-
随着城市化进程的加快和现代工业的迅速发展,大量有机污染物进入到自然水体造成严重污染[1-2]. 这些污染物具有持久性、生物富集性、长距离迁移性及生态毒性等特征,对人民身体健康和生态环境造成巨大的危害. “绿水青山就是金山银山”,开发一种高效的污水处理技术在环保问题愈加受重视的今天显得更加紧迫. 高级氧化技术被认为是水处理中最有应用前景的绿色处理工艺[3-4],其能够将有机污染物有效氧化去除. 作为高级氧化技术的一种,芬顿及类芬顿氧化技术通过催化剂活化H2O2产生强氧化性的羟基自由基(·OH),其标准氧化还原电势达+2.8 V,在自然界中氧化能力仅次于F2,具有对大多数有机物广谱氧化效果,可将污染物分子逐渐降解成小分子产物,甚至彻底转化为H2O、CO2等,达到完全矿化的目标[5-8].
类芬顿氧化技术的关键在于催化剂. 经典芬顿技术以Fe2+作为催化剂,Fe2+具有还原特性,提供电子给H2O2产生·OH,而生成的Fe3+可再被H2O2还原为Fe2+. 即在经典芬顿反应中,整个过程依赖外界添加大量可溶亚铁盐驱动,而在反应后期常因为Fe3+过度累积引起“铁泥”现象,带来二次污染. 随后研究人员基于经典芬顿反应,开发了多种异相催化材料活化H2O2产生·OH等氧化性自由基,一定程度改善了经典芬顿反应的不足. 在众多类芬顿催化剂中[9],以零价铁(zero valance iron,ZVI)为代表的零价金属基催化材料具有较高的活化H2O2潜力,一方面能够降低类芬顿降解体系中的催化剂用量,另一方面可发挥吸附、絮凝等作用联合治理污水问题,在水处理领域被广泛关注.
本文阐述了芬顿及类芬顿技术的基本原理及存在的问题,总结了基于零价金属的类芬顿催化剂的反应特点及发展现状,以期对后续高效催化剂设计和水环境修复提供科学依据.
-
经典芬顿反应是指利用Fe2+激活H2O2,并通过一系列化学反应产生·OH的过程[10],最早由英国化学家芬顿于1893年发现,因此Fe2+与H2O2的组合又被成为芬顿试剂. 经典芬顿反应过程如反应式(1)—(3)所示,Fe2+与H2O2反应生成·OH和Fe3+,此后Fe3+又与H2O2反应生成·O2H等,铁物种在二价态和三价态循环转变,不断催化H2O2产生各种氧化自由基. 由于·OH具有强氧化性,在一定条件下可将众多有机化合物彻底氧化为无机态,在污染物降解过程占据着主导地位,故反应(1)又被认为芬顿氧化的主反应. 另外,反应(1)的标准速率常数要远远高于反应(2),即在实际反应中Fe3+的生成速率要远高于其被还原为Fe2+的速率,这是导致反应后期Fe3+不断积累的主要原因.
尽管经典芬顿反应工艺优势明显,但其存在适用pH范围较窄(pH 2—3)、H2O2利用效率低、稳定性差等缺陷. 在芬顿氧化降解体系中,有机污染物的降解效率与反应体系pH十分敏感:当pH过低时,H2O2则容易质子化形成H3O2+,无法被铁物种催化产生·OH,严重影响了有机物的氧化去除;而pH过高时,铁物种会沉淀导致产生“铁泥”,造成二次污染,同时铁物种脱离降解体系,也大大降低了对H2O2的活化能力. 另外,经典芬顿反应中,铁物种的循环步骤主要依赖反应(2),这导致了有相当数量的H2O2未被有效催化为·OH,而该反应产生的·O2H又会与有机污染物竞争·OH的反应(反应(3)),导致整个芬顿体系降解污染物能力下降[10],循环应用效果差.
-
为弥补经典均相芬顿反应的不足,研究人员在经典芬顿反应上做出了许多改进,开发出了类芬顿氧化技术. 类芬顿反应指的能替代Fe2+活化H2O2产生·OH等活性氧物种的一类技术的总称[11]. 例如铁粉、磁铁矿、氧化铁等与H2O2发生反应产生·OH,将主要降解污染物的场所从溶液均相转移到固液界面,有效降低了铁泥产生[12],同时提高了降解体系的循环稳定性;铜基催化材料能够在较高pH条件下对H2O2实现有效活化,从而将降解污染物所需的pH从酸性扩展到了中性甚至碱性环境,避免了经典芬顿反应添加酸液预调pH,一定程度上简化了污水处理环节.
此外,学者们还开发了利用以过硫酸盐替代H2O2的类芬顿技术体系,通过产生SO4·-或·OH来降解有机污染物. 过硫酸盐与H2O2类似,可被视为后者的衍生物(即H2O2的H原子被硫酸根基团取代). 过硫酸盐类芬顿系统涉及的氧化路径较多,产生的SO4·-一方面会直接降解有机污染物,另一方面可在一定条件下转换为·OH. 由于SO4·-自身拥有很强的氧化性,且在溶液中的寿命较长,能够有效提高类芬顿反应的适用pH范围(pH 2—12). 过硫酸盐类芬顿技术体系同时存在的SO4·-和·OH等氧化性自由基,对不同污染物的降解贡献存在差异,相关氧化路径有待进一步深入研究.
-
近年来,类芬顿氧化技术研究主要集中在研发高效能催化剂. 常见的催化材料包括Fe、Cu、Co、Mn等过渡金属及其氧化物、还有基于以上活性物种的负载型催化材料等. 其中,零价金属类芬顿催化剂整体价态较低,具有较强的电子供给能力,从理论上可活化H2O2或过硫酸盐产生更多的强氧化性自由基,在提升氧化媒介利用效率和污水治理前景极具优势,成为近年来的研究热点.
-
ZVI是研究最广泛的金属基类芬顿催化材料,其电子供给能力突出,内部Fe0会直接供给电子给H2O2裂解活化产生·OH[13-14],或ZVI表面在自然环境中发生氧化形成≡Fe2+,进而与H2O2反应产生·OH. 在经典芬顿反应中,高催化活性Fe2+大量转换为Fe3+,是导致降解性能下降的重要原因,而对于ZVI基催化材料,表面产生的≡Fe3+会与内部的≡Fe0发生归一反应生成更多量的≡Fe2+. 这样的链式反应一方面减少了H2O2的无效分解,提升了H2O2的利用率,另一方面加速了Fe2+/Fe3+的转化,进一步增强了催化活性和循环稳定性[15]. 零价铁基类芬顿体系所涉及的反应见(4)—(8).
-
ZVI具有廉价易得、活化位点丰富、易操作、无二次污染等优势,被广泛应用于难降解污水的处理. 1996年,美国俄勒冈科技研究生院Johnson团队在研究三氯乙烯等卤代烃降解时首先发现了铁粉具有氧化能力[16]. 后续研究证明,铁粉的氧化能力来源于≡Fe0与空气中的O2反应生成Fe2+和H2O2(反应式(9)),随后Fe2+或Fe0与H2O2进一步反应生成了·OH等活性氧自由基. 不过,这种由ZVI产生原位H2O2量相对较少,通过外源添加H2O2构成ZVI/H2O2体系,便可大大增强对有机污染物的降解能力. 都灵大学Minella等[17]在合成实验室系统中使用ZVI/H2O2类芬顿体系降解含布洛芬废水时,通过多次添加H2O2的方法,抵消了由于有机干扰剂和无机干扰剂出现所引起的布洛芬降解效率降低的情况,表现出比Fe2+/H2O2体系更优的稳定性. 此外,研究还发现ZVI/H2O2体系能够在pH = 3—5范围有效降解布洛芬废水,尤其是在较高pH值时比均相芬顿体系的降解活性更高.
ZVI除了类芬顿降解污染物以外,还可通过吸附、离子交换、氧化还原、共沉淀、络合等作用去除污染物质[18-20],即在实际应用中,ZVI能够发挥多种协同作用来达到污水治理的目标. 韩国光州科学技术院Qasim等[21]利用ZVI降解甲基汞废水,发现ZVI首先通过类芬顿反应等产生活性氧自由基(·OH等),将甲基汞快速去甲基化形成Hg2+,随后ZVI再利用还原作用使Hg2+转化为Hg0从而脱离水溶液体系,从而实现对含甲基汞废水的净化治理. Xue等[22]研究ZVI/H2O2体系同时去除含苯胺、Sb和Cr3种污染物复杂废水的能力. 处理过程中,ZVI类芬顿反应产生的·OH对苯胺有氧化降解作用,而废水中Sb和Cr去除贡献来源于ZVI表面纳米级铁(氢)氧化物吸附作用. 这些研究突出展现了ZVI在环境修复领域的独特优势,丰富了ZVI基类芬顿氧化技术的理论体系,尤其是在含多种类型污染物的污水治理上比经典芬顿体系具有不可比拟的优势.
-
由于ZVI在环境治理领域的重要作用,通过低成本的原料和手段制备高效ZVI基类芬顿催化材料具有重要意义. 常见的ZVI材料包括铁屑、铁粉、微米零价铁、纳米零价铁(nano zero valence iron,nZVI)等,对应的材料尺寸依次降低,而比表面积随之升高. 对于类芬顿催化反应而言,材料的催化活性与比表面积呈现正相关特征,即一般情况下,催化材料的尺寸越小,对应的催化活性也越高. nZVI的粒径介于10—100 nm之间,其体积小、比表面积大,与块状或微尺度铁材料相比,理论反应速率更高[23-24],是环境保护材料的前沿研究领域之一.
一般ZVI材料可通过常规方法如球磨法、还原法、雾化法等制得. 但nZVI的制备方法相对复杂,根据已有报道研究可分为“自上而下”和“自下而上”. 表1列举了ZVI材料的制备方法[17, 21, 25-27],球磨法从大尺寸原料出发,通过物理方式使其不断细化,粒径逐渐降低直至纳米级;化学还原、电化学沉积、碳还原法等方法从铁离子状态出发,通过成核和生长历程形成nZVI. 其中,利用可再生植物或废旧塑料等为原料绿色合成ZVI受到广泛关注. 巴西米纳斯联邦大学Puiatti课题组[26]用巨桉叶提取物作为还原剂得到了近似球形且尺寸在50—500 nm之间的nZVI,可在pH 4—5.5的微酸环境下高效去除偶氮染料. Sun等[28]通过热解废旧轮胎获得炭黑,以此成功地将赤泥碳热还原合成了nZVI基复合催化剂. 绿色合成方法避免了高成本的还原剂使用、反应条件相对温和,不会对环境造成新负担,是未来ZVI材料制备的重要发展方向.
-
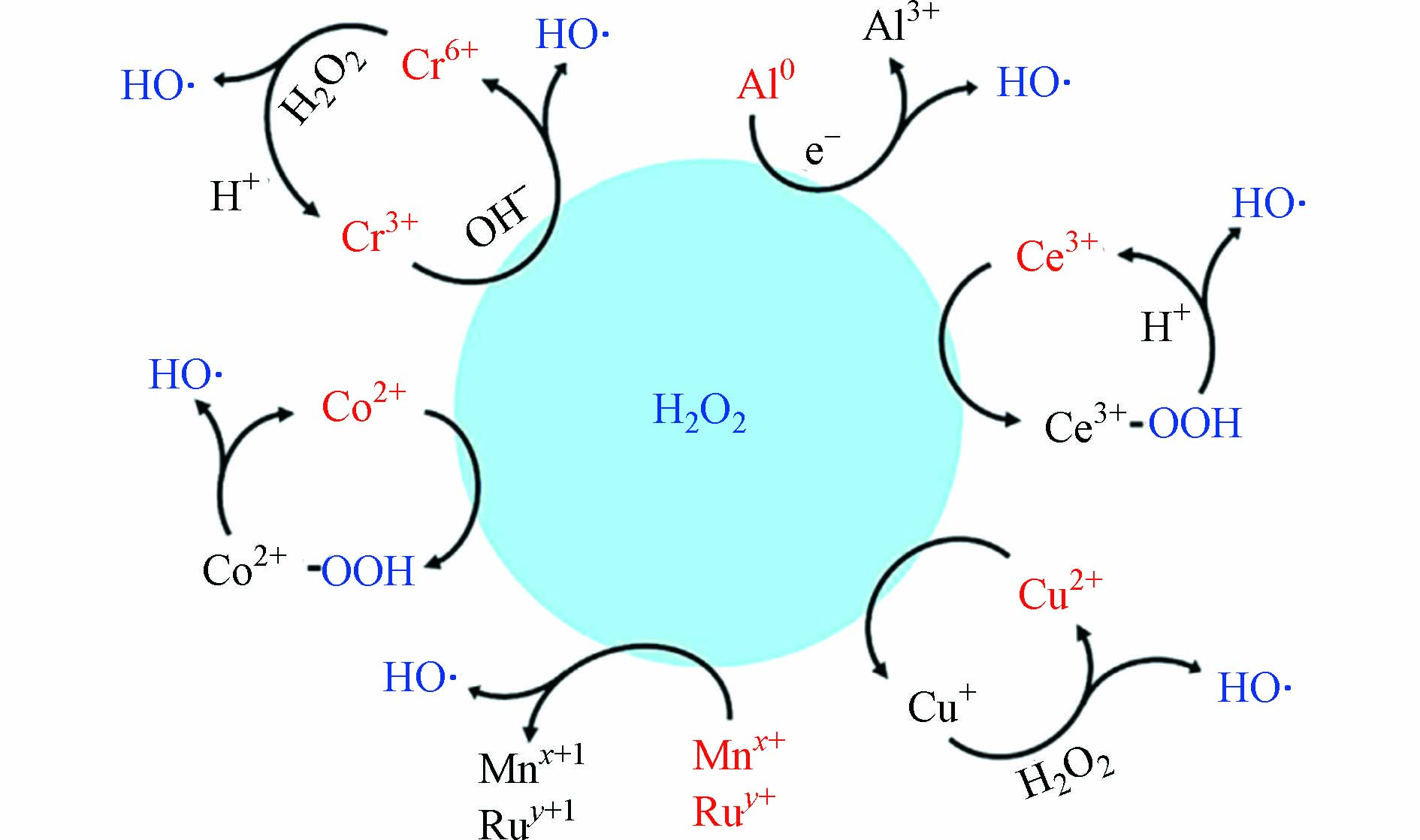
一些金属元素拥有d轨道电子或者空d轨道,d轨道电子易失去或空轨道易夺取电子,表现出较强的氧化还原性能,可拥有多个氧化还原价态. 这些变价元素如Cu、Co、Mn、Ru、Ce、Al等可替代铁参与芬顿反应,活化H2O2或过硫酸盐等产生·OH等强氧化性自由基[10, 29]. 图1为常见金属元素活化H2O2产生·OH的反应机理图. 其中,Cu是除了ZVI之外研究最多的零价态金属催化材料,Co和Mn次之;零价铝具有一定活化H2O2的能力,虽然Al3+无法构成氧化还原离子对,但其庞大的电子供给潜力也使其在类芬顿污水处理拥有一席之地;Cr、Ru和Ce在零价态没有类芬顿催化活性,其常以氧化态或与其他元素复合组成类芬顿催化剂. 因此,除Fe以外的零价金属基类芬顿研究主要集中在Cu、Co、Al、Mn等,以及各元素构成的合金类催化材料. 以上零价金属催化材料的制备方法有球磨法[30-31]、化学还原法[32]、氢热还原法[33]、溶剂热法[34]和高温碳化法[35]等.
-
自然界中的铜元素常见价态有3种,即Cu0,Cu+和Cu2+. 在与铜元素发生反应时,H2O2既可作为还原剂,又可作为氧化剂. 具体而言,Cu0和Cu+提供电子给H2O2,促使H2O2裂解产生·OH,而Cu0和Cu+转化为高价态的Cu2+,该过程与铁基芬顿反应过程类似;当与Cu2+反应时,H2O2提供电子将Cu2+还原为Cu+,并生成·O2H,该过程要比Fe3+/H2O2反应速度快得多. 由于Cu2+/Cu+的循环速率远较Fe3+/Fe+速率高,且Cu(OH)2溶度积常数为2.2×10-22,远高于Fe(OH)3的溶度积常数4.0×10-38,故铜基芬顿在表现出良好的反应活性之外,还具有比铁基芬顿更宽的pH适用范围. 在实际应用中,Cu0,Cu+和Cu2+均可作为催化材料与H2O2构成类芬顿降解体系,其中以Cu+的活性最高[36].
近年来,铜基类芬顿体系在基本机理和实际应用方面都取得了迅速的进展. 与ZVI材料类似,零价铜材料拥有较大的电子供给能力,被认为是非常有前途的类芬顿催化材料. Li等[34]采用简单的溶剂热法制备了一种具有磁性的Cu/CuFe2O4纳米复合材料用来降解亚甲基蓝(MB). 通过改变Cu/CuFe2O4复合材料中Cu0的含量,发现Cu0含量与催化活性呈现正相关特征,并证明了CuFe2O4与Cu0间的协同效应. Yu等[35]利用一种简单的原位碳化方法合成了氮掺杂生物碳包覆的零价铜纳米粒子,形成的零价铜纳米颗粒均匀地分散在生物碳中,这些碳层能对内部包裹的零价铜纳米颗粒起到保护作用,赋予了催化剂良好的环境稳定性,在暴露于空气中4个月后,其催化能力基本不变.
-
Co元素与H2O2的反应活性不高,但对于过硫酸盐活化有着更高的催化性能. 在常见的钴物种中,CoO和Co3O4拥有更低的化学价态和还原潜力,常用于活化过硫酸盐以降解各种污染物[37-38]. 由于零价钴的成本较高,且直接应用于水体易造成重金属离子污染,对人体健康构成潜在危险,阻碍了其应用. 中国环境科学研究院李君超等[39]制备了纳米Fe/Co合金类芬顿催化材料,发现Fe/Co合金化有效拓宽降解体系的pH使用范围,能够在pH 3.0—9.0对盐酸四环素进行有效降解,且长期使用后也不会造成重金属离子在环境中过度累积.
自然界的Mn元素价态丰富,对环境的毒性小于Co,拥有从0至+7多种价态,具备活化H2O2或过硫酸盐的能力. 零价锰还原电势为-1.18 V,因此被广泛应用于高级氧化技术以去除持久性污染物. Shah等[32]将Mn0/过硫酸盐体系降解盐酸环丙沙星(CIP)研究,CIP降解率要高于单独Mn0,这是因为Mn0活化过硫酸盐产生了SO4-·矿化了部分CIP,从而总体降解效果要高于单独Mn0的还原吸附作用. 此外,Mn0表面容易氧化形成MnOx@Mn0核壳结构,相比锰氧化物具有更持久的催化活性.
Al化学性质活泼,具有比Fe、Co、Cu等更强的还原特性,单个原子可提供3个电子活化H2O2或过硫酸盐,能够产生更加丰富的活性氧自由基. 在实际应用中,零价铝的催化活性容易受到其表面致密稳定的氧化层的限制,导致类芬顿降解污染物效率下降. 中国海洋大学Yang等[30]采取NaCl辅助球磨法处理零价铝,一方面NaCl以离子态进入零价铝的外氧化层,促进层间电子转移,另一方面通过球磨方式将零价铁不断剥离出新鲜界面,大大增强了零价铝的催化活性和长期稳定性.
-
此外,不同金属元素还可组成合金基类芬顿催化材料,发挥相互之间的协同作用,以满足日益复杂的水污染治理需求. 王斌等[40]采用溶剂热法分别合成了V掺杂Cu、Fe的双金属催化剂,并研究了二者的非均相芬顿降解亚甲基蓝的性能,结果表明Cu-V复合材料在pH 10时对亚甲基蓝的去除率最高,而Fe-V复合材料在pH 3时对亚甲基蓝的去除率最高. 清华大学王建龙教授团队开发了Ce0-Fe0-石墨烯复合材料,在类芬顿降解磺胺二甲嘧啶时,Ce0构成的Ce3+/Ce4+离子对能够加速Fe2+/Fe3+的循环再生,避免了低活性铁物种的生成积累,增加了催化剂表明的有效催化位点,整体的循环降解效率比单独的Fe0提升了2—3倍. 另外,石墨烯载体有效阻止了零价金属颗粒的聚集长大,对催化稳定性也有较大的贡献[41].
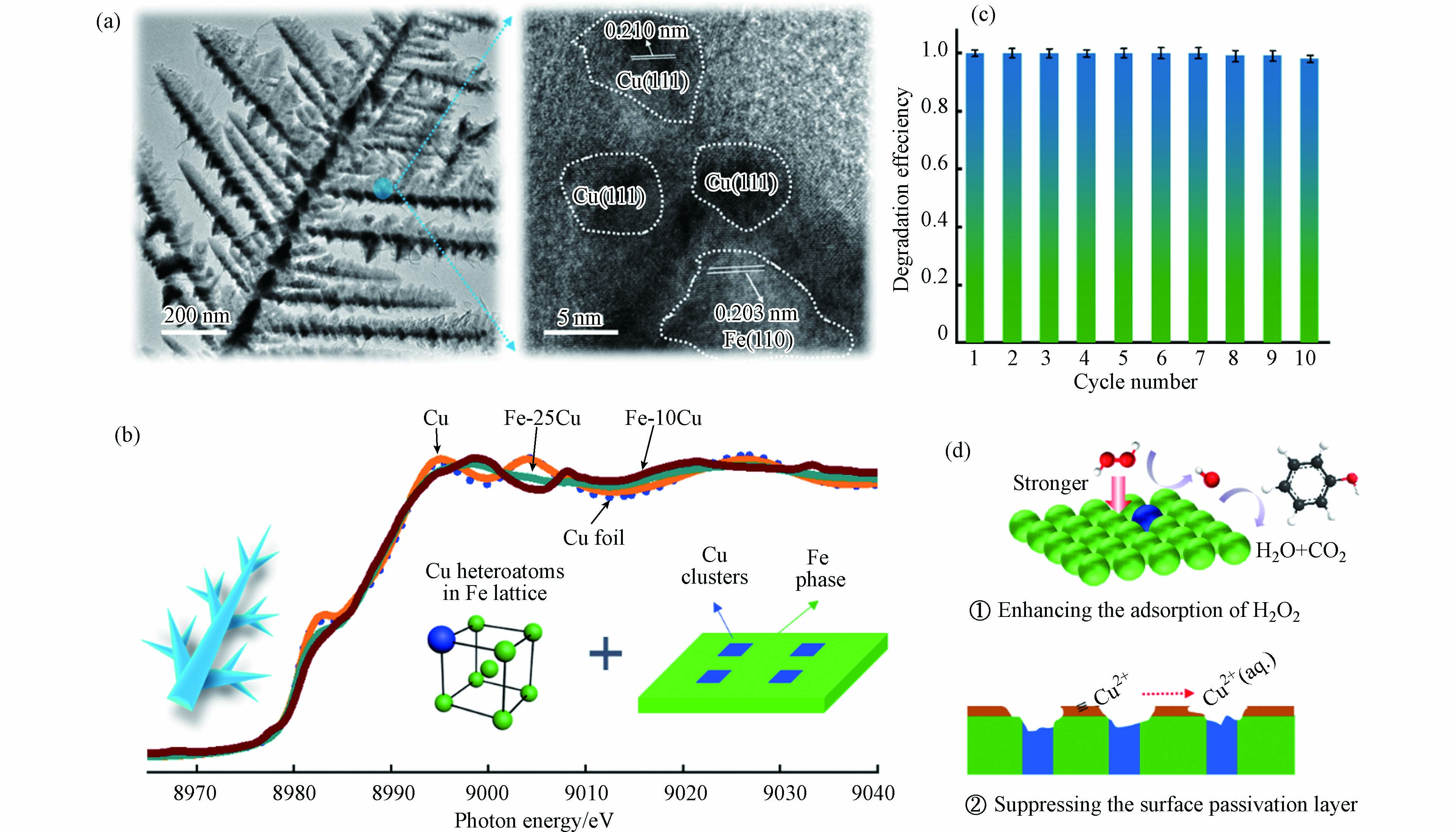
本课题组制备了一系列的Fe-Cu合金类芬顿催化材料[42-43],所制备的催化材料具有微纳分等级树枝状结构,其中的Fe微晶和Cu微晶混合排列,并探讨了Cu杂原子和Cu团簇对类芬顿反应的增强机制. 我们发现尽管Fe和Cu都可以活化H2O2产生·OH,但铁基芬顿占主导作用. 除了Cu+/Cu2+循环和Fe3+/Fe2+循环的协同作用之外,Cu组元根据存在形式的不同具备差异化作用:Cu杂原子增强了相邻Fe原子对H2O2的吸附能力,而独立Cu团簇可有效抑制催化剂表面钝化,提高Fe-Cu合金的可重复使用性(图2). 另外,课题组通过控制氧化法获得了外壳为Cu/Fe3O4而核心为FeCu的复合催化剂,苯酚降解实验证明,酸性条件下为铁物种为活性中心,中性条件下铜物种为活性中心. 这种设计思路有效地综合了Fe与Cu各自的优点,明确了零价金属合金基催化剂各组元之间组织、性能的关系,为高效类芬顿催化剂的设计提供了有益借鉴和思路.
-
由于零价金属基材料为固体催化剂,而基于此所开发的类芬顿氧化技术,就由原来经典芬顿反应的单一均相催化过程转变为异相催化过程. 也就是说,在实际应用中,零价金属基类芬顿氧化技术的降解效率不仅受活性位点与氧化媒介反应产生·OH步骤的影响,同时也涉及了氧化媒介的吸附、氧化性自由基的转移、污染物吸附与降解产物脱附、活性位点再生等步骤. 为进一步提升零价金属材料的类芬顿氧化效果,研究人员寻求多种方法来改良异相催化过程的速度控制步骤反应,以增强零价金属基催化材料对有机污染物的类芬顿降解能力. 目前,关于零价金属基催化材料的性能提升策略可分为3类:助催化剂策略,负载型材料改良策略和外场辅助策略.
-
助催化剂一般是富电子材料,能够对外提供电子,促进过渡金属物种由高价态向低价态转化,进一步改善异相类芬顿反应过程中的活性位点再生、自由基产生等步骤,增强有机污染物降解效果. 目前研究较多的助催化剂有羟胺、抗坏血酸、MoS2等. 华南师范大学方战强课题组系统研究了茶多酚、草酸、盐酸羟胺等对ZVI/H2O2体系类芬顿降解抗生素的效果[44],发现有机助催化剂均能有效提升体系降解能力2—5倍. 其中,茶多酚能够通过还原和螯合作用促进体系产生·OH的速率,而草酸、盐酸羟胺主要通过降低pH和增强均相/异相协同效果加速了抗生素的氧化分解. MoS2表面存着还原态金属活性中心,可增强主催化剂对于H2O2及过硫酸盐活化效果. 四川师范大学Yang等[45]发现,MoS2辅助ZVI/H2O2体系,能有效去除垃圾渗滤液中的难降解有机物(如黄腐酸和腐殖质类物质),降解速率常数比单一ZVI/H2O2体系提升50%以上. 通过活性氧自由基检测、·OH浓度测定、H2O2利用率等综合分析,证明MoS2作为电子供体,有效增强了Fe3+/Fe2+循环,从而大大提高降解有机污染物的能力. 需要指出的是,外源助催化剂持续性效果较差,实际应用中需不断添加;另外,助催化剂在改善类芬顿降解过程中,有可能增加体系的有机负荷或金属离子载量,从而影响整体水体净化进程.
-
外场辅助策略主要包括光[46]、电[47]、微波[48]、超声[49]等,而根据施加外场的不同,又可称之为光芬顿、电芬顿、微波芬顿、超声芬顿等. 由于外界能量的输入,类芬顿降解过程的异相催化步骤被大大增强,如光场可促进H2O2活化效率,改善活性位点的再生;电场能够直接在体系原位产生H2O2,扩展了类芬顿反应适用条件,并加速了金属元素的高低价态转化;微波芬顿和超声芬顿等不仅为氧化媒介的活化步骤供能,同时有利于提升反应物传质效率. 吉林大学Liu等[50]研究发现,将太阳光作为光场辅助芬顿体系的光源处理废水可以使水中·OH浓度升高,从而提高类芬顿反应的效率,这要归因于光场提供能量加速了H2O2的活化效率,拓展了·OH生成的反应途径. 中国科学院城市环境研究所的Chen等[51]在静电纺丝三维纳米纤维网络上用原位合成法合成了一种高活性的电芬顿催化剂——海棉状零价铁纳米颗粒. 海绵状ZVI有效地克服了电催化过程中nZVI容易聚集的缺点,电场持续功能促进了Fe3+转化为Fe2+,避免形成“铁泥”脱离降解体系. 在电场辅助芬顿体系中,ZVI产生·OH使磺胺塞唑的去除率在5 min内达到100%,远高于无电场条件下的去除率(50%). 外场辅助策略不会带来新的有机负荷,降低了催化剂的用量,在时效上具有突出优势,但其需要外界能量持续输入,会不可避免增加污水治理的成本.
-
负载型材料是指具有较大比表面积、高孔隙率或官能团的一类材料,可作为芬顿催化剂的载体. 大比表面积和高孔隙率特性有利于吸附反应媒介和污染物,缩短反应物扩散距离,并为氧化媒介活化和污染物氧化提供反应场所;而载体上的特殊官能团可选择性吸附部分反应物,从而增强特定的氧化降解进程. 大连海事大学Sun[52]等将nZVI固定在碳纳米管薄膜(CNT)中形成Fe0-CNT催化膜,利用膜孔内的液体对流提高传质效果,将对目标污染物降解效率提升了3倍,并对外界因素(如Cl-、HCO3-、NO3-、天然有机物等)表现出良好的抗干扰性. 本团队制备了碳量子点修饰ZVI复合催化材料,其中碳量子点表面富含极性官能团,有效增强了对苯酚的吸附作用,同时碳量子点亦可为铁物种循环提供电子,相比于ZVI材料将pH适用范围由酸性(pH<4)提升至中性条件[53],降解速率增强了2倍以上.
近年来,金属-有机骨架材料(metal-organic frameworks,MOFs)、共价有机骨架材料(covalent organic frameworks,COFs)和MXene材料等被广泛应用于高效类芬顿催化剂的设计,是近年来水处理领域的前沿热点之一. MOFs和COFs具有大比表面积,规整的孔道,易于调控的结构,在改良类芬顿金属位点催化、反应物扩散与吸附等多相催化降解有机污染有着突出优势;MXene是一类二维无机化合物,不仅能够为反应物迁移提供多个通道,同时活性钛组分可加速类芬顿反应中的电子转移. 华东师范大学余承忠团队将Cu/Co纳米合金颗粒装配纳米碳骨架材料[54](即Cu/Co-MOFs衍生材料),发现Cu和Co组元间协同作用有效促进类芬顿活性位点再生,且碳骨架具有的高比表面积提高了合金纳米例子的分散性和稳定性,促进了异相芬顿中的传质效率,连续5次循环降解实验仍能保持90%以上的催化活性. 中国环境科学研究院赵晓丽课题组[55]开发了磁性COFs类芬顿催化材料,该催化剂实现了Fe活性物种与框架结构的合理协调,降解效率是Fe3O4/H2O2体系的3—5倍;华北电力大学郝润龙课题组[56]制备了铁基MXene类芬顿复合催化剂,能够同时处理含亚硝酸钠和Hg废水,这归功于Ti3+/Ti2+加速了活性物种Fe2+的再生和二维载体赋予的优异吸附性能,综合降解效率增加了80%以上. 负载型改良策略直击催化材料自身,又可与另外两种辅助策略联合应用,这为设计新型类芬顿催化剂和构建高效污水处理工艺提供了新思路.
-
零价金属基类芬顿催化剂具有出色的电子供给能力,能够活化H2O2或过硫酸盐等氧化媒介产生强氧化性自由基降解有机污染物,克服了经典芬顿反应稳定性差、易产生铁泥等问题,在水体有机污染物的治理领域具有很好的应用前景. 不同零价金属基材料性质各异,如铁基材料的研究最为广泛,钴基材料活化过硫酸盐性能出色、铜基材料适用pH范围宽等,通过合金化设计和多种提升策略协同运用能进一步提升其类芬顿催化能力. 尽管零价金属类芬顿催化材料在水污染治理的研究已经取得长足进步,但从实用化发展角度出发,仍有以下关键技术和问题亟待深入探索研究.
(1)寻求零价金属基催化材料大规模制备方法. 目前,零价金属催化材料常规制备方法存在一定问题,如球磨法存在耗能高、产品均一性差等问题,化学还原法所采取的还原剂NaBH4价格过高等,距离大规模应用仍有一段距离. 因此,如何批量化制备高效催化剂,尤其是纳米级零价金属基催化材料,是下一步的重点任务.
(2)不同催化活性物种耦合作用下的多相催化机理研究. 零价金属尤其合金基类芬顿催化材料拥有多种活性物种,对氧化媒介的催化、反应物界面吸附、活性位点再生等异相步骤作用呈现各自特色. 厘清多活性物种下的多相催化机理,能够为高效类芬顿催化材料合理设计和实际应用提供理论依据.
(3)现有大多数零价金属基催化材料的应用研究局限在实验室,缺乏在实际污水治理检验. 因此,必须加强零价金属基催化材料在真实废水环境下类芬顿降解速率关键参数、有机污染物氧化/矿化主要路径、催化剂长效稳定方法等研究,这也是未来推进其实用化发展亟待解决的关键工程技术问题.
基于零价金属材料的类芬顿氧化技术研究进展
Research progress of Fenton-like oxidation processes based on zero valent metal materials
-
摘要: 芬顿与类芬顿氧化技术能够活化H2O2,产生强氧化性的羟基自由基(•OH),可对绝大多数污染物彻底氧化. 相较于其他类芬顿催化剂,以零价铁为代表的零价金属材料具有更优的理论电子供给潜力,活化H2O2能力突出,在污水治理领域被重点关注. 本文综述了近年来零价金属类芬顿催化材料的研究进展,系统分析了铁基、铜基、钴基以及合金等零价态金属的反应机理和降解特点,并从异相催化角度探讨了类芬顿氧化处理废水的作用机制和催化剂性能提升策略. 最后,对未来零价金属类芬顿催化材料的发展前景进行了展望.Abstract: Fenton and Fenton-like oxidation technologies can activate H2O2 and produce strong oxidizing hydroxyl radicals (•OH) for completely oxidizing most pollutants. Compared with other Fenton-like catalysts, zero-valent metal materials represented by zero-valent iron have better theoretical electron supply capacity to activate H2O2, which have attracted much attention in the field of water purification. This paper reviews the research progress of zero-valent metal Fenton-like catalysts in recent years, and analyzes systematically the reaction mechanisms and degradation characteristics of zero-valent metals such as Fe-based, Cu-based, Co-based and alloy materials. Furthermore, the effect behaviors and improvement strategy of Fenton-like oxidation for wastewater treatment are discussed from the perspective of heterogeneous catalysis processes. At last, the future development of zero-valent metals Fenton-like catalytic materials is prospected.
-
Key words:
- zero valent metals /
- Fenton reaction /
- heterogeneous catalysis /
- ·OH
-

-
图 2 微纳枝状Fe-Cu合金的微观结构(a)、类芬顿循环降解苯酚实验(b),Fe-Cu合金中铜杂质原子和铜原子簇的存在形式(c)及类芬顿反应机制(d)[42]
Figure 2. Microstructure of micro-nano dendritic Fe-Cu alloy (a), Fenton-like cyclic degradation experiments of phenol (b), Existence forms of Cu impurity atoms and Cu atom clusters in Fe-Cu alloy (c) and their special Fenton-like reaction mechanism (d)[42]
表 1 ZVI材料的制备方法
Table 1. Preparation methods of ZVI materials
制备方法
Preparation methods形貌与尺寸
Morphology and sizes过程
Preparation process球磨法[27] 不规则形状,微米级 在刚性球机械作用下,大尺度ZVI材料被物理粉碎、细化直至目标尺度 化学还原法[17, 21] 近似球形、并以链式聚集,尺寸20—50 nm 搅拌条件下,NaBH4加入至Fe2+/Fe3+溶液,反应后洗涤、分离、干燥 电化学法[25] 树枝多级结构状,2—10 μm、末级尺寸40 nm 以石墨为阳极、红铜为阴极,对FeSO4溶液施加电流,在阴极形成ZVI粉末 绿色合成[26] 近似球形、易形成团聚体,尺寸50—500 nm 将经过研磨、过滤、离心得到的植物提取物加入至含铁溶液,充分搅拌反应,结束后洗涤、分离、干燥 -
[1] 阮挺, 江桂斌. 发现新型环境有机污染物的基本理论与方法 [J]. 中国科学院院刊, 2020, 35(11): 1328-1336. doi: 10.16418/j.issn.1000-3045.20200915004 RUAN T, JIANG G B. Basic theory and analytical methodology for identification of novel environmental organic pollutants [J]. Bulletin of Chinese Academy of Sciences, 2020, 35(11): 1328-1336(in Chinese). doi: 10.16418/j.issn.1000-3045.20200915004
[2] 何阳卓, 魏建宏, 周耀渝, 等. 光芬顿和电芬顿在抗生素降解方面的研究及认识 [J]. 当代化工, 2019, 48(2): 395-398. doi: 10.3969/j.issn.1671-0460.2019.02.045 HE Y Z, WEI J H, ZHOU Y Y, et al. Research and knowledge on photo-Fenton and electro-Fenton for the degradation of antibiotics [J]. Contemporary Chemical Industry, 2019, 48(2): 395-398(in Chinese). doi: 10.3969/j.issn.1671-0460.2019.02.045
[3] 王雪纯. 多孔碳催化剂的制备及其对水中有机污染物的降解研究[D]. 秦皇岛: 燕山大学, 2020. WANG X C. Preparation of porous carbon catalyst and its degradation of organic pollutants in water[D]. Qinhuangdao: Yanshan University, 2020 (in Chinese).
[4] 刘丹, 王渝, 阳明利, 等. 过氧化钙材料用于阿莫西林降解性能研究 [J]. 当代化工, 2022, 51(9): 2136-2140. LIU D, WANG Y, YANG M L, et al. Application of calcium peroxide in amoxicillin degradation [J]. Contemporary Chemical Industry, 2022, 51(9): 2136-2140(in Chinese).
[5] SZABÓ L, STEINHARDT M, HOMLOK R, et al. A microbiological assay for assessing the applicability of advanced oxidation processes for eliminating the sublethal effects of antibiotics on selection of resistant bacteria [J]. Environmental Science & Technology Letters, 2017, 4(6): 251-255. [6] HODGES B C, CATES E L, KIM J H. Challenges and prospects of advanced oxidation water treatment processes using catalytic nanomaterials [J]. Nature Nanotechnology, 2018, 13(8): 642-650. doi: 10.1038/s41565-018-0216-x [7] TUŠAR N N, MAUČEC D, RANGUS M, et al. Manganese functionalized silicate nanoparticles as a Fenton-type catalyst for water purification by advanced oxidation processes (AOP) [J]. Advanced Functional Materials, 2012, 22(4): 820-826. doi: 10.1002/adfm.201102361 [8] BOKARE A D, CHOI W. Review of iron-free Fenton-like systems for activating H2O2 in advanced oxidation processes [J]. Journal of Hazardous Materials, 2014, 275: 121-135. doi: 10.1016/j.jhazmat.2014.04.054 [9] 王爽, 谢良波, 李轶, 等. 芬顿催化剂的活性氧物种生成机制及其在环境治理中的应用 [J]. 稀有金属, 2022, 46(6): 707-723. WANG S, XIE L B, LI Y, et al. Unraveling reactive oxygen species formation mechanism of Fenton catalyst and its application in environmental treatment [J]. Chinese Journal of Rare Metals, 2022, 46(6): 707-723(in Chinese).
[10] 侯琳萌, 清华, 吉庆华. 类芬顿反应的催化剂、原理与机制研究进展 [J]. 环境化学, 2022, 41(6): 1843-1855. doi: 10.7524/j.issn.0254-6108.2021030301 HOU L M, QING H, JI Q H. Research progress on catalysts, principles and mechanisms of Fenton-like reactions [J]. Environmental Chemistry, 2022, 41(6): 1843-1855(in Chinese). doi: 10.7524/j.issn.0254-6108.2021030301
[11] 厉鹏远, 邱立平, 孙绍芳, 等. 强化传统芬顿/类芬顿氧化效能的研究进展 [J]. 中国给水排水, 2021, 37(10): 34-40. doi: 10.19853/j.zgjsps.1000-4602.2021.10.006 LI P Y, QIU L P, SUN S F, et al. Research progress on enhancing the oxidation efficiency of traditional Fenton/Fenton-like process [J]. China Water & Wastewater, 2021, 37(10): 34-40(in Chinese). doi: 10.19853/j.zgjsps.1000-4602.2021.10.006
[12] 吕来, 胡春. 多相芬顿催化水处理技术与原理 [J]. 化学进展, 2017, 29(9): 981-999. doi: 10.7536/PC170552 LYU L, HU C. Heterogeneous Fenton catalytic water treatment technology and mechanism [J]. Progress in Chemistry, 2017, 29(9): 981-999(in Chinese). doi: 10.7536/PC170552
[13] YAO H R, HU S H, WU Y G, et al. The synergetic effects in a Fenton-like system catalyzed by nano zero-valent iron (nZVI) [J]. Polish Journal of Environmental Studies, 2019, 28(4): 2491-2499. doi: 10.15244/pjoes/91939 [14] 邹亚辰, 贾小宁, 冉浪, 等. 零价铁类芬顿法处理含低浓度重金属离子有机废水 [J]. 化学反应工程与工艺, 2021, 37(2): 167-174. ZOU Y C, JIA X N, RAN L, et al. Study on the treatment of organic wastewater containing low concentration heavy metal ions by zero-valent iron Fenton-like process [J]. Chemical Reaction Engineering and Technology, 2021, 37(2): 167-174(in Chinese).
[15] LIANG W, DAI C M, ZHOU X F, et al. Application of zero-valent iron nanoparticles for the removal of aqueous zinc ions under various experimental conditions [J]. PLoS One, 2014, 9(1): e85686. doi: 10.1371/journal.pone.0085686 [16] JOHNSON T L, SCHERER M M, TRATNYEK P G. Kinetics of halogenated organic compound degradation by iron metal [J]. Environmental Science & Technology, 1996, 30(8): 2634-2640. [17] MINELLA M, BERTINETTI S, HANNA K, et al. Degradation of ibuprofen and phenol with a Fenton-like process triggered by zero-valent iron (ZVI-Fenton) [J]. Environmental Research, 2019, 179: 108750. doi: 10.1016/j.envres.2019.108750 [18] VASAREVIČIUS S, DANILA V, PALIULIS D. Application of stabilized nano zero valent iron particles for immobilization of available Cd2+, Cu2+, Ni2+, and Pb2+ ions in soil [J]. International Journal of Environmental Research, 2019, 13(3): 465-474. doi: 10.1007/s41742-019-00187-8 [19] WANG B, DENG C X, MA W, et al. Modified nanoscale zero-valent iron in persulfate activation for organic pollution remediation: A review [J]. Environmental Science and Pollution Research International, 2021, 28(26): 34229-34247. doi: 10.1007/s11356-021-13972-w [20] TOSCO T, PETRANGELI PAPINI M, CRUZ VIGGI C, et al. Nanoscale zerovalent iron particles for groundwater remediation: A review [J]. Journal of Cleaner Production, 2014, 77: 10-21. doi: 10.1016/j.jclepro.2013.12.026 [21] QASIM G H, FAREED H, LEE M, et al. Aqueous monomethylmercury degradation using nanoscale zero-valent iron through oxidative demethylation and reductive isolation [J]. Journal of Hazardous Materials, 2022, 435: 128990. doi: 10.1016/j.jhazmat.2022.128990 [22] XUE G, WANG Q, QIAN Y J, et al. Simultaneous removal of aniline, antimony and chromium by ZVI coupled with H2O2: Implication for textile wastewater treatment [J]. Journal of Hazardous Materials, 2019, 368: 840-848. doi: 10.1016/j.jhazmat.2019.02.009 [23] YIRSAW B D, MEGHARAJ M, CHEN Z L, et al. Environmental application and ecological significance of nano-zero valent iron [J]. Journal of Environmental Sciences (China), 2016, 44: 88-98. doi: 10.1016/j.jes.2015.07.016 [24] MIKHAILOV I, KOMAROV S, LEVINA V, et al. Nanosized zero-valent iron as Fenton-like reagent for ultrasonic-assisted leaching of zinc from blast furnace sludge [J]. Journal of Hazardous Materials, 2017, 321: 557-565. doi: 10.1016/j.jhazmat.2016.09.046 [25] XIA Q, JIANG Z, WANG J, et al. A facile preparation of hierarchical dendritic zero-valent iron for Fenton-like degradation of phenol [J]. Catalysis Communications, 2017, 100: 57-61. doi: 10.1016/j.catcom.2017.06.017 [26] PUIATTI G A, de CARVALHO J P, de MATOS A T, et al. Green synthesis of Fe0 nanoparticles using Eucalyptus grandis leaf extract: Characterization and application for dye degradation by a (Photo)Fenton-like process [J]. Journal of Environmental Management, 2022, 311: 114828. doi: 10.1016/j.jenvman.2022.114828 [27] 王鹏, 王义东, 柳听义. 球磨法制备纳米零价铁的研究进展 [J]. 环境化学, 2021, 40(9): 2924-2933. doi: 10.7524/j.issn.0254-6108.2020050601 WANG P, WANG Y D, LIU T Y. Research progress of preparation of nano zero-valent iron by ball milling [J]. Environmental Chemistry, 2021, 40(9): 2924-2933(in Chinese). doi: 10.7524/j.issn.0254-6108.2020050601
[28] SUN X, NI X, WANG X, et al. Preparation of zero-valent iron-based composite catalyst with red mud and scrap tire as starting materials for Fenton-like degradation of methyl blue [J]. Surfaces and Interfaces, 2022, 31: 102053. doi: 10.1016/j.surfin.2022.102053 [29] 李孟宣, 盛光遥, 何岸飞. 金属催化剂在宽泛pH进行Fenton反应的进展 [J]. 工业水处理, 2021, 41(2): 20-25. LI M X, SHENG G Y, HE A F. Progress of metal catalysts for Fenton reaction at a wide range of pH [J]. Industrial Water Treatment, 2021, 41(2): 20-25(in Chinese).
[30] WU S, YANG S, LIU S, et al. Enhanced reactivity of zero-valent aluminum with ball milling for phenol oxidative degradation [J]. Journal of Colloid and Interface Science, 2020, 560: 260-272. doi: 10.1016/j.jcis.2019.10.075 [31] WANG N. FeCoNiMnCuTi high entropy amorphous alloys and M50Ti50 (M = Fe, Cu, FeCoNiMnCu) amorphous alloys: Novel and efficient catalysts for heterogeneous photo-Fenton decomposition of Rhodamine B [J]. Surfaces and Interfaces, 2022, 33: 102265. doi: 10.1016/j.surfin.2022.102265 [32] SHAH N S, KHAN J A, SAYED M, et al. Hydroxyl and sulfate radical mediated degradation of ciprofloxacin using nano zerovalent Manganese catalyzed S2O82− [J]. Chemical Engineering Journal, 2019, 356: 199-209. doi: 10.1016/j.cej.2018.09.009 [33] ZHOU G, ZHOU R, LIU Y, et al. Efficient degradation of sulfamethoxazole using peracetic acid activated by zero-valent cobalt [J]. Journal of Environmental Chemical Engineering, 2022, 10(3): 107783. doi: 10.1016/j.jece.2022.107783 [34] LI Z L, LYU J C, GE M. Synthesis of magnetic Cu/CuFe2O4 nanocomposite as a highly efficient Fenton-like catalyst for methylene blue degradation [J]. Journal of Materials Science, 2018, 53(21): 15081-15095. doi: 10.1007/s10853-018-2699-0 [35] YU C, HUANG R, XIE Y, et al. In-situ synthesis of N-doped biochar encapsulated Cu(0) nanoparticles with excellent Fenton-like catalytic performance and good environmental stability [J]. Separation and Purification Technology, 2022, 295: 121334. doi: 10.1016/j.seppur.2022.121334 [36] SUN B F, LI H L, LI X Y, et al. Degradation of organic dyes over Fenton-like Cu2O-Cu/C catalysts [J]. Industrial & Engineering Chemistry Research, 2018, 57(42): 14011-14021. [37] DONG X, DUAN X, SUN Z, et al. Natural illite-based ultrafine cobalt oxide with abundant oxygen-vacancies for highly efficient Fenton-like catalysis [J]. Applied Catalysis B:Environmental, 2020, 261: 118214. doi: 10.1016/j.apcatb.2019.118214 [38] LI X N, HUANG X, XI S B, et al. Single cobalt atoms anchored on porous N-doped graphene with dual reaction sites for efficient Fenton-like catalysis [J]. Journal of the American Chemical Society, 2018, 140(39): 12469-12475. doi: 10.1021/jacs.8b05992 [39] 李君超, 蒋进元, 张伟, 等. 纳米Fe/Co合金类Fenton降解盐酸四环素及影响因素 [J]. 环境科学研究, 2018, 31(4): 757-764. doi: 10.13198/j.issn.1001-6929.2018.01.10 LI J C, JIANG J Y, ZHANG W, et al. Oxidative degradation of tetracycline hydrochloride using nano Fe/Co alloy and H2O2 under Fenton conditions [J]. Research of Environmental Sciences, 2018, 31(4): 757-764(in Chinese). doi: 10.13198/j.issn.1001-6929.2018.01.10
[40] 王斌, 杨月红, 阳耀熙. 钒掺杂铜基、铁基双金属催化剂的非均相芬顿氧化性能 [J]. 化工进展, 2021, 40(12): 6705-6713. doi: 10.16085/j.issn.1000-6613.2021-0082 WANG B, YANG Y H, YANG Y X. Heterogeneous Fenton oxidation performance of vanadium-doped copper-based and iron-based bimetallic catalysts [J]. Chemical Industry and Engineering Progress, 2021, 40(12): 6705-6713(in Chinese). doi: 10.16085/j.issn.1000-6613.2021-0082
[41] WAN Z, HU J, WANG J. Removal of sulfamethazine antibiotics using CeFe-graphene nanocomposite as catalyst by Fenton-like process [J]. Journal of Environmental Management, 2016, 182: 284-291. doi: 10.1016/j.jenvman.2016.07.088 [42] XIA Q, ZHANG D, YAO Z, et al. Investigation of Cu heteroatoms and Cu clusters in Fe-Cu alloy and their special effect mechanisms on the Fenton-like catalytic activity and reusability [J]. Applied Catalysis B:Environmental, 2021, 299: 120662. doi: 10.1016/j.apcatb.2021.120662 [43] XIA Q X, ZHANG D J, YAO Z P, et al. Revealing the enhancing mechanisms of Fe-Cu bimetallic catalysts for the Fenton-like degradation of phenol [J]. Chemosphere, 2022, 289: 133195. doi: 10.1016/j.chemosphere.2021.133195 [44] OUYANG Q, LIAN J T, LU B Z, et al. Effects and mechanisms of lincomycin degradation by six promoters in the mZVI/H2O2 systems [J]. Chemical Engineering Journal, 2020, 387: 123417. doi: 10.1016/j.cej.2019.123417 [45] YANG J, ZHANG X, TANG J, et al. Removal efficiency and mechanism of refractory organic matter from landfill leachate MBR effluent by the MoS2-enhanced Fe0/H2O2 system [J]. Journal of Environmental Chemical Engineering, 2022, 10(5): 108391. doi: 10.1016/j.jece.2022.108391 [46] CLARIZIA L, RUSSO D, Di SOMMA I, et al. Homogeneous photo-Fenton processes at near neutral pH: A review [J]. Applied Catalysis B:Environmental, 2017, 209: 358-371. doi: 10.1016/j.apcatb.2017.03.011 [47] 彭媛, 赵朋飞. 响应曲面法优化感应电芬顿处理活性红X-3B废水 [J]. 当代化工, 2022, 51(5): 1014-1019. doi: 10.3969/j.issn.1671-0460.2022.05.002 PENG Y, ZHAO P F. Optimization of induction electro-Fenton for treating reactive red X-3B wastewater by response surface method [J]. Contemporary Chemical Industry, 2022, 51(5): 1014-1019(in Chinese). doi: 10.3969/j.issn.1671-0460.2022.05.002
[48] 左继红, 吴冰. 微波耦合芬顿降解含酚废水反应器及工艺研究 [J]. 当代化工, 2022, 51(8): 2012-2016. doi: 10.3969/j.issn.1671-0460.2022.08.053 ZUO J H, WU B. Microwave coupled Fenton reactor for degradation of phenol wastewater and its process [J]. Contemporary Chemical Industry, 2022, 51(8): 2012-2016(in Chinese). doi: 10.3969/j.issn.1671-0460.2022.08.053
[49] 李光明, 邱珊, 马放. 均相与非均相超声-芬顿催化降解水中罗丹明B [J]. 水处理技术, 2020, 46(5): 19-23. doi: 10.16796/j.cnki.1000-3770.2020.05.004 LI G M, QIU S, MA F. Homogeneous and heterogeneous ultrasound-Fenton for degradation of RhB in water [J]. Technology of Water Treatment, 2020, 46(5): 19-23(in Chinese). doi: 10.16796/j.cnki.1000-3770.2020.05.004
[50] LIU J, WU J Y, KANG C L, et al. Photo-Fenton effect of 4-chlorophenol in ice [J]. Journal of Hazardous Materials, 2013, 261: 500-511. doi: 10.1016/j.jhazmat.2013.07.040 [51] CHEN Y P, YANG L M, PAUL CHEN J, et al. Electrospun spongy zero-valent iron as excellent electro-Fenton catalyst for enhanced sulfathiazole removal by a combination of adsorption and electro-catalytic oxidation [J]. Journal of Hazardous Materials, 2019, 371: 576-585. doi: 10.1016/j.jhazmat.2019.03.043 [52] SUN M, ZOU L, WANG P, et al. Nano valent zero iron (NZVI) immobilized CNTs hollow fiber membrane for flow-through heterogeneous Fenton process [J]. Journal of Environmental Chemical Engineering, 2022, 10(3): 107806. doi: 10.1016/j.jece.2022.107806 [53] XIA Q X, YAO Z P, ZHANG D J, et al. Rational synthesis of micronano dendritic ZVI@Fe3O4 modified with carbon quantum dots and oxygen vacancies for accelerating Fenton-like oxidation [J]. Science of the Total Environment, 2019, 671: 1056-1065. doi: 10.1016/j.scitotenv.2019.03.435 [54] WANG J, LIU C, FENG J, et al. MOFs derived Co/Cu bimetallic nanoparticles embedded in graphitized carbon nanocubes as efficient Fenton catalysts [J]. Journal of Hazardous Materials, 2020, 394: 122567. doi: 10.1016/j.jhazmat.2020.122567 [55] NIU L, ZHAO X, TANG Z, et al. One-Step mechanochemical preparation of magnetic covalent organic framework for the degradation of organic pollutants by heterogeneous and homogeneous Fenton-like synergistic reaction [J]. Separation and Purification Technology, 2022, 294: 121145. doi: 10.1016/j.seppur.2022.121145 [56] CHEN X, TONG X, GAO J B, et al. Simultaneous nitrite resourcing and mercury ion removal using MXene-anchored goethite heterogeneous Fenton composite [J]. Environmental Science & Technology, 2022, 56(7): 4542-4552. -



 下载:
下载: