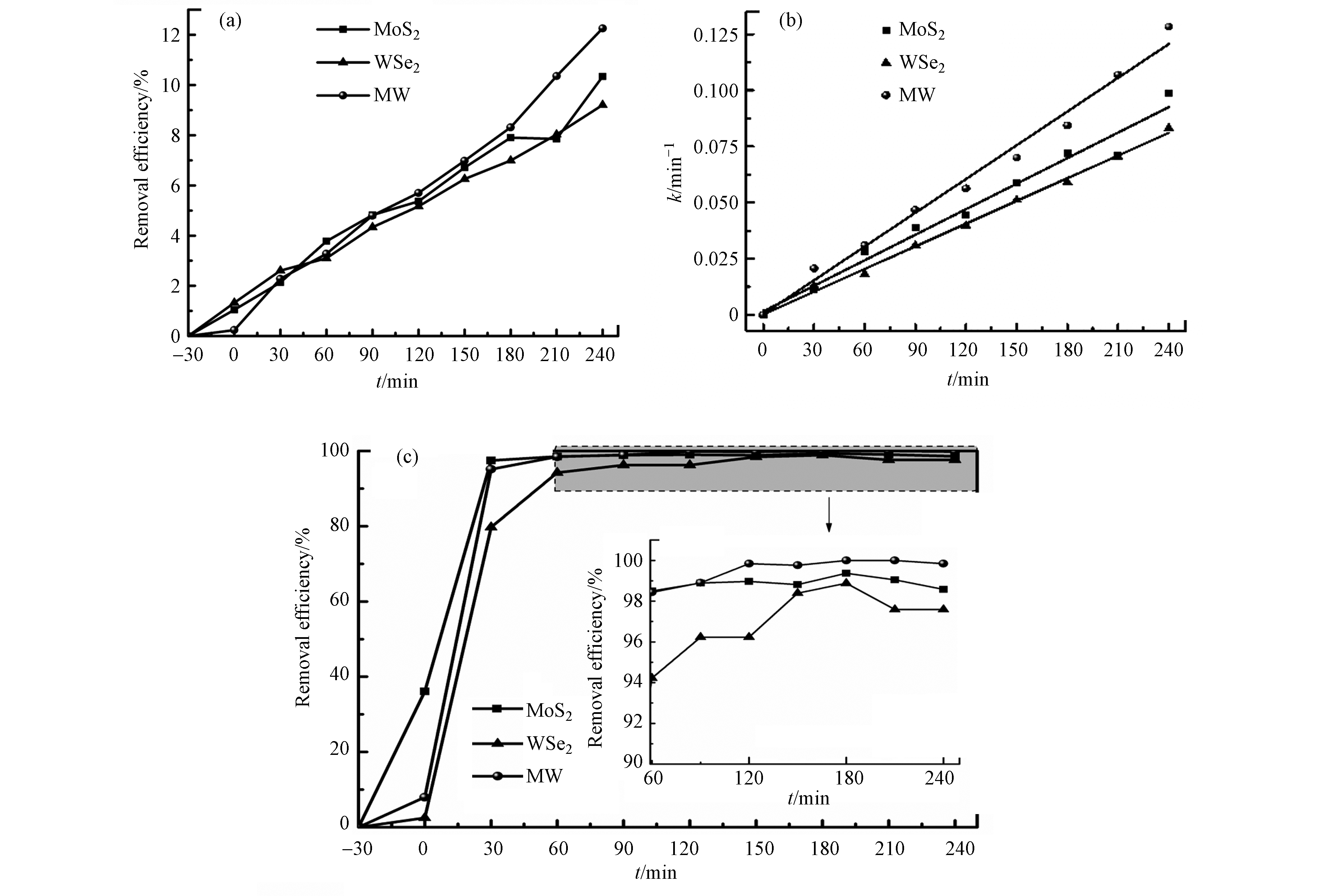
如图5(a)所示,在阴极材料为g-C3N4的条件下,阳极为MW时,燃料电池阴极对于重金属镉(Cd2+)的去除效率12.3%高于WSe2(9.2%)和MoS2(10.3%). 由图5(b)可以看出,MW去除Cd2+的一级反应速率常数k也为三者中的最大值5.01×10−4 min−1。在240 min时,Cd2+去除率仅为12.3%,说明在无光条件下,对低浓度重金属Cd2+的去除能力较弱. 如图5(c)所示,分别以MoS2、WSe2和MW为阳极材料,60 min时,MoS2、WSe2和MW对罗丹明B的去除效率已高达95%,在240 min时,MoS2、WSe2和MW对罗丹明B的去除率分别为98.6%、97.6%、99.8%。MW阳极同样对罗丹明B显示出较高的催化活性。实验过程中,均会产生微小电压.
(2)Cu2+去除性能研究
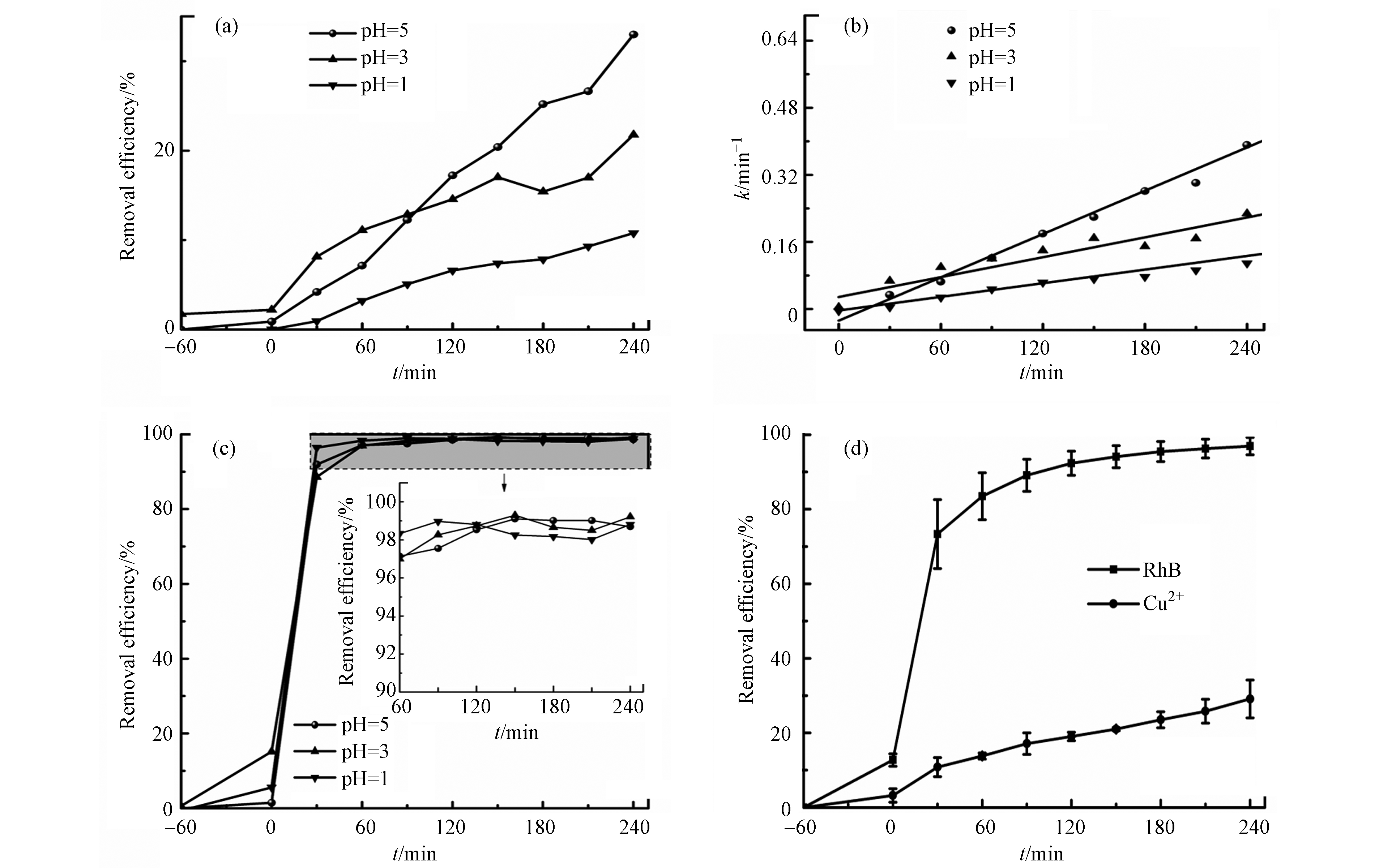
通过对比图5(a)和图6(a)可知,在其他条件不变的情况下,以MW为阳极材料,g-C3N4作为阴极材料,将阴极溶液更改为等浓度的铜离子(Cu2+)溶液,阴极可以得到更高的去除效率,即重金属Cu2+去除效率为29.5%。如图6(a)所示,在阴极材料为g-C3N4的条件下,pH = 5燃料电池对于重金属铜(Cu2+)展示出了最高的去除效率33.0%高于pH = 3的21.8%和pH = 1的10.8%,说明酸性的增强,不利于铜离子的去除。且通过图6(b)可以看出,pH = 5条件下,阴极重金属离子的k值最大,为2×10−3。如图6(c)所示,阴极室溶液pH 值分别为5、3、1时,罗丹明B的去除效率分别为98.7%、99.2%、98.8%,pH对罗丹明B的去除效率影响很小。图6(d)为pH=5时的误差棒,该实验数据误差在10%以内,在误差允许范围内. 研究发现,电池电压随pH值的减小而增大[7],当pH为1时,电池的电压最大. 由以上分析可知,调节pH值对污染物去除效率并没有促进作用,仅可提高电池电压.
(3)电解质浓度对比
如图7(a)所示,在阴极材料为g-C3N4的条件下,电解质浓度为0.1 mol·L−1燃料电池对于重金属铜(Cu2+)也展示出了最高的去除效率43.8%高于0.2 mol·L−1的35.06%、0.3 mg·L−1的26.7%以及0.4 mol·L−1的28.12%. 如图7(b)所示,进行一级动力学拟合后的一级反应速率常数中,0.2 mol·L−1的拟合值最高,为2×10−3 min−1。如图7(c)所示,阴极溶液电解质分别为0.1、0.2、0.3、0.4 mol·L−1时,对应的阳极材料对罗丹明B的去除效率分别为98.1%、98.5%、95.0%以及97.9%. 研究发现,当电解质溶液浓度分别0.1 mol·L−1、0.2 mol·L−1时,对应的阳极材料对罗丹明B的去除效果几乎相同,且电解质浓度为0.1 mg·L−1时电池的催化能力最高.
(4)Cu2+浓度对比
从图8(a)可以看出,改变阴极重金属溶液的浓度,对污染物的去除效率有一定影响,其中,降解240 min时,Cu2+浓度为5 mg·L−1时去除效率为46.4%,对应反应速率常数为1.79×10−3 min-1,浓度为4 mg·L−1时,其去除率最高达64.3%,反应速率常数为0.02 min−1. 图8(c)可以看出,当阴极重金属溶液浓度为2、4、5 mg·L−1时,对阳极罗丹明B的降解效率分别为99.45%、99.06%、98.67%. 由此可以得出,阴极重金属浓度变化,只会影响阴极对重金属离子的去除效果,对阳极有机染料的降解几乎没有影响. 通过对比图8(a)中重金属溶液浓度为8 mg·L−1和5 mg·L−1的降解效果,从而排除吸附对该实验的干扰.
(5)自偏压式燃料电池机理研究
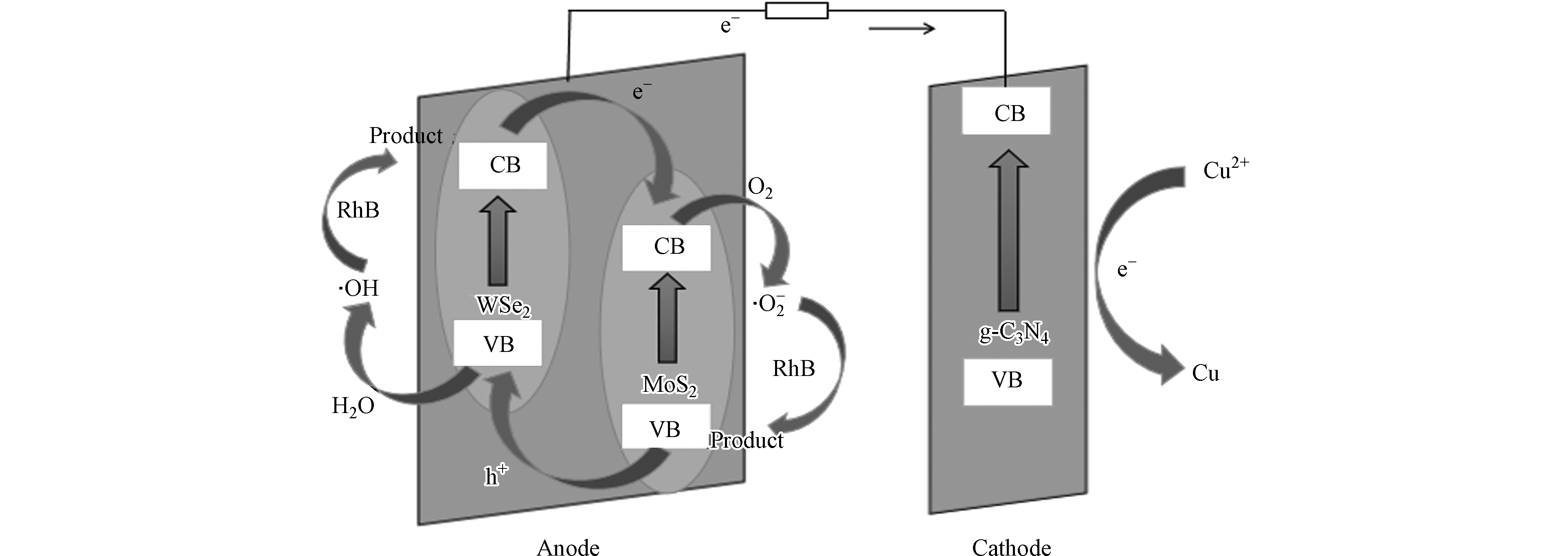
通过自由基捕获实验,得出本实验体系主要是超氧自由基和羟基自由基起作用. 本实验在无光条件下,通过合成p-n型异质结,形成自偏压系统,实现两种污染物同时降解. 如图9所示,MW异质结为p-n型异质结,由于MoS2和WSe2之间存在费米能级差,电子从费米能级高的一侧流向费米能级低的一侧,直至两边的费米能级一致. 且两室载流子之间存在浓度差,引起电荷流动. 同时空穴向相反方向迁移,电子与空穴复合,剩下未经中和的施主离子和不能运动的受主离子,在半导体材料交界面形成空间电荷区,从而建立起稳定的内建电场. MW异质结间的电子流动,活化阳极溶液中的氧气,产生超氧自由基,用于罗丹明B的降解. 随着罗丹明B的降解,亦可产生电子持续活化氧气,电子经电解质和外电路传递到阴极,促进阴极光催化材料产生电子空穴对,还原重金属离子. 经过多次实验得出,该电池稳定性很好,多次重复使用催化电极材料后,催化电极仍具有较高的催化活性.
-
本文通过水热合成法将MoS2与WSe2进行复合,制备MW复合材料,并以其作为阳极材料,g-C3N4作为阴极材料,组建自偏压燃料电池,在降解有机污染物的同时去除废水中的重金属离子. 研究表明,MW的催化能力优于MoS2和WSe2;黑暗条件下,通过改变阴极重金属废水的浓度,电解质浓度以及pH,探究以上因素对燃料电池去除罗丹明B和重金属(Cd或Cu)性能的影响;当溶液pH=5,电解质溶液浓度为0.1 mol·L−1时,铜离子浓度为4 mg·L−1时,重金属的去除率为64.3%。当溶液pH=5,电解质溶液浓度为0.2 mol·L−1,重金属浓度为2 mg·L−1时,有机污染物罗丹明B的去除率最高为99.5%. 通过实验发现,该燃料电池在降解污染物的同时能够产生少量电压,而且通过实验对比自然光和无光条件下的效果,得出自然光条件下的降解效果略优于无光条件. 本实验体系在自然光和无光条件下都展现良好的去除效果,且在实现两种不同类型污染物的去除同时,能耗仅为曝气,无需外加光照或外加偏压,为燃料电池的实际应用提供新思路.
g-C3N4-MoS2/WSe2自偏压系统构建与不同类型污染物同时去除性能研究
Study on simultaneous removal performance of g-C3N4-MoS2 /WSe2 Self-bias system with different types of pollutants
-
摘要: 由于工业化进程的不断推进,染料与重金属废水被非法排入自然水体,水体污染问题日益严峻。为实现两种污染物的同时去除,本文通过调控合成MoS2/WSe2(MW)异质结复合催化材料,提高其催化活性. 黑暗条件下,以MoS2、WSe2以及MW为阳极材料,g-C3N4为阴极材料组建自偏压燃料电池系统,实现在降解有机染料的同时,去除水体中的重金属离子. 通过调控参数,探究影响染料与重金属去除的因素. 研究表明,影响重金属和有机染料去除效果的因素有pH、电解质溶液浓度、重金属溶液浓度. 当溶液pH=5,电解质溶液浓度为0.1 mol·L−1,铜离子浓度为4 mg·L−1时,重金属的去除率为64.3%. 当溶液pH=5,电解质溶液浓度为0.2 mol·L−1,铜离子浓度为2 mg·L−1时,有机染料罗丹明B的去除率为99.5%. 该系统在无外加光照条件下,实现不同类型污染物同时去除,并产生约240 mV的电压,为无光条件下光催化材料的实际应用提供新思路.Abstract: Due to the continuous advancement of industrialization, dye and heavy metal wastewater is illegally discharged into natural water bodies, which makes the problem of water pollution more and more serious. In order to realize simultaneous degradation of the two pollutants, MoS2/WSe2 (MW) heterojunction composite was synthesized to improve its catalytic activity in this paper. Under dark conditions, the self-biased fuel cell was constructed with MoS2, WSe2 and MW as anode materials, g-C3N4 as cathode material to degrade organic pollutants and remove heavy metal ions in water. By adjusting the parameters and exploring the factors affecting the removal of dyes and heavy metals, the study showed that the removal efficiency of heavy metals and organic dyes was affected by pH, electrolyte and heavy metal concentration. When the pH 5, electrolyte solution concentration was 0.1 mol·L−1, and copper ion concentration was 4 mg·L−1, the removal rate of heavy metals was 64.3%. When the pH 5, electrolyte solution concentration was 0.2 mol·L−1, and copper ion concentration was 2 mg·L−1, the removal rate of organic dye rhodamine B was 99.45%. This system can remove different kinds of pollutants and generate a voltage of about 240 mV at the same time in the absence of illumination, which provides a new idea for the practical application of photocatalytic materials in dark conditions.
-
Key words:
- g-C3N4 /
- MoS2/WSe2 /
- self-biased fuel cell /
- dye /
- heavy metal
-

-
-
[1] FU F, WANG Q. Removal of heavy metal ions from wastewaters: a review [J]. Journal of Environmental Management, 2011, 92(3): 407-418. [2] LI Z, MA Z, KUIJP T J, et al. A review of soil heavy metal pollution from mines in China: pollution and health risk assessment [J]. Science of Total Environment, 2014, 468/469: 843-853. doi: 10.1016/j.scitotenv.2013.08.090 [3] LI Z, CHEN J, GUO H, et al. Triboelectrification-enabled self-powered detection and removal of heavy metal ions in wastewater [J]. Advanced Materials, 2016, 28(15): 2983-2991. doi: 10.1002/adma.201504356 [4] RABÉ K, LIU L, NAHYOON N A, et al. Enhanced Rhodamine B and coking wastewater degradation and simultaneous electricity generation via anodic g-C3N4/Fe0(1%)/TiO2 and cathodic WO3 in photocatalytic fuel cell system under visible light irradiation [J]. Electrochimica Acta, 2019, 298: 430-439. doi: 10.1016/j.electacta.2018.12.121 [5] 张林, 冯江涛, 王宁, 等. 甘氨酸改性TiO2材料的合成及其对染料的吸附性能 [J]. 环境化学, 2018, 37(12): 2621-2629. doi: 10.7524/j.issn.0254-6108.2018012905 ZHANG L, FENG J T, WANG N, et al. Preparation of glycine functionalized TiO2 adsorbent and its adsorption performance for organic dyes [J]. Environmental Chemistry, 2018, 37(12): 2621-2629(in Chinese). doi: 10.7524/j.issn.0254-6108.2018012905
[6] WANG Y, WANG Y, SONG X M, et al. BiOCl-based photocathode for photocatalytic fuel cell [J]. Applied Surface Science, 2020, 506: 144949. doi: 10.1016/j.apsusc.2019.144949 [7] RABÉ K, LIU L, NAHYOON N A, et al. Visible-light photocatalytic fuel cell with Z-scheme g-C3N4/Fe0/TiO2 anode and WO3 cathode efficiently degrades berberine chloride and stably generates electricity [J]. Separation and Purification Technology, 2019, 212: 774-782. doi: 10.1016/j.seppur.2018.11.089 [8] LIU N, HAN M, SUN Y, et al. A g-C3N4 based photoelectrochemical cell using O2/H2O redox couples [J]. Energy & Environmental Science, 2018, 11(7): 1841-1847. [9] BAI Y, YANG P, WANG L, et al. Ultrathin Bi4O5Br2 nanosheets for selective photocatalytic CO2 conversion into CO [J]. Chemical Engineering Journal, 2019, 360: 473-482. doi: 10.1016/j.cej.2018.12.008 [10] WANG H-N, CHEN X, CHEN R, et al. A ternary hybrid CuS/Cu2O/Cu nanowired photocathode for photocatalytic fuel cell [J]. Journal of Power Sources, 2019, 435: 226766. doi: 10.1016/j.jpowsour.2019.226766 [11] LI M, LIU Y, DONG L, et al. Recent advances on photocatalytic fuel cell for environmental applications-The marriage of photocatalysis and fuel cells [J]. Science of Total Environment, 2019, 668: 966-978. doi: 10.1016/j.scitotenv.2019.03.071 [12] 吴斌, 方艳芬, 任慧君, 等. g-C3N4光催化降解2, 4-DCP的活性及机理 [J]. 环境化学, 2017, 36(7): 1484-1491. doi: 10.7524/j.issn.0254-6108.2017.07.2016102508 WU B, FANG Y F, REN H J, et al. Activity and mechanism of photocatalytic degradation for 2, 4-DCP over g-C3N4 [J]. Environmental Chemistry, 2017, 36(7): 1484-1491(in Chinese). doi: 10.7524/j.issn.0254-6108.2017.07.2016102508
[13] 张聪, 米屹东, 马东, 等. CeO2/g-C3N4光催化剂的制备及性能 [J]. 环境化学, 2017, 36(1): 147-152. doi: 10.7524/j.issn.0254-6108.2017.01.2016051706 ZHANG C, MI Y D, MA D, et al. Preparation and photocatalytic performance of CeO2/g-C3N4 photocatalysts [J]. Environmental Chemistry, 2017, 36(1): 147-152(in Chinese). doi: 10.7524/j.issn.0254-6108.2017.01.2016051706
[14] LAN M, FAN G, YANG L, et al. Enhanced visible-light-induced photocatalytic performance of a novel ternary semiconductor coupling system based on hybrid Zn-in mixed metal oxide/g-C3N4 composites [J]. RSC Advances, 2015, 5(8): 5725-5734. doi: 10.1039/C4RA07073A [15] ZHAO L, WANG L, YU P, et al. A Chromium Nitride/carbon nitride containing graphitic carbon nanocapsule hybrid as a pt-free electrocatalyst for oxygen reduction [J]. Chemical Communications, 2015, 51(62): 12399-12402. doi: 10.1039/C5CC04482K [16] LUO W B, CHOU S L, WANG J Z, et al. A metal-free, free-standing, macroporous graphene@ g-c3n4 composite air electrode for high-energy lithium oxygen batteries [J]. Small, 2015, 11(23): 2817-2824. doi: 10.1002/smll.201403535 [17] GAI P, SONG R, ZHU C, et al. A ternary hybrid of carbon nanotubes/graphitic carbon nitride nanosheets/gold nanoparticles used as robust substrate electrodes in enzyme biofuel cells [J]. Chemical Communications, 2015, 51(79): 14735-14738. doi: 10.1039/C5CC06062A [18] HAN D, MING W, XU H, et al. Chemical trend of transition-metal doping in WSe2 [J]. Physical Review Applied, 2019, 12(3): 034038. doi: 10.1103/PhysRevApplied.12.034038 [19] YIN C, WANG X, CHEN Y, et al. A ferroelectric relaxor polymer-enhanced p-type WSe2 transistor [J]. Nanoscale, 2018, 10(4): 1727-1734. doi: 10.1039/C7NR08034D [20] WU J M, SUN Y-G, CHANG W E, et al. Piezoelectricity induced water splitting and formation of hydroxyl radical from active edge sites of MoS2 nanoflowers [J]. Nano Energy, 2018, 46: 372-382. doi: 10.1016/j.nanoen.2018.02.010 [21] XUE H, DAI Y, KIM W, et al. High photoresponsivity and broadband photodetection with a band-engineered WSe2/SnSe2 heterostructure [J]. Nanoscale, 2019, 11(7): 3240-3247. doi: 10.1039/C8NR09248F [22] 齐中, 王熙, 李来胜, 等. 基于水热法制备的TiO2 /MoS2复合光催化剂及其光催化制氢活性 [J]. 环境化学, 2016, 35(5): 1027-1034. doi: 10.7524/j.issn.0254-6108.2016.05.2015112403 QI Z, WANG X, LI L, et al. Development of TiO2 /MoS2 by hydrothermal method for photocatalytic hydrogen generation under solar light [J]. Environmental Chemistry, 2016, 35(5): 1027-1034(in Chinese). doi: 10.7524/j.issn.0254-6108.2016.05.2015112403
[23] TEICH J, DVIR R, HENNING A, et al. Light and complex 3D MoS2/graphene heterostructures as efficient catalysts for the hydrogen evolution reaction [J]. Nanoscale, 2020, 12(4): 2715-2725. doi: 10.1039/C9NR09564K [24] MUKHERJEE S, BISWAS S, DAS S, et al. Solution-processed, hybrid 2D/3D MoS2/Si heterostructures with superior junction characteristics [J]. Nanotechnology, 2017, 28(13): 135203. doi: 10.1088/1361-6528/aa5e42 [25] AGARWAL V, VARGHESE N, DASGUPTA S, et al. Engineering a 3D MoS2 foam using keratin exfoliated nanosheets [J]. Chemical Engineering Journal, 2019, 374: 254-262. doi: 10.1016/j.cej.2019.05.185 [26] ANWER S, HUANG Y, LI B, et al. Nature-Inspired, Graphene-Wrapped 3D MoS2 Ultrathin Microflower Architecture as a High-Performance Anode Material for Sodium-Ion Batteries [J]. ACS Applied Materials and Interfaces, 2019, 11(25): 22323-22331. doi: 10.1021/acsami.9b04260 [27] CHENG R, LI D, ZHOU H, et al. Electroluminescence and photocurrent generation from atomically sharp WSe2/MoS2 heterojunction p-n diodes [J]. Nano Letters, 2014, 14(10): 5590-5597. doi: 10.1021/nl502075n [28] PESCI F M, SOKOLIKOVA M S, GROTTA C, et al. MoS2/WS2 heterojunction for photoelectrochemical water oxidation [J]. ACS Catalysis, 2017, 7(8): 4990-4998. doi: 10.1021/acscatal.7b01517 [29] WU J M, CHANG W E, CHANG Y T, et al. Piezo-catalytic effect on the enhancement of the ultra-high degradation activity in the dark by single- and few-layers MoS2 nanoflowers [J]. Advanced Materials, 2016, 28(19): 3718-3725. doi: 10.1002/adma.201505785 [30] LIU W, WANG M, XU C, et al. Facile synthesis of g-C3N4/ZnO composite with enhanced visible light photooxidation and photoreduction properties [J]. Chemical Engineering Journal, 2012, 209: 386-393. doi: 10.1016/j.cej.2012.08.033 [31] SI K, MA J, LU C, et al. A two-dimensional MoS2/WSe2 van der Waals heterostructure for enhanced photoelectric performance [J]. Applied Surface Science, 2020, 507: 145082. doi: 10.1016/j.apsusc.2019.145082 -




 下载:
下载: